Abstract
Background and Objectives: In view of the increasing prevalence of morbidly obese patients, the influence of excessive total bodyweight (TBW) on the pharmacokinetics and pharmacodynamics of propofol was characterized in this study using bispectral index (BIS) values as a pharmacodynamic endpoint.
Methods: A population pharmacokinetic and pharmacodynamic model was developed with the nonlinear mixed-effects modelling software NONMEM VI, on the basis of 491 blood samples from 20 morbidly obese patients (TBW range 98–167 kg) and 725 blood samples from 44 lean patients (TBW range 55–98 kg) from previously published studies. In addition, 2246 BIS values from the 20 morbidly obese patients were available for pharmacodynamic analysis.
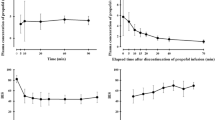
Results: In a three-compartment pharmacokinetic model, TBW proved to be the most predictive covariate for clearance from the central compartment (CL) in the 20 morbidly obese patients (CL 2.33L/min × [TBW/70]^[0.72]). Similar results were obtained when the morbidly obese patients and the 44 lean patients were analysed together (CL 2.22 L/min × [TBW/70]^[0.67]). No covariates were identified for other pharmacokinetic parameters. The depth of anaesthesia in the morbidly obese patients was adequately described by a two-compartment biophase-distribution model with a sigmoid maximum possible effect (Emax) pharmacodynamic model (concentration at half-maximum effect [EC50] 2.12 mg/L) without covariates.
Conclusion: We developed a pharmacokinetic and pharmacodynamic model of propofol in morbidly obese patients, in which TBW proved to be the major determinant of clearance, using an allometric function with an exponent of 0.72. For the other pharmacokinetic and pharmacodynamic parameters, no covariates could be identified.








Similar content being viewed by others
References
Ogden CL, Carroll MD, Curtin LR, et al. Prevalence of overweight and obesity in the United States, 1999–2004. JAMA 2006 Apr 5; 295(13): 1549–55
Whitlock G, Lewington S, Sherliker P, et al. Body-mass index and cause-specific mortality in 900 000 adults: collaborative analyses of 57 prospective studies. Lancet 2009 Mar 28; 373(9669): 1083–96
World Health Organization. Obesity: preventing and managing the global epidemic. Geneva: World Health Organization, 1997
Cheymol G. Effects of obesity on pharmacokinetics implications for drug therapy. Clin Pharmacokinet 2000 Sep; 39(3): 215–31
Hanley MJ, Abernethy DR, Greenblatt DJ. Effect of obesity on the pharmacokinetics of drugs in humans. Clin Pharmacokinet 2010; 49(2): 71–87
Servin F, Farinotti R, Haberer JP, et al. Propofol infusion for maintenance of anesthesia in morbidly obese patients receiving nitrous oxide: a clinical and pharmacokinetic study. Anesthesiology 1993 Apr; 78(4): 657–65
Schuttler J, Ihmsen H. Population pharmacokinetics of propofol: a multicenter study. Anesthesiology 2000 Mar; 92(3): 727–38
McLeay SC, Morrish GA, Kirkpatrick CM, et al. Encouraging the move towards predictive population models for the obese using propofol as a motivating example. Pharm Res 2009 Jul; 26(7): 1626–34
Cortínez LI, Anderson BJ, Penna A, et al. Influence of obesity on propofol pharmacokinetics: derivation of a pharmacokinetic model. Br J Anaesth 2010 Oct; 105(4): 448–56
Knibbe CA, Voortman HJ, Aarts LP, et al. Pharmacokinetics, induction of anaesthesia and safety characteristics of propofol 6% SAZN vs propofol 1 % SAZN and Diprivan-10 after bolus injection. Br J Clin Pharmacol 1999 Jun; 47(6): 653–60
Knibbe CA, Zuideveld KP, DeJongh J, et al. Population pharmacokinetic and pharmacodynamic modeling of propofol for long-term sedation in critically ill patients: a comparison between propofol 6% and propofol 1%. Clin Pharmacol Ther 2002 Dec; 72(6): 670–84
van Kralingen S, Diepstraten J, van de Garde EM, et al. Comparative evaluation of propofol 350 and 200 mg for induction of anaesthesia in morbidly obese patients: a randomized double-blind pilot study. Eur J Anaesthesiol 2010 Jun; 27(6): 572–4
Egan TD, Huizinga B, Gupta SK, et al. Remifentanil pharmacokinetics in obese versus lean patients. Anesthesiology 1998 Sep; 89(3): 562–73
Knibbe CA, Koster VS, Deneer VH, et al. Determination of propofol in low-volume samples by high-performance liquid chromatography with fluorescence detection. J Chromatogr B Biomed Sci Appl 1998 Mar 20; 706(2): 305–10
Beal SL, Sheiner LB, Boeckmann A. NONMEM user’s guide. San Francisco (CA): University of California, 1999
