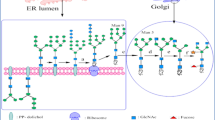
Abstract
A host of bacteria and viruses are dependent on O-linked and N-linked glycosylation to perform vital biological functions. Pathogens often have integral proteins that participate in host-cell interactions such as receptor binding and fusion with host membrane. Fusion proteins from a broad range of disparate viruses, such as paramyxovirus, HIV, ebola, and the influenza viruses share a variety of common features that are augmented by glycosylation. Each of these viruses contain multiple glycosylation sites that must be processed and modified by the host post-translational machinery to be fusogenically active. In most viruses, glycosylation plays a role in biogenesis, stability, antigenicity and infectivity. In bacteria, glycosylation events play an important role in the formation of flagellin and pili and are vitally important to adherence, attachment, infectivity and immune evasion. With the importance of glycosylation to pathogen survival, it is clear that a better understanding of the processes is needed to understand the pathogen requirement for glycosylation and to capitalize on this requirement for the development of novel therapeutics.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Witze E.S., Old W.M., Resing K.A., Ahn N.G., Mapping protein post-translational modifications with mass spectrometry, Nat. Methods, 2007, 4, 798–806
Spiro R.G., Protein glycosylation: nature, distribution, enzymatic formation, and disease implications of glycopeptide bonds, Glycobiology, 2002, 12, 43R–56R
Stepper J., Shastri S., Loo T.S., Preston J.C., Novak P., Man P., et al., Cysteine S-glycosylation, a new post-translational modification found in glycopeptide bacteriocins, FEBS letters, 585, 645–650
Furmanek A., Hofsteenge J., Protein C-mannosylation: facts and questions, Acta Biochim. Polon., 2000, 47, 781–789
Falzarano D., Krokhin O., Van Domselaar G., Wolf K., Seebach J., Schnittler H.J., et al., Ebola sGP—the first viral glycoprotein shown to be C-mannosylated, Virology, 2007, 368, 83–90
Hartmann S., Hofsteenge J., Properdin, the positive regulator of complement, is highly C-mannosylated, J. Biol. Chem., 2000, 275, 28569–72854
Ristl R., Steiner K., Zarschler K., Zayni S., Messner P., Schaffer C., The s-layer glycome-adding to the sugar coat of bacteria, Int. J. Microbiol., 2011, pi 127870
Apweiler R., Hermjakob H., Sharon N., On the frequency of protein glycosylation, as deduced from analysis of the SWISS-PROT database, Biochim. Biophys. Acta, 1999, 1473, 4–8
Messner P., Prokaryotic glycoproteins: unexplored but important, J. Bacteriol., 2004, 186, 2517–2519
Weerapana E., Imperiali B., Asparagine-linked protein glycosylation: from eukaryotic to prokaryotic systems, Glycobiology, 2006, 16, 91R–101R
Neuberger A., Carbohydrates in protein: The carbohydrate component of crystalline egg albumin, Biochem. J., 1938, 32, 1435–1451
Sleytr, U. B. and Thorne, K. J. (1976) Chemical characterization of the regularly arranged surface layers of Clostridium thermosaccharolyticum and Clostridium thermohydrosulfuricum. J Bacteriol 126, 377–83.
