Abstract
Background and Objectives: As compared with individual tablets, saxagliptin/metformin extended-release (XR) fixed-dose combination (FDC) tablets offer potential for increased patient compliance with the convenience of once-daily dosing. Two bioequivalence studies assessed the fed-state bioequivalence of saxagliptin/metformin XR 5 mg/500 mg FDC (study 1) and saxagliptin/metformin XR 5mg/1000 mg FDC (study 2) relative to the same dosage strengths of individual component tablets administered concurrently. The effect of food on saxagliptin and metformin pharmacokinetics from the saxagliptin/metformin XR 5 mg/500 mg FDC and their steady-state pharmacokinetics from the saxagliptin/metformin XR 5 mg/1000 mg were also investigated.
Methods: These were randomized, open-label, single-dose, three-period, three-treatment, crossover studies in healthy subjects (n=30 in each study). The treatments in study 1 were a saxagliptin/metformin XR 5 mg/500 mg FDC tablet in the fed and fasted states on separate occasions, and saxagliptin 5 mg and metformin XR 500 mg co-administered in the fed state. The treatments in study 2 were a saxagliptin/metformin XR 5 mg/1000 mg FDC tablet in the fed state, saxagliptin 5 mg and 2 × metformin XR 500 mg co-administered in the fed state, and saxagliptin/metformin XR 5 mg/1000 mg FDC once daily for 4 days in the fed state to assess steady-state pharmacokinetics. The safety and tolerability of each treatment were also evaluated.
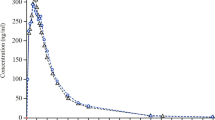
Results: For both studies, saxagliptin and metformin in the FDCs were bio-equivalent to the individual components as the limits of the 90% confidence interval of the ratio of adjusted geometric means for all key pharmacokinetic parameters were contained within 0.800 to 1.250. Compared with the fasted state, food did not have a meaningful effect on the pharmacokinetics of saxagliptin and metformin when administered as the saxagliptin/metformin XR 5 mg/500 mg FDC. The saxagliptin/metformin XR 5 mg/1000 mg FDC showed consistent pharmacokinetics at steady state without evidence of dose dumping. Co-administration of saxagliptin and metformin XR was generally safe and well tolerated as the FDCs or as individual tablets.
Conclusion: Saxagliptin/metformin XR 5 mg/500 mg and saxagliptin/metformin XR 5 mg/1000 mg FDCs were bioequivalent to individual tablets of saxagliptin and metformin of the same strengths. Additionally, food had little effect on the pharmacokinetics of saxagliptin and metformin administered in the saxagliptin/metformin XR 5 mg/500 mg FDC and the steady-state pharmacokinetics of the saxagliptin/metformin XR 5 mg/1000 mg FDC was consistent over time. No unexpected safety findings were observed with saxagliptin/metformin XR administration. The tolerability of the FDC of saxagliptin/metformin XR was comparable to that of the co-administered individual components. These results indicate that the safety and efficacy profile of co-administration of saxagliptin and metformin can be extended to the saxagliptin/metformin XR FDC tablets.
Trials Registration: ClinicalTrials.gov Identifiers: NCT01192139 and NCT01192152.






Similar content being viewed by others
References
Monnier L, Benichou M, Charra-Ebrard S, et al. An overview of the rationale for pharmacological strategies in type 2 diabetes: from the evidence to new perspectives. Diabetes Metab 2005 Apr; 31(2): 101–9
Deacon CF. Dipeptidyl peptidase-4 inhibitors in the treatment of type 2 diabetes: a comparative review. Diabetes Obes Metab 2011 Jan; 13(1): 7–18
Kirby M, Yu DM, O’Connor S, et al. Inhibitor selectivity in the clinical application of dipeptidyl peptidase-4 inhibition. Clin Sci (Lond) 2009 Sep 28; 118(1): 31–41
United States Prescribing Information for Onglyza [online]. Available from URL: http://packageinserts.bms.com/pi/pi_onglyza.pdf [Accessed 2011 Feb 1]
United States Prescribing Information for Glucophage/Glucophage XR [online]. Available from URL: http://packageinserts.bms.com/pi/pi_glucophage.pdf [Accessed 2009 Jan 1]
Hinke SA, Kuhn-Wache K, Hoffmann T, et al. Metformin effects on dipeptidylpeptidase IV degradation of glucagon-like peptide-1. Biochem Biophys Res Commun 2002; 291: 1302–8
Lenhard JM, Croom DK, Minnick DT. Reduced serum dipeptidyl peptidase-IV after metformin and pioglitazone treatments. Biochem Biophys Res Commun 2004; 324: 92–7
DeFronzo RA, Hissa MN, Garber AJ, et al. The efficacy and safety of saxagliptin when added to metformin therapy in patients with inadequately controlled type 2 diabetes with metformin alone. Diabetes Care 2009; 32: 1649–55
Jadzinsky M, Pfützner A, Paz-Pacheco E, et al. Saxagliptin given in combination with metformin as initial therapy improves glycaemic control in patients with type 2 diabetes compared with either monotherapy: a randomized controlled trial. Diabetes Obes Metab 2009; 11: 611–22
Patel CG, Komoroski BJ, Brenner E, et al. No meaningful pharmacokinetic drug-drug interaction between saxagliptin and metformin in healthy subjects [abstract 213]. Presented at the American College of Clinical Pharmacy Annual Meeting, Denver (CO), October 2007
Fujioka K, Pans M, Joyal S. Glycemic control in patients with type 2 diabetes mellitus switched from twice-daily immediate-release metformin to a once-daily extended-release formulation. Clin Ther 2003 Feb; 25(2): 515–29
Data on file, Bristol-Myers Squibb Company, 2011
Acknowledgements
The funding for this study was provided by Bristol-Myers Squibb Co. and AstraZeneca. All authors are stockholders and/or employees of either Bristol-Myers Squibb Co. or PPD, Inc. The authors wish to thank the clinical site staff and subjects for their contributions to these studies.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Boulton, D.W., Smith, C.H., Li, L. et al. Bioequivalence of Saxagliptin/Metformin Extended-Release (XR) Fixed-Dose Combination Tablets and Single-Component Saxagliptin and Metformin XR Tablets in Healthy Adult Subjects. Clin. Drug Investig. 31, 619–630 (2011). https://doi.org/10.2165/11590290-000000000-00000
Published:
Issue Date:
DOI: https://doi.org/10.2165/11590290-000000000-00000