Struys MM, Coppens MJ, De Neve N, et al. Influence of administration rate on propofol plasma-effect site equilibration. Anesthesiology 2007 Sep; 107(3): 386–96
Pai MP, Paloucek FP. The origin of the “ideal” body weight equations. Ann Pharmacother 2000 Sep; 34(9): 1066–9
Janmahasatian S, Duffull SB, Ash S, et al. Quantification of lean bodyweight. Clin Pharmacokinet 2005; 44(10): 1051–65
van Kralingen S, van de Garde EMW, van Dongen EPA, et al. Maintenance of anesthesia in morbidly obese patients using propofol with continuous BIS-monitoring: a comparison of propofol-remifentanil and propofol-epidural anesthesia. Acta Anaesthesiol Belg. In press
Janmahasatian S, Duffull SB, Chagnac A, et al. Lean body mass normalizes the effect of obesity on renal function. Br J Clin Pharmacol 2008 Jun; 65(6): 964–5
Han PY, Duffull SB, Kirkpatrick CM, et al. Dosing in obesity: a simple solution to a big problem. Clin Pharmacol Ther 2007 Nov; 82(5): 505–8
Mathieu P, Lemieux I, Despres JP. Obesity, inflammation, and cardiovascular risk. Clin Pharmacol Ther 2010 Apr; 87(4): 407–16
Lloret Linares C, Decleves X, Oppert JM, et al. Pharmacology of morphine in obese patients: clinical implications. Clin Pharmacokinet 2009; 48(10): 635–51
Bjornsson MA, Norberg A, Kalman S, et al. A two-compartment effect site model describes the bispectral index after different rates of propofol infusion. J Pharmacokinet Pharmacodyn 2010 Jun; 37(3): 243–55
Sheiner LB, Stanski DR, Vozeh S, et al. Simultaneous modeling of pharmacokinetics and pharmacodynamics: application to d-tubocurarine. Clin Pharmacol Ther 1979 Mar; 25(3): 358–71
Mandema JW, Veng-Pedersen P, Danhof M. Estimation of amobarbital plasma-effect site equilibration kinetics: relevance of polyexponential conductance functions. J Pharmacokinet Biopharm 1991 Dec; 19(6): 617–34
Visser SA, Smulders CJ, Reijers BP, et al. Mechanism-based pharmacokinetic-pharmacodynamic modeling of concentration-dependent hysteresis and biphasic electroencephalogram effects of alphaxalone in rats. J Pharmacol Exp Ther 2002 Sep; 302(3): 1158–67
Wang LP, McLoughlin P, Paech MJ, et al. Low and moderate remifentanil infusion rates do not alter target-controlled infusion propofol concentrations necessary to maintain anesthesia as assessed by bispectral index monitoring. Anesth Analg 2007 Feb; 104(2): 325–31
Ferreira DA, Nunes CS, Antunes LM, et al. The effect of a remifentanil bolus on the bispectral index of the EEG (BIS) in anaesthetized patients independently from intubation and surgical stimuli. Eur J Anaesthesiol 2006 Apr; 23(4): 305–10
Bouillon TW, Bruhn J, Radulescu L, et al. Pharmacodynamic interaction between propofol and remifentanil regarding hypnosis, tolerance of laryngoscopy, bispectral index, and electroencephalographic approximate entropy. Anesthesiology 2004 Jun; 100(6): 1353–72
Liu N, Chazot T, Huybrechts I, et al. The influence of a muscle relaxant bolus on bispectral and datex-ohmeda entropy values during propofol-remifentanil induced loss of consciousness. Anesth Analg 2005 Dec; 101(6): 1713–8
Bonhomme V, Hans P. Muscle relaxation and depth of anaesthesia: where is the missing link? Br J Anaesth 2007 Oct; 99(4): 456–60
Marsh B, White M, Morton N, et al. Pharmacokinetic model driven infusion of propofol in children. Br J Anaesth 1991 Jul; 67(1): 41–8
Schnider TW, Minto CF, Gambus PL, et al. The influence of method of administration and covariates on the pharmacokinetics of propofol in adult volunteers. Anesthesiology 1998 May; 88(5): 1170–82
Absalom AR, Mani V, De Smet T, et al. Pharmacokinetic models for propofol: defining and illuminating the devil in the detail. Br J Anaesth 2009 Jul; 103(1): 26–37
La Colla L, Albertin A, La Colla G, et al. No adjustment vs adjustment formula as input weight for propofol target-controlled infusion in morbidly obese patients. Eur J Anaesthesiol 2009 May; 26(5): 362–9
Acknowledgements
Simone van Kralingen and Jeroen Diepstraten contributed equally to this work.
The authors would like to thank Brigitte Bliemer and Silvia Samson for their enthusiastic support and participation in this study.
No source of funding was used to conduct this study. The authors have no conflicts of interest that are relevant to the content of this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
van Kralingen, S., Diepstraten, J., Peeters, M.Y.M. et al. Population Pharmacokinetics and Pharmacodynamics of Propofol in Morbidly Obese Patients. Clin Pharmacokinet 50, 739–750 (2011). https://doi.org/10.2165/11592890-000000000-00000
Published:
Issue Date:
DOI: https://doi.org/10.2165/11592890-000000000-00000