Sleytr U.B., Heterologous reattachment of regular arrays of glycoproteins on bacterial surfaces, Nature, 1975, 257, 400–402
Szymanski C.M., Logan S.M., Linton D., Wren B.W., Campylobacter—a tale of two protein glycosylation systems, Trends Microbiol., 2003, 11, 233–238
Wacker M., Linton D., Hitchen P.G., Nita-Lazar M., Haslam S.M., North S.J., et al., N-linked glycosylation in Campylobacter jejuni and its functional transfer into E. coli, Science, 2002, 298, 1790–1793
Abu-Qarn M., Eichler J., Protein N-glycosylation in Archaea: defining Haloferax volcanii genes involved in S-layer glycoprotein glycosylation, Mol. Microbiol., 2006, 61, 511–525
Logan S.M., Flagellar glycosylation — a new component of the motility repertoire?, Microbiology, 2006, 152, 1249–1262
Castric P., pilO, a gene required for glycosylation of Pseudomonas aeruginosa 1244 pilin, Microbiology, 1995, 141, 1247–1254
Fletcher C.M., Coyne M.J., Villa O.F., Chatzidaki-Livanis M., Comstock L.E. A general O-glycosylation system important to the physiology of a major human intestinal symbiont, Cell, 2009, 137, 321–331
Coyne M.J., Reinap B., Lee M.M., Comstock L.E., Human symbionts use a host-like pathway for surface fucosylation, Science, 2005, 307, 1778–1781
Power P.M., Seib K.L., Jennings M.P., Pilin glycosylation in Neisseria meningitidis occurs by a similar pathway to wzy-dependent O-antigen biosynthesis in Escherichia coli, Biochem. Biophys. Res. Commun., 2006, 347, 904–908
Stimson E., Virji M., Makepeace K., Dell A., Morris H., Payne G., et al., Meningococcal pilin: a glycoprotein substituted with digalactosyl 2,4-diacetamido-2,4,6-trideoxyhexose, Mol. Microbiol., 1995, 17, 1201–1214
Virji M., Post-translational modifications of meningococcal pili. Identification of common substituents: glycans and alpha-glycerophosphate—a review, Gene, 1997, 192, 141–147
Hegge F.T., Hitchen P.G., Aas F.E., Kristiansen H., Lovold C., Egge-Jacobsen W., Unique modifications with phosphocholine and phosphoethanolamine define alternate antigenic forms of Neisseria gonorrhoeae type IV pili, Proc. Natl. Acad. Sci. USA, 2004, 101, 10798–107803
Castric P., Cassels F.J., Carlson R.W., Structural characterization of the Pseudomonas aeruginosa 1244 pilin glycan, J. Biol. Chem., 2001, 276, 26479–26485
Schirm M., Soo E.C., Aubry A.J., Austin J., Thibault P., Logan S.M., Structural, genetic and functional characterization of the flagellin glycosylation process in Helicobacter pylori, Mol. Microbiol., 2003, 48, 1579–1592
McNally D.J., Aubry A.J., Hui J.P., Khieu N.H., Whitfield D., Ewing C.P., et al., Targeted metabolomics analysis of Campylobacter coli VC167 reveals legionaminic acid derivatives as novel flagellar glycans, J. Biol. Chem., 2007, 282, 14463–14475
Thibault P., Logan S.M., Kelly J.F., Brisson J.R., Ewing C.P., Trust T.J., et al., Identification of the carbohydrate moieties and glycosylation motifs in Campylobacter jejuni flagellin, J. Biol. Chem., 2001, 276, 34862–34870
Sambri V., Stefanelli C., Cevenini R., Detection of glycoproteins in Borrelia burgdorferi, Arch. Microbiol., 1992, 157, 205–208
Swanson A.F., Kuo C.C., Binding of the glycan of the major outer membrane protein of Chlamydia trachomatis to HeLa cells, Infect. Immun., 1994, 62, 24–28
Swanson A.F., Kuo C.C., The characterization of lectin-binding proteins of Chlamydia trachomatis as glycoproteins, Microb. Pathog., 1991, 10, 465–473
Swanson A.F., Kuo C.C., Evidence that the major outer membrane protein of Chlamydia trachomatis is glycosylated, Infect. Immun., 1991, 59, 2120–2125
