Abstract
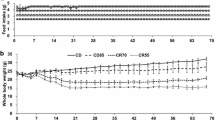
It is generally accepted that the location of body fat deposits may play an important role in the risk of developing some endocrine and metabolic diseases. We have studied the effect of food restriction and food restriction/refeeding, often practiced by individuals trying to lose body weight, on the expression of genes which are associated with obesity and certain metabolic disorders in inguinal, epididymal, and perirenal rat white adipose tissues. Gene expression was analyzed by real time semi-quantitative polymerase chain reaction and by Western blot. We found that prolonged food restriction caused a significant decrease of body and adipose tissue mass as well as the increase of Scd1 and Elovl6 gene expressions in all main rat adipose tissue deposits. Food restriction/refeeding caused increases of: a) Scd1 and Elovl6 mRNA levels in adipose tissue, b) Scd1 protein level and c) desaturation index in adipose tissue. The increased expression of both genes was unusually high in inguinal adipose tissue. The results suggest that the increase of Scd1 and Elovl6 gene expressions in white adipose tissue by prolonged food restriction and prolonged food restriction/refeeding may contribute to accelerated fat recovery that often occurs in individuals after food restriction/refeeding.
Similar content being viewed by others
References
Flowers M.T., Ntambi J.M., Role of stearoylcoenzyme A desaturase in regulating lipid metabolism, Curr. Opin. Lipidol., 2008, 19, 248–256
Ntambi J.M., Miyazaki M., Regulation of stearoyl-CoA desaturases and role in metabolism, Prog. Lipid Res., 2004, 43, 91–104
Miyazaki M., Ntambi J.M., Role of stearoylcoenzyme A desaturase in lipid metabolism, Prostaglandins Leukot. Essent. Fatty Acids, 2003, 68, 113–121
Scaglia N., Chisholm J.W., Igal R.A., Inhibition of stearoyl-CoA desaturase-1 inactivates acetyl-CoA carboxylase and impairs proliferation in cancer cells: role of AMPK, PLoS One, 2009, 4, e6812
Kaestner K.H., Ntambi J.M., Kelly T.J. Jr., Lane M.D., Differentiation-induced gene expression in 3T3-L1 preadipocytes. A second differentially expressed gene encoding stearoyl-CoA desaturase, J. Biol. Chem., 1989, 264, 14755–14761
Mihara K., Structure and regulation of rat liver microsomal stearoyl-CoA desaturase gene, J. Biochem., 1990, 108, 1022–1029
Cohen P., Miyazaki M., Socci N.D., Hagge-Greenberg A., Liedtke W., Soukas A.A., et al., Role for stearoyl-CoA desaturase-1 in leptin-mediated weight loss, Science, 2002, 297, 240–243
Dobrzyn A., Ntambi J.M., Stearoyl-CoA desaturase as a new drug target for obesity treatment, Obes. Rev., 2005, 6, 169–174
Dobrzyn A., Ntambi J.M., Stearoyl-CoA desaturase: A therapeutic target of insulin resistance and diabetes, Drug Discov. Today Ther. Strateg., 2005, 2, 125–128
Dobrzyn P., Jazurek M., Dobrzyn A., Stearoyl-CoA desaturase and insulin signaling — What is the molecular switch?, Biochim. Biophys. Acta, 2010, 1797, 1189–1194
Ntambi J.M., Dietary regulation of stearoyl-CoA desaturase 1 gene expression in mouse liver, J. Biol. Chem., 1992, 267, 10925–10930
Matsuzaka T., Shimano H., Yahagi N., Yoshikawa T., Amemiya-Kudo M., Hasty A.H., et al., Cloning and characterization of a mammalian fatty acyl-CoA elongase as a lipogenic enzyme regulated by SREBPs, J. Lipid Res., 2002, 43, 911–920
American Diabetes Association, Nutrition recommendations and principles for people with diabetes mellitus, Diabetes Care, 2000, 23Suppl.1, S43–S46
Mainieri D., Summermatter S., Seydoux J., Montani J.P., Rusconi S., Russell A.P., et al., A role for skeletal muscle stearoyl-CoA desaturase 1 in control of thermogenesis, FASEB J., 2006, 20, 1751–1753
Turyn J., Stojek M., Swierczynski J., Up-regulation of stearoyl-CoA desaturase 1 and elongase 6 genes expression in rat lipogenic tissues by chronic food restriction and chronic food restriction/refeeding, Mol. Cell. Biochem., 2010, 345, 171–179
