Abstract
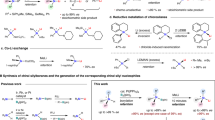
Hydroboration of trialkyn-1-yl(organo)silanes with one equivalent and two equivalents of 9-borabicyclo[3.3.1]nonane, 9-BBN afford dialkyn-1-ylsilanes and alkyn-1-ylsilanes, respectively. The alkyn-1-ylsilane derivatives are stable at room temperature and can be store under dry argon for prolong period of time. These compounds are attractive materials for further rearrangements to afford novel 1-silacyclobutene derivatives bearing Si-alkenyl or Si-alkynyl functionalities. The hydroboration reaction is well controlled by the Si-R1 function, i.e., the starting silanes with R1 = Ph selectively afford hydroboration of one Si-C≡C bond with one equivalent of 9-BBN, leaving the other two functionalities untouched. Under mild reaction conditions (25°C), the starting silanes with R1 = Me lead to mixture containing dialkyn-1-ylsilane, alkyn-1-ylsilane and their respective 1-silacyclobutene derivatives. All new compounds are sensitive towards air and moisture and were studied by multinuclear magnetic resonance spectroscopy (1H, 13C, 11B, 29Si NMR) in solution.
Similar content being viewed by others
References
T. Takahashi, Z. Xi, Y. Obora, N. Suzuki, J. Am. Chem. Soc. 117, 2665 (1995)
M. Horacek, N. Bazyakina, P. Stepnicka, R. Gyepes, I. Cisarova, S. Bredeau, P. Meunier, J. Kubista, K. Mach, J. Organomet. Chem. 628, 30 (2001)
A.C. Dema, C.M. Lukehart, A.T. McPhail, D.R. McPhail, J. Am. Chem. Soc. 112, 7229 (1990)
J. Mohseni-Ala, N. Auner, Inorg. Chim. Acta 359, 4673 (2006)
K.C. Jin, V.T. Dang, Y. Ikee, T. Yamada, S. Sano, M. Shiro, Y. Nagao, Heterocycles 70, 71 (2006)
N. Auner, C.R. Heikenwälder, C. Wagner, Organometallics 12, 4135 (1993)
G.T. Burns, T.J. Barton, J. Am. Chem. Soc. 105, 2006 (1983)
B. Wrackmeyer, H.E. Maisel, E. Molla, A. Motalib, A. Badshah, M.H. Bhatti, S. Ali, Appl. Organomet. Chem. 17, 465 (2003)
B. Wrackmeyer, E. Khan, R. Kempe, Appl. Organomet. Chem. 21, 39 (2007)
B. Wrackmeyer, E. Khan, S. Bayer, K. Shahid, Z. Naturforsch. 62b, 1174 (2007)
E. Khan, S. Bayer, B. Wrackmeyer, Z. Naturforsch. 64b, 47 (2009)
E. Khan, B. Wrackmeyer, Turk. J. Chem. (In press)
B. Wrackmeyer, E. Khan, R. Kempe, Appl. Organomet. Chem. 22, 383 (2008)
W.E. Davidsohn, M.C. Henry, Chem. Rev. 67, 73 (1976)
L. Brandsma, Preparative Acetylenic Chemistry, 2nd edition (Elsevier, Amsterdam, 1988)
L. Brandsma, Synthesis of Acetylenes, Allenes, Cumulenes — Methods and Techniques (Elsevier, Amsterdam, 2004)
B. Wrackmeyer, E. Khan, A. Badshah, E. Molla, P. Thoma, O.L. Tok, W. Milius, R. Kempe, J. Senker, Z. Naturforsch. 65b, 119 (2010)
B. Wrackmeyer, E. Khan, S. Bayer, O.L. Tok, E.V. Klimkina, W. Milius, R. Kempe, Z. Naturforsch. 65b, 725 (2010)
E. Khan, B. Wrackmeyer, R. Kempe, Eur. J. Inorg. Chem. 5367 (2008)
E. Khan, R. Kempe, B. Wrackmeyer, Appl. Organomet. Chem. 23, 204 (2009)
G.A. Morris, R. Freeman, J. Am. Chem. Soc. 101, 760 (1979)
G.A. Morris, J. Am. Chem. Soc. 102, 428 (1980)
G.A. Morris, J. Magn. Reson. 41, 185 (1980)
D.P. Burum, R.R. Ernst, J. Magn. Reson. 39, 163 (1980)
B. Wrackmeyer, E. Khan, R. Kempe, Z. Anorg. Allg. Chem. 633, 453 (2007)
B. Wrackmeyer, H. E. Maisel, W. Milius, M.H. Bhatti, S. Ali, J. Organomet. Chem. 669, 72 (2003)
B. Wrackmeyer, E. Khan, W. Milius, Z. Naturforsch. 63b, 1267 (2008)
B. Wrackmeyer, E. Khan, R. Kempe, Z. Naturforsch. 62b, 75 (2007)
B. Wrackmeyer, Progr. NMR Spectrosc. 12, 227 (1979)
B. Wrackmeyer, Annu. Rep. NMR Spectrosc. 20, 61 (1988)
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Khan, E., Wrackmeyer, B. Synthesis and NMR spectroscopic characterization of Si-substituted 1-silacyclobutene derivatives. cent.eur.j.chem. 9, 126–132 (2011). https://doi.org/10.2478/s11532-010-0122-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11532-010-0122-z