Lindenthal C., Elsinghorst E.A., Enterotoxigenic Escherichia coli TibA glycoprotein adheres to human intestine epithelial cells, Infect. Immun., 2001, 69, 52–57
Lindenthal C., Elsinghorst E.A., Identification of a glycoprotein produced by enterotoxigenic Escherichia coli, Infect. Immun., 1999, 67, 4084–4091
Espitia C., Mancilla R., Identification, isolation and partial characterization of Mycobacterium tuberculosis glycoprotein antigens, Clin. Exp. Immunol., 1989, 77, 378–383
Mondotte J.A., Lozach P.Y., Amara A., Gamarnik A.V., Essential role of dengue virus envelope protein N glycosylation at asparagine-67 during viral propagation, J. Virol., 2007, 81, 7136–7148
Korber B., Gaschen B., Yusim K., Thakallapally R., Kesmir C., Detours V., Evolutionary and immunological implications of contemporary HIV-1 variation, Br. Med. Bull., 2001, 58, 19–42
Wei X., Decker J.M., Wang S., Hui H., Kappes J.C., Wu X., et al., Antibody neutralization and escape by HIV-1, Nature, 2003, 422, 307–312
Land A., Braakman I., Folding of the human immunodeficiency virus type 1 envelope glycoprotein in the endoplasmic reticulum, Biochimie, 2001, 83, 783–790
Slater-Handshy T., Droll D.A., Fan X., Di Bisceglie A.M., Chambers T.J., HCV E2 glycoprotein: mutagenesis of N-linked glycosylation sites and its effects on E2 expression and processing, Virology, 2004, 319, 36–48
Meunier J.C., Fournillier A., Choukhi A., Cahour A., Cocquerel L., Dubuisson J., et al., Analysis of the glycosylation sites of hepatitis C virus (HCV) glycoprotein E1 and the influence of E1 glycans on the formation of the HCV glycoprotein complex, J. Gen. Virol., 1999, 80, 887–896
Wagner R., Wolff T., Herwig A., Pleschka S., Klenk H.D., Interdependence of hemagglutinin glycosylation and neuraminidase as regulators of influenza virus growth: a study by reverse genetics, J. Virol., 2000, 74, 6316–6323
Klenk H.D., Wagner R., Heuer D., Wolff T., Importance of hemagglutinin glycosylation for the biological functions of influenza virus, Virus Res., 2002, 82, 73–75
Baigent S.J., McCauley J.W., Glycosylation of haemagglutinin and stalk-length of neuraminidase combine to regulate the growth of avian influenza viruses in tissue culture, Virus Res., 2001, 79, 177–185
Tsuchiya E., Sugawara K., Hongo S., Matsuzaki Y., Muraki Y., Nakamura K., Role of overlapping glycosylation sequons in antigenic properties, intracellular transport and biological activities of influenza A/H2N2 virus haemagglutinin, J. Gen. Virol., 2002, 83, 3067–3074
Tsuchiya E., Sugawara K., Hongo S., Matsuzaki Y., Muraki Y., Li Z.N., et al., Effect of addition of new oligosaccharide chains to the globular head of influenza A/H2N2 virus haemagglutinin on the intracellular transport and biological activities of the molecule, J. Gen. Virol., 2002, 83, 1137–1146
Abe Y., Takashita E., Sugawara K., Matsuzaki Y., Muraki Y., Hongo S., Effect of the addition of oligosaccharides on the biological activities and antigenicity of influenza A/H3N2 virus hemagglutinin, J. Virol., 2004, 78, 9605–9611
Kaverin N.V., Rudneva I.A., Ilyushina N.A., Varich N.L., Lipatov A.S., Smirnov Y.A., et al., Structure of antigenic sites on the haemagglutinin molecule of H5 avian influenza virus and phenotypic variation of escape mutants, J. Gen. Virol., 2002, 83, 2497–2505
Londrigan S.L., Turville S.G., Tate M.D., Deng Y.M., Brooks A.G., Reading P.C., N-linked Glycosylation Facilitates Sialic Acid-Independent Attachment and Entry of Influenza A Viruses into Cells Expressing DC-SIGN or L-SIGN, J. Virol., 2010, 85, 2990–3000
