Abstract
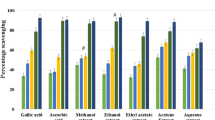
Hieracium pilosella L. (Asteraceae) is a well-known plant used in ethno-medicine as its inflorescences are particularly rich in beneficial polyphenolics. This research aimed to elucidate the structure of a new flavone glycoside isolated from the inflorescences of Hieracium pilosella and evaluate its antioxidant, antimicrobial and antiproliferative activities. The chromatographic methods were successfully applied to isolate the new flavonoid. Its structure was determined by subsequent UV, NMR and MS experiments and identified as isoetin 4′-O-β-D-glucopyranoside. Free radical scavenging capacity was examined by measuring the scavenging activity of the new isoetin derivative on 2,2-diphenyl-1-picrylhydrazyl (DPPH). The compound was also screened for spectrum of antimicrobial activity using the agar well diffusion method. Minimum inhibitory concentration (MIC) for Pseudomonas aeruginosa ATCC 9027 was performed by the micro-dilution broth method. The antiproliferative effect of tested glycoside was assessed in two human tumor cell lines derived from lung (A549) and colon (HT-29) carcinoma and cell proliferation was determined by means of MTT method. The tested compound showed high antiradical activity, reducing the DPPH? with EC50 7.9 μM (3.7 µg/ml) and exhibited narrow antimicrobial spectrum among tested microorganisms. The compound was active against Pseudomonas aeruginosa ATCC 9027 (MIC 125 μg/ml) which is prone to causing infections that are difficult to treat due to it developing extremely rapid antibiotic resistance. In the antiproliferative studies, cell proliferation of the colon (HT-29) carcinoma cell line was significantly decreased after exposure to the compound. The results indicate that isoetin 4′-O-β-D-glucopyranoside possesses antioxidant capacity and very promising antibacterial activity and could have uses as an effective antipseudomonal agent as well a antiproliferative agent.
Similar content being viewed by others
References
Lakić N.S., Mimica-Dukić N.M., Isak J.M., Božin B.N., Antioxidant properties of Galium verum L. (Rubiaceae) extracts, Cent. Eur. J. Biol., 2010, 5, 331–337
Harborne J.B., The Flavonoids: Advances in research since 1986, Chapman & Hall, London, 1999
Bishop G.F., Davy A.J., Biological flora of the British Isles. Hieracium pilosella L. (Pilosella officinarum F. Schultz & Schultz-Bip.), J. Ecol., 1994, 82, 195–210
Beaux D., Fleurentin J., Mortier F., Effect of extracts of Ortosiphon stamineus Benth, Hieracium pilosella L., Sambucus nigra L. and Arctostaphylos uva-ursi (L.) spreng. in Rats, Phytother. Res., 1999, 13, 222–225
Bolle P., Bello U., Faccendini P., Martinoli L., Tita B., Hieracium pilosella L.: pharmacological effect of ethanol extract, Pharmacol. Res., 1993, 27, 29–30
Goetz P., Wuyts D., Phytothérapie et nutrithérapie de l’hypertension artérielle (Phytotherapy and nutritherapy of hypertension), Phytothérapie, 2008, 6, 247–252, (in French)
Moro C.O., Basile G., Obesity and medicinal plants, Fitoterapia, 2000, 71, S73–S82
Dombrowicz E., Šwiątek L., Kopycki W., Phenolic acids in Inflorescentia Helichrysi and Herba Hieracii pilosellae, Pharmazie, 1992, 47, 469–470
Stanojević L.P., Stanković M.Z., Nikolić V.D., Nikolić L.B., Anti-oxidative and antimicrobial activities of Hieracium pilosella L. extracts, J. Serb. Chem. Soc., 2008, 73, 531–540
Barbour E.K., Sharif M.A., Sagherian V.K., Habre A.N., Talhouk R.S., Talhouk S.N., Screening of selected plants of Lebanon for antimicrobial activity, J. Ethnopharmacol., 2004, 93, 1–7
Bedoya L.M., Sanchez-Palomino S., Abad M.J., Bermejo P., Alcami J., Anti-HIV activity of medicinal plant extracts, J. Ethnopharmacol., 2001, 77, 113–116
