Abstract
Disease stabilization after first-line chemotherapy, also known as induction chemotherapy, is defined, according to the Response Evaluation Criteria in Solid Tumours (RECIST), as having neither sufficient shrinkage to qualify as a partial response (PR) nor sufficient increase to qualify as progressive disease (PD). In oncology, stable disease (SD) has often been viewed as an equivocal result and is therefore of unclear clinical value.
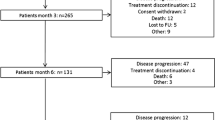
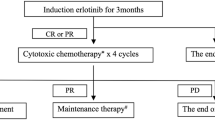
In SD patients with advanced non-small cell lung cancer (NSCLC) who have already received four cycles of first-line chemotherapy with platinum agents plus a third-generation agent (gemcitabine, vinorelbine, docetaxel or paclitaxel) or pemetrexed, the continuation of the original treatment is not recommended according to the American Society of Clinical Oncology (ASCO) guidelines. The ASCO guidelines recommend maintenance with bevacizumab or cetuximab, as tolerated until progression, only for platinum-based chemotherapy combined with bevacizumab or cetuximab. Several trials and a meta-analysis have, however, suggested a role for maintenance treatment in patients without progression after induction chemotherapy. The National Comprehensive Cancer Network guidelines recently suggested that maintenance therapy may be considered after four to six cycles of induction platinum doublets for patients with tumour responses or SD, and recommended first-line treatment with epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors in EGFR mutated patients to continue until PD. More recently, two randomized clinical trials that compared pemetrexed or erlotinib with a placebo demonstrated a better overall survival in favour of maintenance therapy. In subgroup analyses for both trials, patients with SD after first-line induction chemotherapy had pronounced survival benefits when erlotinib or pemetrexed maintenance therapy was given, although this result was not achieved in patients with a complete response or PR after induction chemotherapy. The management of patients with SD after first-line chemotherapy is an important issue because only a minority of patients with advanced NSCLC experience tumour shrinkage after standard platinum-based chemotherapy. Many more patients experience either SD or PD. The notion that the prognosis of SD patients varies greatly due to the complexity of SD should, however, be taken into careful consideration for the treatment decision. Therefore, suggestions for the further classification of SD are urgently needed to enable the use of an alternative therapy at an early time.
Similar content being viewed by others
References
Therasse P, Arbuck SG, Eisenhauer EA, et al. New guidelines to evaluate the response to treatment in solid tumors: European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 2000 Feb 2; 92(3): 205–16
Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 2009 Jan; 45(2): 228–47
Miller AB, Hoogstraten B, Staquet M, et al. Reporting results of cancer treatment. Cancer 1981 Jan 1; 47(1): 207–14
Choi H, Charnsangavej C, Faria SC, et al. Correlation of computed tomography and positron emission tomography in patients with metastatic gastrointestinal stromal tumor treated at a single institution with imatinib mesylate: proposal of new computed tomography response criteria. J Clin Oncol 2007 May 1; 25(13): 1753–9
Sirohi B, Ashley S, Norton A, et al. Early response to platinum-based first-line chemotherapy in non-small cell lung cancer may predict survival. J Thorac Oncol 2007 Aug; 2(8): 735–40
Fidias PM, Dakhil SR, Lyss AP, et al. Phase III study of immediate compared with delayed docetaxel after frontline therapy with gemcitabine plus carboplatin in advanced non-small-cell lung cancer. J Clin Oncol 2009 Feb 1; 27(4): 591–8
Lara Jr PN, Redman MW, Kelly K, et al., Southwest Oncology Group. Disease control rate at 8 weeks predicts clinical benefit in advanced non-small-cell lung cancer: results from Southwest Oncology Group randomized trials. J Clin Oncol 2008 Jan 20; 26(3): 463–7
Suzuki C, Jacobsson H, Hatschek T, et al. Radiologic measurements of tumor response to treatment: practical approaches and limitations. Radiographics 2008 Mar-Apr; 28(2): 329–44
Shepherd FA, Rodrigues Pereira J, Ciuleanu T, et al. Erlotinib in previously treated non-small-cell lung cancer. N Engl J Med 2005 Jul 14; 353(2): 123–32
Azzoli CG, Baker Jr S, Temin S, et al. American Society of Clinical Oncology Clinical Practice Guideline update on chemotherapy for stage IV non-small-cell lung cancer. J Clin Oncol 2009 Dec 20; 27(36): 6251–66
Sandler A, Gray R, Perry MC, et al. Paclitaxel-carboplatin alone or with bevacizumab for non-small-cell lung cancer. N Engl J Med 2006 Dec 14; 355(24): 2542–50
Reck M, von Pawel J, Zatloukal P, et al. Overall survival with cisplatin-gemcitabine and bevacizumab or placebo as first-line therapy for nonsquamous non-small-cell lung cancer: results from a randomised phase III trial (AVAiL). Ann Oncol 2010 Sep; 21(9): 1804–9