Daniels R., Kurowski B., Johnson A.E., Hebert D.N., N-linked glycans direct the cotranslational folding pathway of influenza hemagglutinin, Mol. Cell, 2003, 11, 79–90
Skehel J.J., Wiley D.C., Receptor binding and membrane fusion in virus entry: the influenza hemagglutinin, Ann. Rev. Biochem., 2000, 69, 531–569
Vigerust D.J., Ulett K.B., Boyd K.L., Madsen J., Hawgood S., McCullers J.A., N-linked glycosylation attenuates H3N2 influenza viruses, J. Virol., 2007, 81, 8593–8600
Job E.R., Deng Y.M., Tate M.D., Bottazzi B., Crouch E.C., Dean M.M., et al., Pandemic H1N1 influenza A viruses are resistant to the antiviral activities of innate immune proteins of the collectin and pentraxin superfamilies, J. Immunol., 2010, 185, 4284–4291
Deshpande K.L., Fried V.A., Ando M., Webster R.G., Glycosylation affects cleavage of an H5N2 influenza virus hemagglutinin and regulates virulence, Proc. Natl. Acad. Sci. USA, 1987, 84, 36–40
Zambon M.C., Epidemiology and pathogenesis of influenza, J. Antimicrob. Chemother., 1999, 44, 3–9
Das S.R., Puigbo P., Hensley S.E., Hurt D.E., Bennink J.R., Yewdell J.W., Glycosylation focuses sequence variation in the influenza A virus H1 hemagglutinin globular domain, PLoS Pathog., 2010, 6, e1001211
Martin-Gallardo A., Fleischer E., Doyle S.A., Arumugham R., Collins P.L., Hildreth S.W., et al., Expression of the G glycoprotein gene of human respiratory syncytial virus in Salmonella typhimurium, J. Gen. Virol., 1993, 74, 453–458
Collins P.L., Mottet G., Oligomerization and posttranslational processing of glycoprotein G of human respiratory syncytial virus: altered O-glycosylation in the presence of brefeldin A, J. Gen. Virol., 1992, 73, 849–863
Aguilar H.C., Matreyek K.A., Filone C.M., Hashimi S.T., Levroney E.L., Negrete O.A., et al., N-glycans on Nipah virus fusion protein protect against neutralization but reduce membrane fusion and viral entry, J. Virol., 2006, 80, 4878–4889
Moll M., Kaufmann A., Maisner A., Influence of N-glycans on processing and biological activity of the nipah virus fusion protein, J. Virol., 2004, 78, 7274–7278
Bossart K.N., Crameri G., Dimitrov A.S., Mungall B.A., Feng Y.R., Patch J.R., et al., Receptor binding, fusion inhibition, and induction of crossreactive neutralizing antibodies by a soluble G glycoprotein of Hendra virus, J. Virol., 2005, 79, 6690–6702
Oostra M., de Haan C.A., de Groot R.J., Rottier P.J., Glycosylation of the severe acute respiratory syndrome coronavirus triple-spanning membrane proteins 3a and M., J. Virol., 2006, 80, 2326–2336
Schowalter R.M., Smith S.E., Dutch R.E., Characterization of Human Metapneumovirus F Protein-Promoted Membrane Fusion: Critical Roles for Proteolytic Processing and Low pH, J. Virol., 2006, 80, 10931–10941
Bowden T.A., Crispin M., Harvey D.J., Jones E.Y., Stuart D.I., Dimeric architecture of the Hendra virus attachment glycoprotein: evidence for a conserved mode of assembly, J. Virol., 2010, 84, 6208–6217
Bowden T.A., Crispin M., Harvey D.J., Aricescu A.R., Grimes J.M., Jones E.Y., et al., Crystal structure and carbohydrate analysis of Nipah virus attachment glycoprotein: a template for antiviral and vaccine design, J. Virol., 2008, 82, 11628–11636
Botarelli P., Houlden B.A., Haigwood N.L., Servis C., Montagna D., Abrignani S., N-glycosylation of HIV-gp120 may constrain recognition by T lymphocytes, J. Immunol., 1991, 147, 3128–3132
Zhu, X., Borchers, C., Bienstock, R. J., Tomer, K. B. (2000) Mass spectrometric characterization of the glycosylation pattern of HIV-gp120 expressed in CHO cells. Biochemistry 39, 11194–204.
