Abstract
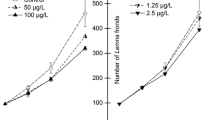
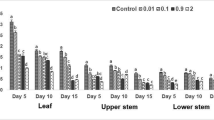
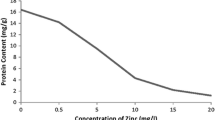
The effects of mercury (Hg), cadmium (Cd) and chromium (Cr) in concentrations ranging from 0.02 to 20 mg L−1 applied for 24 h were assessed in Lemna minor and Lemna gibba by measuring changes in protein concentration, ascorbic acid, phenolics, malondialdehyde (MDA), hydrogen peroxide (H2O2), the activity of guaiacol peroxidase (G-POX) and catalase (CAT). Ascorbic acid, phenolics, catalase and guaiacol peroxidase played a key role in the antioxidative response of L. gibba. Inadequate activity of antioxidant enzymes in the L. minor resulted in MDA and H2O2 accumulation. In both used species, Hg treatment decreased protein content and increased CAT and G-POX activity, but decreased MDA and H2O2 levels. Cadmium and chromium had opposite impacts on two used Lemna species on almost all observed parameters. Enhanced antioxidative responses of L. gibba to lower concentrations of Hg, Cd and Cr indicated greater abiotic stress tolerance than L. minor.
Similar content being viewed by others
References
Dietz K.-J., Krämer U., Baier M., Free radicals and reactive oxygen species as madiators of heavy metal toxicity in plants, In: Prasad M.N.V., Hagemeyer J. (Eds.), Heavy metal stress in plants: From molecules to ecosystems, Springer, Berlin, 1999
Apel K., Hirt H., Reactive oxygen species: metabolism, oxidative stress and signal transduction, Annu. Rev. Plant Biol., 2004, 55, 373–399
Khan N.A., Singh S., Abiotic stress and plant responses, IK International, New Delhi, 2008
Wang B., Peng L., Zhu L., Ren P., Protective effect of total flavonoids from Spirodela polyrrhiza (L.) Schleid on human umbilical vein endothelial cell damage induced by hydrogen peroxide, Colloids Surf. B: Biointerfaces, 2007, 60, 36–40
Michalak A., Phenolic compounds and their antioxidant activity in plants growing under heavy metal stress, Polish J. Environ. Stud., 2006, 115, 523–530
Saqrane S., El Ggazali I., Ouahid Y., El Hassni M., El Haframi I., Bouarab L., et al., Phytotoxic effects of cyanobacteria extract on the aquatic plant Lemna gibba: Microcystin accumulation, detoxication and oxidative stress induction, Aquatic Toxicol., 2007, 83, 284–294
Israr M., Sahi S., Datta R., Sarkar D., Bioaccumulation and physiological effects of mercury in Sesbania drummondii, Chemosphere, 2006, 65, 591–598
Ortega-Villasante C., Rallán-Álvarez R., Del Campo F.F., Carpena-Ruiz R.O., Cellular damage induced by cadmium and mercury in Medicago sativa, J. Exp. Bot., 2005, 56, 2239–2251
Sparks D.L., Toxic metals in the environment: the role of surfaces, Elements, 2005, 1, 193–197
Mendelssohn I.A., McKee K.L., Kong T., A comparison of physiological indicators of sublethal cadmium stress in wetland plants, Environ. Exp. Bot., 2001, 46, 263–275
Schützendübel A., Polle A., Plant response to abiotic stressors: heavy metal-induced oxidative stress and protection by mycorrhization, J. Exp. Bot., 2002, 53, 1351–1365
Tkalec M., Prebeg T., Roje V., Pevalek-Kozlina B., Ljubešić N., Cadmium-induced responses in duckweed Lemna minor L., Acta Physiol. Plant, 2008, 30, 881–890
Cobbett S.C., Phytochelatins and their roles in heavy metal detoxification, Plant Physiol., 2000, 123, 825–832
Salnikow K., Zhitkovich A., Genetic and epigenetic mechanisms in metal carcinogenesis and cocarcinogenesis: nickel, arsenic and chromium, Chem. Res. Toxicol., 2008, 21, 28–44
Sinha S., Saxena R., Singh S., Chromium induced lipid peroxidation in plants of Pistia stratiotes L: role of antioxidants and antioxidant enzymes, Chemosphere, 2005, 58, 595–604
Zell- und stoffwechselphysiologiche Untersuchungen an der Wurzel von Lemna minor unter besonderer Berücksichtigung von Kalliumund Calciummangel. Planta, 1950, 38, 431–473
Bradford M.M., A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding, Anal. Biochem., 1976, 72, 248–254
Siegel B.Z., Galston A.W., The isoperoxidases of Pisum sativum, Plant Physiol., 1967, 42, 221–226
Aebi H., Catalase in vitro, Methods Enzymol., 1984, 105, 121–126
Mukherjee S.P., Choudhuri M.A., Implications of water stress-induced changes in the level of endogenous ascorbic acid and hydrogen peroxide in Vigna seedlings, Physiol. Plantarum, 1983, 58, 166–170
Varma S., Dubey R.S., Lead toxicity induces lipid peroxidation and alters the activities of antioxidant enzymes in growing rice plants, Plant Sci., 2003, 164, 645–655
Liang Y., Lu J., Zhang L., Wu S., Wu Y., Estimation of black tea quality by analysis of chemical composition and colour difference of tea infusions, Food Chem., 2003, 80, 286–290
Sharma S.S., Dietz K.J., The relationship between metal toxicity and cellular redox imbalance, Trends Plant Sci., 2009, 14, 43–50
