Abstract
Background: The intake of medicines during pregnancy can have negative or toxic effects on the fetus, possibly leading to adverse pregnancy outcomes.
Objective: The aim of this study was to describe the level of drug exposure during pregnancy in a rural area of Mozambique and its relation to pregnancy outcome.
Methods: A total of 3105 pregnant women were interviewed in a cohort study. Information on disease, treatments received during pregnancy, and pregnancy outcome was collected. Newborns were examined at birth for clinical signs, birthweight, and presence of any congenital malformation.
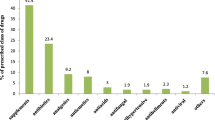
Results: Malaria and sexually transmitted diseases were the most frequently reported diseases (30.5% and 24.1 %, respectively), and 41% (1276/3105) of participants reported at least one drug exposure. The mean number of drugs taken per pregnant woman was 3.9 (SD 2.1). Antibiotics were the most commonly (41.2%) reported agents, followed by antimalarials (23.8%). There were more stillbirths (p < 0.007) among those reporting to be exposed to drugs compared with no exposure. Polydactyly was the most frequent malformation observed.
Conclusions: Drug exposure during pregnancy, including drugs with recognized potential pregnancy risk, was high in this rural area of southern Africa. The association of stillbirths with drug exposure might be a consequence of the disease that led to drug administration, although a direct causality of the drugs cannot be excluded. These findings emphasize the need for reinforcing pharmacovigilance systems in rural Africa, especially, or at least, in pregnant women.





Similar content being viewed by others
References
Lagoy CT, Joshi N, Cragan JD, et al. Medication use during pregnancy an urgent call for public health action. J Womens Health 2005; 14Suppl. 2: 104–9
Andrade SE, Gurwitz JH, Davis RL, et al. Prescription drug use in pregnancy. Am J Obstet Gynecol 2004; 191: 398–407
Osorio-de-Castro CGS, Pepe VLE, Luiza VL, et al. Prescribed and reported drug use during pregnancy. Cad Saúde Pública 2004; 1: S73–82
Eze UI, Eferakeya AE, Oparah AC, et al. Assessment of prescription profile of pregnant women visiting antenatal clinics. Pharm Pract 2007; 5Suppl. 3: 135–9
Engeland A, Bramness JG, Daltveit AK, et al. Prescription drug use among fathers and mothers before and during pregnancy: a population-based cohort study of 106,000 pregnancies in Norway 2004–2006. Br J Clin Pharmacol 2008; 65 Suppl. 5: 653–60
Yang T, Walker MC, Krewski D, et al. Maternal characteristics associated with pregnancy exposure to FDA category C, D, and X drugs in a Canadian population. Pharmacoepidemiol Drug Saf 2008; 17: 270–7
World Health Organization. Antimalarial drug combination therapy: report of a WHO technical consultation. WHO/CDS/RBM/2001.35. Geneva: World Health Organization, 2001 [online]. Available from URL: http://whqlibdoc.who.int/hq/2001/WHO_CDS_RBM_2001.35.pdf [Accessed 2011 Oct 12]
Ward SA, Sevene E, Hastings IM, et al. Antimalarial drugs and pregnancy: safety, pharmacokinetics, and pharmacovigilance. Lancet Infect Dis 2007; 7: 136–44
Dellicour S, ter Kuile F, Stergachis A. Pregnancy exposure registries for assessing antimalarial drug safety in pregnancy in malaria-endemic countries. PloS Med 2008; 5Suppl. 9: 1310–4
INDEPTH. Population and health in developing countries. Ottawa (ON): International Development Research Centre (IDRC), 2002: 189–95
Guinovart C, Bassat Q, Sigaúque B, et al. Malaria in rural Mozambique: part I. Children attending the outpatient clinic. Malar J 2008; 7: 36–45
Nhacolo AQ, Nhalungo DA, Sacoor CN, et al. Levels and trends of demographic indices in southern rural Mozambique: evidence from demographic surveillance in Manhiça district. BMC Public Health 2006; 6: 291
Bardají A, Sigauque B, Bruni L, et al. Clinical malaria in African pregnant women. Malar J 2008; 7Suppl. 27: 1–7
Programa Nacional de Controlo da Malária. Normas de tratamento da malaria. Maputo: MISAU, 2002
Lahuerta M, Aparicio E, Bardaji A, et al. Rapid spread and genetic diversification of HIV type 1 subtype C in a rural area of souther Mozambique. AIDS Res Hum Retroviruses 2008; 24Suppl. 2: 327–35
Programa Nacional de Controlo das ITS/HIV/SIDA. Plano estratégico nacional de combate ás ITS/HIV/SIDA do MISAU 2004–2008. Maputo: MISAU, 2004
World Health Organization. International statistical classification of diseases (ICD) and related health problems. Geneva: World Health Organization, 2007 [online]. Available from URL: http://www.who.int/classifications/apps/icd/icd10online/ [Accessed 2008 Jun 13]