Doores K.J., Bonomelli C., Harvey D.J., Vasiljevic S., Dwek R.A., Burton D.R., et al., Envelope glycans of immunodeficiency virions are almost entirely oligomannose antigens, Proc. Natl. Acad. Sci. USA, 2010, 107, 13800–13805
Fenouillet E., Gluckman J.C., Bahraoui E., Role of N-linked glycans of envelope glycoproteins in infectivity of human immunodeficiency virus type 1, J. Virol., 1990, 64, 2841–2848
Montefiori D.C., Robinson W.E. Jr., Mitchell W.M., Role of protein N-glycosylation in pathogenesis of human immunodeficiency virus type 1, Proc. Natl. Acad. Sci. USA, 1988, 85, 9248–9252
Wyatt R., Kwong P.D., Desjardins E., Sweet R.W., Robinson J., Hendrickson W.A., et al., The antigenic structure of the HIV gp120 envelope glycoprotein, Nature, 1998, 393, 705–711
Poumbourios P., Maerz A.L., Drummer H.E., Functional evolution of the HIV-1 envelope glycoprotein 120 association site of glycoprotein 41, J. Biol. Chem., 2003, 278, 42149–42160
Bunnik E.M., Euler Z., Welkers M.R., Boeser-Nunnink B.D., Grijsen M.L., Prins J.M., et al., Adaptation of HIV-1 envelope gp120 to humoral immunity at a population level, Nat. Med., 2010, 16, 995–997
Chaillon A., Braibant M., Moreau T., Thenin S., Moreau A., Autran B., et al., The V1V2 domain and a N-linked glycosylation site in the V3 loop of the HIV-1 envelope glycoprotein modulate neutralization sensitivity to the human broadly neutralizing antibody 2G12, J. Virol., 2011, 85, 3642–3648
Sanders R.W., Venturi M., Schiffner L., Kalyanaraman R., Katinger H., Lloyd K.O., et al., The mannose-dependent epitope for neutralizing antibody 2G12 on human immunodeficiency virus type 1 glycoprotein gp120, J. Virol., 2002, 76, 7293–7305
Calarese D.A., Lee H.K., Huang C.Y., Best M.D., Astronomo R.D., Stanfield R.L., et al., Dissection of the carbohydrate specificity of the broadly neutralizing anti-HIV-1 antibody 2G12, Proc. Natl. Acad. Sci. USA, 2005, 102, 13372–13377
Calarese D.A., Scanlan C.N., Zwick M.B., Deechongkit S., Mimura Y., Kunert R., et al., Antibody domain exchange is an immunological solution to carbohydrate cluster recognition, Science, 2003, 300, 2065–2071
Lai J., Bernhard O.K., Turville S.G., Harman A.N., Wilkinson J., Cunningham A.L., Oligomerization of the macrophage mannose receptor enhances gp120-mediated binding of HIV-1, J. Biol. Chem., 2009, 284, 11027–110238
Frost S.D., Wrin T., Smith D.M., Kosakovsky Pond S.L., Liu Y., Paxinos E., et al., Neutralizing antibody responses drive the evolution of human immunodeficiency virus type 1 enveloped
Bonhomme C.J., Capul A.A., Lauron E.J., Bederka L.H., Knopp K.A., Buchmeier M.J. Glycosylation modulates arenavirus glycoprotein expression and function, Virology, 2011, 409, 223–233
Feldmann H., Nichol S.T., Klenk H.D., Peters C.J., Sanchez A., Characterization of filoviruses based on differences in structure and antigenicity of the virion glycoprotein, Virology, 1994, 199, 469–473
Simmons G., Wool-Lewis R.J., Baribaud F., Netter R.C., Bates P., Ebola virus glycoproteins induce global surface protein down-modulation and loss of cell adherence, J. Virol., 2002, 76, 2518–2528
Dowling W., Thompson E., Badger C., Mellquist J.L., Garrison A.R., Smith J.M., et al., Influences of glycosylation on antigenicity, immunogenicity, and protective efficacy of ebola virus GP DNA vaccines, J. Virol., 2007, 81, 1821–1837
Shi X., Elliott R.M., Analysis of N-linked glycosylation of hantaan virus glycoproteins and the role of oligosaccharide side chains in protein folding and intracellular trafficking, J. Virol., 2004, 78, 5414–5422
Zheng F., Ma L., Shao L., Wang G., Chen F., Zhang Y., et al., Defining the N-linked glycosylation site of Hantaan virus envelope glycoproteins essential for cell fusion, J. Microbiol., 2007, 45, 41–47
Robinson M.J., Sancho D., Slack E.C., LeibundGut-Landmann S., Reis e Sousa C., Myeloid C-type lectins in innate immunity, Nat. Immunol., 2006, 7, 1258–1265