Panda S.K., Impact of copper on reactive oxygen species, lipid peroxidation and antioxidans in Lemna minor, Biol. Plant., 2008, 52, 561–564
Razinger J., Dermastia M., Drinovec L., Drobne D., Zrimec A., Dolenec Koce J., Antioxidative responses of duckweed (Lemna minor L.) to shortterm copper exposure, Environ. Sci. Pollut. Res. Int., 2007, 14, 194–201
Razinger J., Dermastia M., Dolenc Koce J., Zrimec A., Oxidative stress in duckweed (Lemna minor L.) caused by short term cadmium exposure, Environ. Pollut., 2008, 153, 687–694
Gupta M., Chandra P., Bioaccumulation and toxicity in rooted-submerged macrophyte Vallisneria spiralis, Environ. Pollut., 1998, 103, 327–332
John R., Ahmad P., Gadgil K., Sharma S., Effect of cadmium and lead on growth, biochemical parameters and uptake in Lemna polyrrhiza L., Plant. Soil Environ., 2008, 54, 262–270
Sinha S., Gupta M., Chandra P., Bioaccumulation and biochemical effects of mercury in the plant Bacopa monneri L., Environ. Toxicol. Water Qual., 1996, 11, 105–112
Palma J.M., Sandalio L.M., Corpas F.J., Romero-Puertas M.C., McCarthy I., del Rio L.A., Plant proteases, protein degradation and oxidative stress: role of peroxisomes, Plant. Physiol. Biochem., 2002, 40, 521–530
Gardea-Torresdey J.L., Peralta-Videa J.R., Montes M., de la Rosa G., Corral-Diaz B., Bioaccumulation of cadmium, chromium and copper by Convolvulus arvensis L.: impact on plant growth and uptake of nutritional elements, Biores. Technol., 2004, 92, 229–235
Hou W., Chen X., Song G., Wang Q., Chang C.C., Effects of copper and cadmium on heavy metal polluted waterbody restoration by duckweed (Lemna minor), Plant Physiol. Biochem., 2007, 45, 62–69
Sun W., Van Montagu M., Verbruggen N., Small heat shock proteins and stress tolerance in plants, Biochim. Phys. Acta, 2002, 1577, 1–9
Hall J.L., Cellular mechanisms for heavy metal detoxification and tolerance, J. Exp. Bot., 2002, 53, 1–11
Prado C., Rosa M., Pagano E., Hilal M., Prado F.E., Seasonal variability of physiological and biochemical aspects of chromium accumulation in outdoor-grown Salvinia minima, Chemosphere, 2010, 81, 584–593
Vajpayee P., Tripathi R.D., Rai U.N., Ali M.B., Singh S.N., Chromium (VI) accumulation reduces chlorophyll biosynthesis, nitrate reductase activity and protein content in Nymphaea alba L., Chemosphere, 2000, 41, 1075–1082
Bhattacharjee S., Reactive oxygen species and oxidative burst: roles in stress, senescence and signal transduction in plant, Curr. Sci., 2005, 89, 1113–1121
Foyer C.H., Noctor G., Oxidant and antioxidant signalling in plants: a re-evaluation of the concept of oxidative stress in a physiological context, Plant Cell Environ., 2005, 28, 1056–1071
Uruç Parlak K., Demirezen Yilmaz D., Response of antioxidant defence to Zn stress in three duckweed species, Ecotoxicol. Environ. Saf., 2012, 85, 52–58
Subhadra A.V., Nanda A.K., Bahera P.K., Panda B.B., Acceleration of catalase and peroxidase activities in Lemna minor L. and Allium cepa L. in response to low levels of aquatic mercury, Environ. Pollut., 1991, 69, 169–179
Dhir B., Sharmila P., Pardha Saradhi P., Nasim S.A., Physiological and antioxidant responses of Salvinia natans exposed to chromium-rich wastewater, Ecotoxicol. Environ. Saf., 2009, 72, 1790–1797
Mittler R., Oxidative stress, antioxidants and stress tolerance, Trends Plant Sci., 2002, 7, 405–410
Oláh V., Lakatos G., Bertók C., Kanalas P., Szőllősi E., Kis J., Short-term chromium (VI) stress induces different photosynthetic responses in two duckweed species, Lemna gibba L. and Lemna minor L., Photosynthetica, 2010, 48, 513–520
Gill S.S., Tuteja N., Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants, Plant Physiol. Biochem., 2010, 48, 909–930
Babić M., Radić S., Cvjetko P., Roje V., Pevalek-Kozlina B., Pavlica M., Antioxidative responses of Lemna minor plants exposed to thallium(I)-acetate, Aquat. Bot., 2009, 91, 166–172
Asada K., Takahashi M., Production and scavenging of active oxygen in photosynthesis, In: Kyle D.J., Osmond C.B., Arntzen C.J. (Eds.), Photoinhibition, Elsevier, Amsterdam, 1987
Dai L.P., Xiong Z.T., Huang Y., Li M.J., Cadmium-induced changes in pigments, total phenolics and phenylalanine ammonia-lyase activity in fronds of Azolla imbricate, Environ. Toxicol., 2006, 21, 505–512
Forni C., Braglia R., Harren F.J.M., Cristescu S.M., Stress responses of duckweed (Lemna minor L) and water velvet (Azolla filiculoides Lam.) to anionic surfactant sodium-dodecyl-sulphate (SDS), Aquatic Toxicol., 2012, 110–111, 107–113
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Varga, M., Horvatić, J. & Čelić, A. Short term exposure of Lemna minor and Lemna gibba to mercury, cadmium and chromium. cent.eur.j.biol. 8, 1083–1093 (2013). https://doi.org/10.2478/s11535-013-0238-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11535-013-0238-1