Comissão Técnica de Terapêutica e Farmácia. Formulário Nacional de Medicamentos. Maputo: MISAU, 1999
Duplay D. Physicians desk reference. 59th ed. Montvale (NJ): Thomson PDR, 2005
Drug Safety Site. Drug safety during pregnancy and breastfeeding, 2003–2006, 2008 [online]. Available from URL: http://www.drugsafetysite.com [Accessed 2008 Jun 13]
Black RA, Hill A. Over-the-counter medications in pregnancy. Am Fam Physician 2003; 67Suppl. 12: 2517–24
White TE, Bushdid PB, Ritter S, et al. Artesunate-induced depletion of embryonic erythroblasts precedes embryolethality and teratogenicity in vivo. Birth Defects Res B Dev Reprod Toxicol 2006; 77Suppl. 5: 413–29
World Health Organization. Assessment of the safety of artemisinin compounds in pregnancy. WHO/CDS/MAL/20903.1094. WHO/GMP/TDR/Artemisinin/07.1. Geneva: World Health Organization, 2006 [online]. Available from URL: http://whqlibdoc.who.int/publications/2007/9789241596114_eng.pdf [Accessed 2011 Oct 12]
Watkins WM, Sibley CH, Hastings IM. The search for effective and sustainable treatments for plasmodium falciparum malaria in Africa: a model of the selection of resistance by antifolate drugs and their combinations. Am J Trop Med Hyg 2005; 72Suppl. 2: 163–73
van Geertruyden JP, Thomas F, Erhart A, et al. The contribution of malaria in pregnancy to perinatal mortality. Am Soc Trop Med Hyg 2004; 71Suppl. 2:35–40
Kromberg JG, Jenkins T. Common birth defects in South African Blacks. S Afr Med J 1982; 62Suppl. 17: 599–602
Sengeyi MA, Tshibangu K, Tozin R, et al. Etiopathogenesis and type of congenital malformations observed in Kinshasa (Zaire). J Gynecol Obstet Biol Reprod (Paris) 1990; 19Suppl. 8: 955–61
Scott-Emuakpor AB, Madueke ED. The study of genetic variation in Nigeria II: the genetics of polydactyly. Hum Hered 1976; 26Suppl. 3: 198–202
Cheng B, Dong Y, He L, et al. Crossed polydactyly type I caused by a point mutation in the GLI3 gene in a large Chinese pedigree. J Clin Lab Anal 2006; 20Suppl. 4: 133–8
Shaw GM, Wasserman C, O’Malley CD, et al. Maternal pesticide exposure from multiple sources and selected congenital anomalies. Epidemiology 1999; 10Suppl. 1: 60–6
Chambers CD, Polifkaje J, Friedman JM. Drug safety in pregnant women and their babies: ignorance not bliss. Clin Pharmacol Ther 2008; 83Suppl. 1: 181–3
Acknowledgments
We are grateful to all the pregnant women who participated in this study. We also thank the health personnel from the Manhiça Health Centre, especially those at the maternity clinic, and the staff at the Manhiça Health Research Centre (CISM). Our special thanks to Mr Llorenç Quinto, Miss Sonia Amos, and Miss Ana Sofia Roberto for their dedication throughout the study.
Esperança Sevene has received a Career Development Fellowship from the ‘European and Developing Countries Clinical Trials Partnership (EDCTP)’. This study was integrated into an intervention trial for malaria prevention in pregnancy entitled “A randomized placebo-controlled trial of intermittent preventive treatment in pregnant women in the context of insecticide treated nets delivered through the antenatal clinic” which received financial support from the Banco Bilbao Vizcaya Argentaria (BBVA) Foundation (grant number BBVA 02-0). The Centro de Investigação em Saúde de Manhiça (CISM) receives core funding from the Spanish Agency for International Cooperation (AECI).
The funding source did not have any involvement in the study design and conduct of the study; collection, management, analysis, and interpretation of data; preparation, review, or approval of the study manuscript; or in the decision to submit the paper for publication.
Esperança Sevene, Clara Menendez, Pedro Alonso, and Xavier Carné conceived and designed the study; Esperança Sevene, Azucena Bardají, Alda Mariano, Betuel Sigauqúe, Pedro Alonso, Xavier Carné, and Clara Menendez contributed to the study work; Edgar Ayala, Esperança Sevene, John Aponte, Sónia Machevo, and Clara Menendez analyzed the data; and Esperança Sevene, Azucena Bardají, Alda Mariano, Sónia Machevo, Betuel Sigaúque, Edgar Ayala, John Aponte, Xavier Carné, Pedro Alonso, and Clara Menendez wrote the paper. All authors declared no conflict of interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sevene, E., Bardají, A., Mariano, A. et al. Drug Exposure and Pregnancy Outcome in Mozambique. Pediatr Drugs 14, 43–49 (2012). https://doi.org/10.2165/11591270-000000000-00000
Published:
Issue Date:
DOI: https://doi.org/10.2165/11591270-000000000-00000