Weis W.I., Taylor M.E., Drickamer K., The C-type lectin superfamily in the immune system, Immunol. Rev., 1998, 163, 19–34
Cambi A., de Lange F., van Maarseveen N.M., Nijhuis M., Joosten B., van Dijk E.M., et al., Microdomains of the C-type lectin DC-SIGN are portals for virus entry into dendritic cells, J. Cell Biol., 2004, 64, 145–155
Cambi A., Figdor C.G., Dual function of C-type lectin-like receptors in the immune system, Curr. Opin. Cell Biol., 2003, 15, 539–546
Geijtenbeek T.B., van Vliet S.J., Engering A., Hart B.A., van Kooyk Y., Self- and nonself-recognition by C-type lectins on dendritic cells, Annu. Rev. Immunol., 2004, 22, 33–54
Rogers N.C., Slack E.C., Edwards A.D., Nolte M.A., Schulz O., Schweighoffer E., et al., Sykdependent cytokine induction by Dectin-1 reveals a novel pattern recognition pathway for C type lectins, Immunity, 2005, 22, 507–517
Meyer-Wentrup F., Cambi A., Adema G.J., Figdor C.G., “Sweet talk”: closing in on C type lectin signaling, Immunity, 2005, 22, 399–400
Upham J.P., Pickett D., Irimura T., Anders E.M., Reading P.C., Macrophage receptors for influenza A virus: role of the macrophage galactose-type lectin and mannose receptor in viral entry, J. Virol., 2010, 84, 3730–3737
Reading P.C., Miller J.L., Anders E.M., Involvement of the mannose receptor in infection of macrophages by influenza virus, J. Virol., 2000, 74, 5190–5197
Nguyen D.G., Hildreth J.E., Involvement of macrophage mannose receptor in the binding and transmission of HIV by macrophages, Eur. J. Immunol., 2003, 33, 483–493
Vigerust D.J., Egan B.S., Shepherd V.L., HIV-1 Nef mediates post-translational down-regulation and redistribution of the mannose receptor, J. Leukoc. Biol., 2005, 77, 522–534
Reiling N., Blumenthal A., Flad H.D., Ernst M., Ehlers S., Mycobacteria-induced TNF-alpha and IL-10 formation by human macrophages is differentially regulated at the level of mitogenactivated protein kinase activity, J. Immunol., 2001, 167, 3339–3345
Bergman M., Del Prete G., van Kooyk Y., Appelmelk B., Helicobacter pylori phase variation, immune modulation and gastric autoimmunity, Nat. Rev. Microbiol., 2006, 4, 151–159
Cocquerel L., Voisset C., Dubuisson J., Hepatitis C virus entry: potential receptors and their biological functions, J. Gen. Virol., 2006, 87, 1075–1084
Lozach P.Y., Lortat-Jacob H., de Lacroix de Lavalette A., Staropoli I., Foung S., Amara, A., et al., DC-SIGN and L-SIGN are high affinity binding receptors for hepatitis C virus glycoprotein E2, J. Biol. Chem., 2003, 278, 20358–20366
Martina B.E., Koraka P., van den Doel P., Rimmelzwaan G.F., Haagmans B.L., Osterhaus A.D., DC-SIGN enhances infection of cells with glycosylated West Nile virus in vitro and virus replication in human dendritic cells induces production of IFN-alpha and TNF-alpha, Virus Res., 2008, 135, 64–71
Hacker K., White L., de Silva A.M., N-linked glycans on dengue viruses grown in mammalian and insect cells, J. Gen. Virol., 2009, 90, 2097–2106
Davis C.W., Mattei L.M., Nguyen H.Y., Ansarah-Sobrinho C., Doms R.W., Pierson T.C., The location of asparagine-linked glycans on West Nile virions controls their interactions with CD209 (DCSIGN), J. Biol. Chem., 2006, 281, 37183–37194
Davis C.W., Nguyen H.Y., Hanna S.L., Sanchez M.D., Doms R.W., Pierson T.C., West Nile virus discriminates between DC-SIGN and DC-SIGNR for cellular attachment and infection, J. Virol., 2006, 80, 1290–1301
Lin G., Simmons G., Pohlmann S., Baribaud F., Ni H., Leslie G.J., et al., Differential N-linked glycosylation of human immunodeficiency virus and Ebola virus envelope glycoproteins modulates interactions with DC-SIGN and DC-SIGNR, J. Virol., 2003, 77, 1337–1346
Turville S.G., Arthos J., Donald K.M., Lynch G., Naif H., Clark G., et al., HIV gp120 receptors on human dendritic cells, Blood, 2001, 98, 2482–2488