Chomczynski P., Sacchi N., Single-step method of RNA isolation by acid guanidinium thiocyanatephenol-chloroform extraction, Anal. Biochem., 1987, 162, 156–159
Folch J., Lees M., Sloane Stanley G.H., A simple method for the isolation and purification of total lipides from animal tissues, J. Biol. Chem., 1957, 226, 497–509
Wang Y., Botolin D., Christian B., Busik J., Xu J., Jump D.B., Tissue-specific, nutritional, and developmental regulation of rat fatty acid elongases, J. Lipid Res., 2005, 46, 706–715
Atzmon G., Yang X.M., Muzumdar R., Ma X.H., Gabriely I., Barzilai N., Differential gene expression between visceral and subcutaneous fat depots, Horm. Metab. Res., 2002, 34, 622–628
Dusserre E., Moulin P., Vidal H., Differences in mRNA expression of the proteins secreted by the adipocytes in human subcutaneous and visceral adipose tissues, Biochim. Biophys. Acta, 2000, 1500, 88–96
Lefebvre A.M., Laville M., Vega N., Riou J.P., van Gaal L., Auwerx J., et al., Depot-specific differences in adipose tissue gene expression in lean and obese subjects, Diabetes, 1998, 47, 98–103
Linder K., Arner P., Flores-Morales A., Tollet-Egnell P., Norstedt G., Differentially expressed genes in visceral or subcutaneous adipose tissue of obese men and women, J. Lipid Res., 2004, 45, 148–154
Montague C.T., Prins J.B., Sanders L., Digby J.E., O’Rahilly S., Depot- and sex-specific differences in human leptin mRNA expression: implications for the control of regional fat distribution, Diabetes, 1997, 46, 342–347
Stelmanska E., Sucajtys-Szulc E., Korczynska J., Adrych K., Swierczynski J., Diversity of SREBP-1 gene expression in rat adipose tissue depots in response to refeeding after food restriction, Biochim. Biophys. Acta, 2005, 1733, 130–136
Summermatter S., Marcelino H., Arsenijevic D., Buchala A., Aprikian O., Assimacopoulos-Jeannet F., et al., Adipose tissue plasticity during catchup fat driven by thrifty metabolism: relevance for muscle-adipose glucose redistribution during catch-up growth, Diabetes, 2009, 58, 2228–2237
Bruss M.D., Khambatta C.F., Ruby M.A., Aggarwal I., Hellerstein M.K., Calorie restriction increases fatty acid synthesis and whole body fat oxidation rates, Am. J. Physiol. Endocrinol. Metab., 2010, 298, E108–E116
Hulver M.W., Berggren J.R., Carper M.J., Miyazaki M., Ntambi J.M., Hoffman E.P., et al., Elevated stearoyl-CoA desaturase-1 expression in skeletal muscle contributes to abnormal fatty acid partitioning in obese humans, Cell Metab., 2005, 2, 251–261
Green C.D., Olson L.K., Modulation of palmitateinduced endoplasmic reticulum stress and apoptosis in pancreatic β-cells by stearoyl-CoA desaturase and Elovl6, Am. J. Physiol. Endocrinol. Metab., 2011, 300, E640–E649
Wahrenberg H., Lonnqvist F., Arner P., Mechanisms underlying regional differences in lipolysis in human adipose tissue, J. Clin. Invest., 1989, 84, 458–467
Richelsen B., Pedersen S.B., Moller-Pedersen T., Bak J.F., Regional differences in triglyceride breakdown in human adipose tissue: effects of catecholamines, insulin, and prostaglandin E2, Metabolism, 1991, 40, 990–996
Bjorntorp P., Metabolic implications of body fat distribution, Diabetes Care, 1991, 14, 1132–1143
Kannel W.B., Cupples L.A., Ramaswami R., Stokes J. III, Kreger B.E., Higgins M., Regional obesity and risk of cardiovascular disease; the Framingham Study, J. Clin. Epidemiol., 1991, 44, 183–190
Kissebah A.H., Krakower G.R., Regional adiposity and morbidity, Physiol. Rev., 1994, 74, 761–811
Krotkiewski M., Bjorntorp P., Sjostrom L., Smith U., Impact of obesity on metabolism in men and women. Importance of regional adipose tissue distribution, J. Clin. Invest., 1983, 72, 1150–1162
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Turyn, J., Mika, A., Stepnowski, P. et al. Unusual increase of Scd1 and Elovl6 expression in rat inguinal adipose tissue. cent.eur.j.biol. 7, 192–200 (2012). https://doi.org/10.2478/s11535-012-0007-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11535-012-0007-6