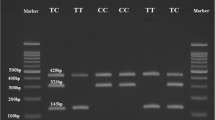
Abstract
Type 1 Diabetes mellitus (T1DM) begins with aberrant inflammatory process followed by auto-destruction in genetically susceptible individuals. Therefore, we hypothesized that gain-of-function allelic variants TNF-α-238A, -308A and PTPN22 1858T could be associated not only with T1DM development but also with the clinical outcome in patients of Bosnia and Herzegovina. A total of 402 subjects were enrolled in the association study. SNPs were determined by PCR-RFLP. Data was analyzed by GraphPad Prism and Sigma Stat 3.5 software. Genotypes frequencies at TNF-α-238 and -308 loci were not statistically different between patients and controls. In contrast, distribution of genotypes at the 1858 position of PTPN22 was significantly different, due to higher frequency of gain-of-function gene variants in patients than controls. Moreover, long term glucose regulation (based on HbA1c level) was significantly worse in patients with the risk TNF-α-308A allele than in patients with non-risk (G) allele. However, patients with the risk allele of both genes (TNF-α-308A and PTPN22 1858T) had the worst glycemic control, suggesting that those two work synergistically. In conclusion, in a cohort from Bosnia and Herzegovina TNF-α-308A allele is significantly associated with the worse long-term glucose control, but PTPN22 1858T allele is significantly associated with diabetes development.
Similar content being viewed by others
References
Hawa M.I., Beyan H., Buckley L.R., Leslie R.D., Impact of genetic and non-genetic factors in type 1 diabetes, Am. J. Med. Genet., 2002, 115, 8–17
Movahedi B., Van de Casteele M., Caluwe N., Stange G., Breckpot K., Thielemans K,. Human pancreatic duct cells can produce tumour necrosis factor-α that damages neighbouring beta cells and activates dendritic cells, Diabetologia, 2004, 47, 998–1008
Christen U., Wolfe T., Mohrle U., Hughes A.C., Rodrigo E., Green E.A., et al., A dual role for TNF-alpha in type 1 diabetes: islet-specific expression abrogates the ongoing autoimmune process when induced late but not early during pathogenesis, J. Immunol., 2001, 166, 7023–7032
Hamaguchi K., Kimura A., Seki N., Higuchi T., Yasunaga S., Takahashi M., Analysis of tumor necrosis factor-alpha promoter polymorphism in type 1 daibetes: HLA-B and -DRB1 alleles are primarly associated with the disease in Japanese, Tissue Antigens, 2000, 55, 10–16
Anderson M.S, Bluestone J.A., The NOD mouse: a model of immune dysregulation, Annu. Rev. Immunol., 2005, 23, 447–485
Vang T., Congia M., Macis M.D., Musumeci L., Orru V., Zavattari P., Autoimmune-associated lymphoid tyrosine phosphatase is a gain-of-function variant, Nat. Genet., 2005, 37, 1317–1319
Bottini N., Vang T., Cucca F., Mustelin T., Role of PTPN22 in type 1 diabetes and other autoimmune diseases, Semin Immunol, 2006, 18, 207–213
Miller S.A., Dykes DD, Polesky H.F., A simple salting out procedure for extracting DNA from human nucleated cells, Nucleic Acids Res., 1988, 16, 1215
Wieser F., Fabjani G., Tempfer C., Schneeberger C., Zeillinger R., Huber C.J., et al., Tumor necrosis factor -alpha promotor polymorphisms and endometriosis, J]_Soc. Gynecol. Investig., 2002, 9, 313–318
Zheng W., She J.X., Genetic association between a lymphoid tyrosine phosphatase (PTPN22) and type 1 diabetes, Diabetes, 2005, 54, 906–908
Hosking L., Lumsden S., Lewis K., Yeo A., McCarthy L., Bansal A., et al., Detection of genotyping errors by Hardy-Weinberg equilibrium testing, Eur. J. Hum. Genet., 2004, 12, 395–399
Gomes I., Collins A., Lonjou C., Thomas N.S., Wilkinson J., Watson M., et al., Hardy-Weinberg quality control, Ann Hum Genet, 1999, 63, 535–538
Wilson A.G., Symons J.A., McDowell T.L., McDevitt H.O., Duff G.W., Effects of a polymorphism in the human tumor necrosis factor alpha promoter on transcriptional activation, Proc. Natl. Acad. Sci. USA, 1997, 94, 3195–3199