Liu Y., Liu H., Kim B.O., Gattone V.H., Li J., Nath A., et al., CD4-independent infection of astrocytes by human immunodeficiency virus type 1: requirement for the human mannose receptor, J. Virol., 2004, 78, 4120–4133
Gramberg T., Hofmann H., Moller P., Lalor P.F., Marzi A., Geier M., et al., LSECtin interacts with filovirus glycoproteins and the spike protein of SARS coronavirus, Virology, 2005, 340, 224–236
Pipirou Z., Powlesland A.S., Steffen I., Pohlmann S., Taylor M.E., Drickamer K., Mouse LSECtin as a model for a human Ebola virus receptor, Glycobiology, 2011, 21, 806–812
Li Y., Hao B., Kuai X., Xing G., Yang J., Chen J., et al., C-type lectin LSECtin interacts with DCSIGNR and is involved in hepatitis C virus binding, Mol. Cell. Biochem., 2009, 327, 183–190
Francois K.O., Balzarini J., Potential of carbohydrate-binding agents as therapeutics against enveloped viruses, Med. Res. Rev., 2010, 31
Witvrouw M., Fikkert V., Hantson A., Pannecouque C., O’Keefe B.R., McMahon J., et al., Resistance of human immunodeficiency virus type 1 to the high-mannose binding agents cyanovirin N and concanavalin A, J. Virol., 2005, 79, 7777–7784
Williams D.C. Jr., Lee J.Y., Cai M., Bewley C.A., Clore G.M., Crystal structures of the HIV-1 inhibitory cyanobacterial protein MVL free and bound to Man3GlcNAc2: structural basis for specificity and high-affinity binding to the core pentasaccharide from n-linked oligomannoside, J. Biol. Chem., 2005, 280, 29269–29276
Balzarini J., Van Herrewege Y., Vermeire K., Vanham G., Schols D., Carbohydrate-binding agents efficiently prevent dendritic cell-specific intercellular adhesion molecule-3-grabbing nonintegrin (DC-SIGN)-directed HIV-1 transmission to T lymphocytes, Mol. Pharmacol., 2007, 71, 3–11
O’Keefe B.R., Smee D.F., Turpin J.A., Saucedo C.J., Gustafson K.R., Mori T., et al., Potent antiinfluenza activity of cyanovirin-N and interactions with viral hemagglutinin, Antimicrob. Agents Chemother., 2003, 47, 2518–2525
Balzarini J., Carbohydrate-binding agents: a potential future cornerstone for the chemotherapy of enveloped viruses?, Antivir. Chem. Chemother., 2007, 18, 1–11
Bertaux C., Daelemans D., Meertens L., Cormier E.G., Reinus J.F., Peumans W.J., et al., Entry of hepatitis C virus and human immunodeficiency virus is selectively inhibited by carbohydrate-binding agents but not by polyanions, Virology, 2007, 36, 40–50
Micewicz E.D., Cole A.L., Jung C.L., Luong H., Phillips M.L., Pratikhya P., et al., Grifonin-1: a small HIV-1 entry inhibitor derived from the algal lectin, Griffithsin, PLoS One, 2010, 5, e14360
Romanelli F., Smith K.M., Hoven A.D., Chloroquine and hydroxychloroquine as inhibitors of human immunodeficiency virus (HIV-1) activity, Curr. Pharm. Des., 2004, 10, 2643–2648
Boelaert J.R., Sperber K., Piette J., Chloroquine exerts an additive in vitro anti-HIV type 1 effect when associated with didanosine and hydroxyurea, AIDS Res. Hum. Retroviruses, 1999, 15, 1241–7124
Sperber K., Louie M., Kraus T., Proner J., Sapira E., Lin S., et al., Hydroxychloroquine treatment of patients with human immunodeficiency virus type 1, Clin. Ther., 1995, 17, 622–636
Paton N.I., Aboulhab J., Hydroxychloroquine, hydroxyurea and didanosine as initial therapy for HIV-infected patients with low viral load: safety, efficacy and resistance profile after 144 weeks, HIV Med., 2005, 6, 13–20
Naarding M.A., Baan E., Pollakis G., Paxton W.A., Effect of chloroquine on reducing HIV-1 replication in vitro and the DC-SIGN mediated transfer of virus to CD4+ T-lymphocytes, Retrovirology, 2007, 4, 6
Vincent M.J., Bergeron E., Benjannet S., Erickson B.R., Rollin P.E., Ksiazek T.G., et al., Chloroquine is a potent inhibitor of SARS coronavirus infection and spread, Virol. J., 2005, 2, 69
De Clercq E., Potential antivirals and antiviral strategies against SARS coronavirus infections, Expert Rev. Anti Infect. Ther., 2006, 4, 291–302