Reck M, von Pawel J, Zatloukal P, et al. Phase III trial of cisplatin plus gemcitabine with either placebo or bevacizumab as first-line therapy for nonsquamous non-small-cell lung cancer: AVAil. J Clin Oncol 2009 Mar 10; 27(8): 1227–34
Pirker R, Pereira JR, Szczesna A, et al. Cetuximab plus chemotherapy in patients with advanced non-small-cell lung cancer (FLEX): an open-label randomised phase III trial. Lancet 2009 May 2; 373(9674): 1525–31
Brodowicz T, Krzakowski M, Zwitter M, et al. Cisplatin and gemcitabine first-line chemotherapy followed by maintenance gemcitabine or best supportive care in advanced non-small cell lung cancer: a phase III trial. Lung Cancer 2006; 52: 155–63
Park JO, Kim SW, Ahn JS, et al. Phase III trial of two versus four additional cycles in patients who are nonprogressive after two cycles of platinum based chemotherapy in non-small-cell lung cancer. J Clin Oncol 2007; 25: 5233–9
Socinski MA, Schell MJ, Peterman A, et al. Phase III trial comparing a defined duration of therapy versus continuous therapy followed by second line therapy in advanced-stage IIIB/IV non-small-cell lung cancer. J Clin Oncol 2002; 20: 1335–43
von Plessen C, Bergman B, Andresen O, et al. Palliative chemotherapy beyond three courses conveys no survival or consistent quality-of-life benefits in advanced non-small-cell lung cancer. Br J Cancer 2006; 95: 966–73
Ciuleanu T, Brodowicz T, Zielinski C, et al. Maintenance pemetrexed plus best supportive care versus placebo plus best supportive care for non-small-cell lung cancer: a randomised, double-blind, phase 3 study. Lancet 2009 Oct 24; 374(9699): 1432–40
Cappuzzo F, Ciuleanu T, Stelmakh L, et al. Erlotinib as maintenance treatment in advanced non-small-cell lung cancer: a multicentre, randomised, placebo-controlled phase 3 study. Lancet Oncol 2010 Jun; 11(6): 521–9
Soon YY, Stockler MR, Askie LM, et al. Duration of chemotherapy for advanced non-small-cell lung cancer: a systematic review and meta-analysis of randomized trials. J Clin Oncol 2009 Jul 10; 27(20): 3277–83
NCCN clinical practice guidelines in oncology: non-small cell lung cancer. National Comprehensive Cancer Network, 2011. http://www.nccn.org/ [Accessed 2011 Feb 14
D’Addario G, Früh M, Reck M, et al. Metastatic non-small-cell lung cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 2010 May; 21 Suppl. 5: v116–19
Goldie JH, Coldman AJ. A mathematical model for relating the drug sensitivity of tumors to their spontaneous mutation rate. Cancer Treat Rep 1979; 63: 1727–33
Day RS. Treatment sequencing, asymmetry, and uncertainty: protocol strategies for combination chemotherapy. Cancer Res 1986; 46: 3876–85
Grossi F, Aita M, Follador A, et al. Sequential, alternating, and maintenance/consolidation chemotherapy in advanced non-small cell lung cancer: a review of the literature. Oncologist 2007 Apr; 12(4): 451–64
Belani CP, Brodowicz T, Ciuleanu T, et al. Maintenance pemetrexed (Pem) plus best supportive care (BSC) versus placebo (Plac) plus BSC: a randomized phase III study in advanced non-small cell lung cancer (NSCLC). J Clin Oncol 2009; 27 Suppl. 18s: 806s (abstr CRA8000)
Available at: http://www.fda.gov/AboutFDA/CentersOffices/CDER/ucm170660.htm-accessed. [Accessed 2010 Aug 16
Available at: http://www.ema.europa.eu/docs/en_GB/document_library/EPAR__WC500090680.pdf. [Accessed 2010 Aug 16
Coudert B, Ciuleanu T, Park K, et al. Survival benefit with erlotinib maintenance therapy relative to prior chemotherapeutic response: a subanalysis of the phase III SATURN study in NSCLC. 2nd European Lung Cancer Conference; 28 April–1 May, 2010; Geneva, Switzerland. Abstract 204O
Available at: http://www.fda.gov/AboutFDA/CentersOffices/CDER/ucm209058.htm-accesed. [Accessed 2010 Aug 16
Available at: http://www.ema.europa.eu/ema/index.jsp?curl_pages/medicines/human/medicines/000618/smops/Positive/human_smop_000085.jsp-accessed. [Accessed 2010 Aug 16
Perol M, Chouaid C, Milleron J, et al. Maintenance with either gemcitabine or erlotinib versus observation with predefined second-line treatment after cisplatin-gemcitabine induction chemotherapy in advanced NSCLC: IFCT-GFPC 0502 phase III study. J Clin Oncol 2010; 28(15s): abstr 7507
Paz-Ares LG, De Marinis F, Dediu M, et al. PARAMOUNT: phase III study of maintenance pemetrexed (pem) plus best supportive care (BSC) versus placebo plus BSC immediately following induction treatment with pem plus cisplatin for advanced nonsquamous non-small cell lung cancer (NSCLC). J Clin Oncol 2011; 29(18s): abstr CRA7510
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Grossi, F. Management of Non-Small Cell Lung in Cancer Patients with Stable Disease. Drugs 72 (Suppl 1), 20–27 (2012). https://doi.org/10.2165/1163013-S0-000000000-00000
Published:
Issue Date:
DOI: https://doi.org/10.2165/1163013-S0-000000000-00000