Louis E., Franchimont D., Piron A., Gevaert Y., Schaaf-Lafontaine N., Roland S., et al., Tumour necrosis factor (TNF) gene polymorphism influences TNF-alpha production in lipopolysaccharide (LPS)-stimulated whole blood cell culture in healthy humans, Clin. Exp. Immunol., 1998, 113, 401–406
Kaluza W., Reuss E., Grossmann S., Hug R., Schopf R.E., Galle P.R., et al., Different transcriptional activity and in vitro TNF-alpha production in psoriasis patients carrying the TNF-alpha 238A promoter polymorphism, J. Invest. Dermatol., 2000, 114, 1180–1183
Das S.N., Baniasadi V., Kapuria V., Association of -308 TNF-alpha promoter polymorphism with type 1 diabetes in North Indians, Int. J. Immunogenet., 2006, 33, 411–416
Korolija M., Hadzija M., Pape Medvidovic E., Pavkovic P., Kapitanovic S., Pavlic Renar I., et al., Genetic evaluation of the TNF-α-238G/A and -308 G/A promoter polymorphisms in Craotian patients with type 1 diabetes, Hum. Immunl., 2010, 71, 1228–1232
Pérez C., González F.E, Pavez V., Araya A.V., Aguirre A., Cruzat A., et al., The -308 polymorphism in the promoter region of the tumor necrosis factor-alpha (TNF-alpha) gene and ex vivo lipopolysaccharideinduced TNF-alpha expression in patients with aggressive periodontitis and/or type 1 diabetes mellitus, Eur. Cytokine Netw., 2004, 15, 364–370
Korolija M, Renar I.P., Hadzija M., Medvidovic E.P., Pavkovic P., Jokic M., et al., Association of PTPN22 C1858T and CTLA-4 A49G polymorphisms with Type 1 Diabetes in Croatians, Diabetes Res. Clin. Pract., 2009, 86, e54–e57
Peng H., Zhou M., Xu W.D., Xu K., Zhai Y., Li R., et al., Association of PTPN22 C1858T polymorphism and type 1 diabetes: A meta analysis, Immunol. Invest., 2012, 41, 484–496
Nielsen C., Hansen D., Husby S., Lillevang S.T., Sex-specific association of the human PTPN22 1858T allele with type 1 diabetes, Int. J. Immunogenet., 2007, 34, 469–473
Zhernakova A., Eerligh P., Wijmenga C., Barrera P., Roep B.O., Koeleman B.P.C., Differential association of the PTPN22 coding variant with autoimmune diseases in a Dutch population, Genes Immun., 2005, 6, 459–461
Fedetz M., Matesanz F., Caro-Maldonado A., Smirnov I.I., Chvorostinka V.N., Moiseenko T.A., et al., The 1858 PTPN22 gene variant contributes to a genetic risk of type 1 diabetes in a Ukrainian population, Tissue Antigens, 2006, 67, 430–433
Kahles H., Ramos-Lopez E., Lange B., Zwermann O., Reincke M., Sex-specific association of PTPN22 1858T with type 1 diabetes but not woth Hashimoto’sthyroiditis or Addison’s disease in the German population, Eur. J. Endocrinol., 2005, 153, 895–899
Elahi M.M., Asotra K., Matata B.M., Mastana S.S., Tumor necrosis factor alpha-308 gene locus promoter polymorphism: An analysis of association with health and disease, BBA-Mol. Basis Dis., 2009, 1792, 163–172
Wilson A.G., di Giovine F.S., Blakemore A.I., Duff G.W., Single base polymorphism in the human tumour necrosis factor alpha gene detectable by NcoI restriction of PCR product, Hum. Mol. Genet., 1992, 1, 353
Laki J., Kiszel P., Blasko B., Kovaca B.B., Korner A., Madacsy L., et al., The HLA 8.1 ancestral haplotype is strongly linked to the C allele of −429T>C promoter polymorphism of receptor of the advanced glycation endproduct (RAGE) gene. Haplotype-independent association of the −429C allele with high hemoglobin(A1C) levels in diabetic patients, Mol. Immunol., 2006, 44, 648–655
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Hadžija, M.P., Korolija, M., Vukadinović, G. et al. Association of TNF-α and PTPN22 SNPs with the risk and clinical outcome of type 1 diabetes. cent.eur.j.biol. 8, 513–519 (2013). https://doi.org/10.2478/s11535-013-0166-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11535-013-0166-5