Gawrońska-Grzywacz M., Krzaczek T., Flavonoids and coumarins from Hieracium pilosella L. (Asteraceae), Acta Soc. Bot. Pol., 2009, 78, 189–195
Gawrońska-Grzywacz M., Krzaczek T., Free and bound phenolic acids in inflorescences and rhizomes with roots of Hieracium pilosella L. (Asteraceae), Acta Soc. Bot. Pol., 2006, 75, 215–218
Zidorn C., Gottschlich G., Stuppner H., Chemosystematic investigations on phenolics from flowerheads of Central European taxa of Hieracium sensu lato (Asteraceae), Plant Syst. Evol., 2002, 231, 39–58
Gluchoff-Fiasson K., Favre-Bonvin J., Fiasson J.L., Glycosides and acylated glycosides of isoetin from European species of Hypochoeris, Phytochemistry, 1991, 30, 1673–1675
Harborne J.B., Revised structures for three isoetin glycosides, yellow flower pigments in Heywoodiella oligocephala, Phytochemistry, 1991, 30, 1677–1678
Marco J.A., Barbera O., Rodriguez S., Domingo C., Adell J., Flavonoids and other phenolics from Artemisia hispanica, Phytochemistry, 1988, 27, 3155–3159
Pauli G.F., Junior P., Phenolic glycosides from Adonis aleppica, Phytochemistry, 1995, 38, 1245–1250
Shi S., Zhang Y., Zhao Y., Huang K., Preparative isolation and purification of three flavonoid glycosides from Taraxacum mongolicum by highspeed counter-current chromatography, J. Sep. Sci., 2008, 31, 683–688
Shi S., Zhao Y., Zhou H., Zhang Y., Jiang X., Huang K., Identification of antioxidants from Taraxacum mongolicum by high-performance liquid chromatography-diode array detection-radicalscavenging detection-electrospray ionization mass spectrometry and nuclear magnetic resonance experiments, J. Chromatogr. A, 2008, 1209, 145–152
Rahman M.A.A., Moon S.S., Isoetin 5′-Methyl Ether, A Cytotoxic Flavone from Trichosanthes kirilowii, Bull. Korean Chem. Soc., 2007, 28, 1261–1264
Mabry T.J., Markham K.R., Thomas M.B., The Systematic Identification of Flavonoids, Springer-Verlag, Berlin-Heidelberg-New York, 1970
Brand-Williams W., Cuvelier M.E., Berset C., Use of free radical method to evaluate antioxidant activity, Lebensm. Wiss. Technol., 1995, 28, 25–30
Nowak R., Gawlik-Dziki U., Polyphenols of Rosa L. leaves extracts and their radical scavenging activity, Z. Naturforsch. C., 2007, 62, 32–38
Juszczak M., Matysiak J., Brzana W., Niewiadomy A., Rzeski W., Evaluation of antiproliferative activity of 2-(monohalogenphenylamino)-5-(2,4-dihydroxyphenyl)-1,3,4-thiadiazoles, Arzneimittelforschung, 2008, 58, 353–357
Harborne J.B., Williams C.A., Advances in flavonoid research since 1992, Phytochemistry, 2000, 55, 481–504
Rice-Evans C.A., Miller N.J., Paganga G., Structure-antioxidant activity relationships of flavonoids and phenolic acids, Free Radic. Biol. Med., 1996, 20, 933–956
Lambert R.J.W., Pearson J., Susceptibility testing: accurate and reproducible minimum inhibitory concentration (MIC) and non-inhibitory concentration (NIC) values, J. Appl. Microbiol., 2008, 8, 784–790
Fluit A.C., Verhoef J., Schmitz F.J., Antimicrobial resistance in European isolates of Pseudomonas aeruginosa, European SENTRY Participants, Eur. J. Clin. Microbiol. Infect. Dis., 2000, 19, 370–374
Karlowsky J.A., Draghi D.C., Jones M.E., Thornsberry C., Friedland I.R., Saham D.F., Surveillance for antimicrobial susceptibility among clinical isolates of Pseudomonas aeruginosa and Acinetobacter baumanii from hospitalized patients in the United States, 1998 to 2001, Antimicrob. Agents Chemother., 2003, 47, 1681–1688
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Gawrońska-Grzywacz, M., Krzaczek, T., Nowak, R. et al. Biological activity of new flavonoid from Hieracium pilosella L.. cent.eur.j.biol. 6, 397–404 (2011). https://doi.org/10.2478/s11535-011-0017-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11535-011-0017-9