Keyaerts E., Vijgen L., Maes P., Neyts J., Van Ranst M., In vitro inhibition of severe acute respiratory syndrome coronavirus by chloroquine, Biochem. Biophys. Res. Commun., 2004, 323, 264–268
Di Trani L., Savarino A., Campitelli L., Norelli S., Puzelli S., D’Ostilio D., et al., Different pH requirements are associated with divergent inhibitory effects of chloroquine on human and avian influenza A viruses, Virol. J., 2007, 4, 39
Ooi E.E., Chew J.S., Loh J.P., Chua R.C., In vitro inhibition of human influenza A virus replication by chloroquine, Virol. J., 2006, 3, 39
Savarino A., Di Trani L., Donatelli I., Cauda R., Cassone A., New insights into the antiviral effects of chloroquine, Lancet Infect. Dis., 2006, 6, 67–69
Rolain J.M., Colson P., Raoult D., Recycling of chloroquine and its hydroxyl analogue to face bacterial, fungal and viral infections in the 21st century, Int. J. Antimicrob. Agents, 2007, 30, 297–308
Vigerust D.J., McCullers J.A., Chloroquine is effective against influenza A virus in vitro but not in vivo, Influenza Other Respi Viruses, 2007, 1, 189–192
Fedson D.S., Confronting an influenza pandemic with inexpensive generic agents: can it be done?, Lancet Infect. Dis., 2008, 8, 571–576
Dwek R.A., Butters T.D., Platt F.M., Zitzmann N., Targeting glycosylation as a therapeutic approach, Nat. Rev. Drug Discov., 2002, 1, 65–75
Chang J., Schul W., Butters T.D., Yip A., Liu B., Goh A., et al., Combination of alpha-glucosidase inhibitor and ribavirin for the treatment of dengue virus infection in vitro and in vivo, Antiviral Res., 2011, 89, 26–34
Chang J., Wang L., Ma D., Qu X., Guo H., Xu X., et al., Novel imino sugar derivatives demonstrate potent antiviral activity against flaviviruses, Antimicrob. Agents Chemother., 2009, 53, 1501–1508
Vancova M., Nebesarova J., Grubhoffer L., Lectin-binding characteristics of a Lyme borreliosis spirochete Borrelia burgdorferi sensu stricto, Folia Microbiol., (Praha), 2005, 50, 229–238
Logan S.M., Hui J.P., Vinogradov E., Aubry A.J., Melanson J.E., Kelly J.F., et al., Identification of novel carbohydrate modifications on Campylobacter jejuni 11168 flagellin using metabolomics-based approaches, FEBS J., 2009, 276, 1014–1023
Appelmelk B.J., den Dunnen J., Driessen N.N., Ummels R., Pak M., Nigou J., et al., The mannose cap of mycobacterial lipoarabinomannan does not dominate the Mycobacterium-host interaction, Cell Microbiol., 2008, 10, 930–944
Twine S.M., Paul C.J., Vinogradov E., McNally D.J., Brisson J.R., Mullen J.A., et al., Flagellar glycosylation in Clostridium botulinum, FEBS J., 2008, 275, 4428–4444
Romanova J., Katinger D., Ferko B., Voglauer R., Mochalova L., Bovin N., et al., Distinct host range of influenza H3N2 virus isolates in Vero and MDCK cells is determined by cell specific glycosylation pattern, Virology, 2003, 307, 90–97
Wright K.E., Spiro R.C., Burns J.W., Buchmeier M.J., Post-translational processing of the glycoproteins of lymphocytic choriomeningitis virus, Virology, 1990, 177, 175–183
Branco L.M., Garry R.F., Characterization of the Lassa virus GP1 ectodomain shedding: implications for improved diagnostic platforms, Virol. J., 2009, 6, 147
Martinez I., Melero J.A., Binding of human respiratory syncytial virus to cells: implication of sulfated cell surface proteoglycans, J. Gen. Virol., 2000, 81, 2715–2722
Feinberg H., Castelli R., Drickamer K., Seeberger P.H., Weis W.I., Multiple modes of binding enhance the affinity of DC-SIGN for high mannose N-linked glycans found on viral glycoproteins, J. Biol. Chem., 2007, 282, 4202–4209
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Vigerust, D.J. Protein glycosylation in infectious disease pathobiology and treatment. cent.eur.j.biol. 6, 802–816 (2011). https://doi.org/10.2478/s11535-011-0050-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11535-011-0050-8