Abstract
Background
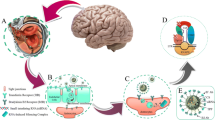
HIV infection of the CNS is the principle cause of HIV-associated dementia in adults and encephalopathy in children. Gene therapy techniques such as small interfering RNA (siRNA) possess great potential in drug development, but first they must overcome the key obstacle of reaching the interior of the affected cells. A successful delivery vector for anti-HIV drugs that is capable of crossing the blood-brain barrier (BBB) could provide a way of addressing this issue. Non-viral vectors such as dendrimers offer a means for effectively delivering and transfecting siRNA to the target cells.
Objective
To evaluate the application of gene therapy for reducing HIV replication in human astrocytes.
Methods
We used the 2G-NN16 amino-terminated carbosilane dendrimer as a method for delivering siRNA to HIV-infected human astrocytes. We tested the cytotoxicity in human astrocytoma cells caused by 2G-NN16 and dendriplexes formed with siRNA (siRNA/2G-NN16) by 3-(4,5-dimethylthiazol-2-yl)-2, 5-diphenyl-tetrazolium-bromide (MTT) and lactate dehydrogenase assays. The ability to transfect human astrocytes with siRNA/2G-NN16 dendriplexes was tested by flow cytometry and immunofluorescence microscopy. To assess the potential capability of siRNA/2G-NN16 dendriplexes for crossing the BBB, we used an in vitro transcytosis assay with bovine brain microvascular endothelial cells. HIV-1 inhibition assays using 2G-NN16 and siRNA/2G-NN16 dendriplexes were determined by quantification of the viral load from culture supernatants of the astrocytes.
Results
A gradual time-controlled degradation of the 2G-NN16 dendrimer and liberation of its siRNA cargo between 12 and 24 hours was observed via gel electrophoresis. There was no cytotoxicity in HIV-infected or non-infected human astrocytoma cells when treated with up to 24 μg/mL of 2G-NN16 dendrimer or siRNA/2G-NN16 dendriplexes, and siRNA/2G-NN16 dendriplexes were seen to successfully transfect human astrocytes even after crossing an in vitro BBB model. More interestingly, transfected siRNA was observed to exert a biologic effect, as dendriplexes were shown to down-regulate the housekeeping gene GAPDH and to reduce replication of HIV-1 strains X4-HIV NL4-3 and R5-HIV BaL in human astrocytes.
Conclusions
The 2G-NN16 dendrimer successfully delivers and transfects siRNA to HIV-infected human astrocytes and achieves gene silencing without causing cytotoxicity.Both authors have contributed equally to this paper.






Similar content being viewed by others
References
Sacktor N, Lyles RH, Skolasky R, et al. HIV-associated neurologic disease incidence changes: multicenter AIDS cohort study, 1990–1998. Neurology 2001 Jan 23; 56(2): 257–60
Lindl KA, Marks DR, Kolson DL, et al. HIV-associated neurocognitive disorder: pathogenesis and therapeutic opportunities. J Neuroimmune Pharmacol. Epub 2010 Apr 16
Ellis RJ, Deutsch R, Heaton RK, et al. Neurocognitive impairment is an independent risk factor for death in HIV infection. San Diego HIV Neurobehavioral Research Center Group. Arch Neurol 1997 Apr; 54(4): 416–24
Sanchez-Ramon S, Resino S, Bellon Cano JM, et al. Neuroprotective effects of early antiretrovirals in vertical HIV infection. Pediatr Neurol 2003 Sep; 29(3): 218–21
Canto-Nogues C, Sanchez-Ramon S, Alvarez S, et al. HIV-1 infection of neurons might account for progressive HIV-1-associated encephalopathy in children. J Mol Neurosci 2005; 27(1): 79–89
Gonzalez-Scarano F, Martin-Garcia J. The neuropathogenesis of AIDS. Nat Rev Immunol 2005 Jan; 5(1): 69–81
Kerza-Kwiatecki AP, Amini S. CNS as an HIV-1 reservoir: BBB and drug delivery. J Neurovirol 1999 Apr; 5(2): 113–4
Brew BJ. HIV, the brain, children, HAART and ‘neuro-HAART': a complex mix. Aids 2009 Sep 10; 23(14): 1909–10
Ghafouri M, Amini S, Khalili K, et al. HIV-1 associated dementia: symptoms and causes. Retrovirology 2006; 3: 28
McArthur JC, Haughey N, Gartner S, et al. Human immunodeficiency virus-associated dementia: an evolving disease. J Neurovirol 2003 Apr; 9(2): 205–21
Clifford DB. HIV-associated neurocognitive disease continues in the antiretroviral era. Top HIV Med 2008 Jun–Jul; 16(2): 94–8
Tozzi V, Balestra P, Bellagamba R, et al. Persistence of neuropsychologic deficits despite long-term highly active antiretroviral therapy in patients with HIV-related neurocognitive impairment: prevalence and risk factors. J Acquir Immune Defic Syndr 2007 Jun 1; 45(2): 174–82
Wynn HE, Brundage RC, Fletcher CV. Clinical implications of CNS penetration of antiretroviral drugs. CNS Drugs 2002; 16(9): 595–609
Sharp PA. RNA interference: 2001. Genes Dev 2001 Mar 1; 15(5): 485–90
Coburn GA, Cullen BR. siRNAs: a new wave of RNA-based therapeutics. J Antimicrob Chemother 2003 Apr; 51(4): 753–6
Lee MT, Coburn GA, McClure MO, et al. Inhibition of human immunodeficiency virus type 1 replication in primary macrophages by using Tat- or CCR5-specific small interfering RNAs expressed from a lentivirus vector. J Virol 2003 Nov; 77(22): 11964–72
Han W, Wind-Rotolo M, Kirkman RL, et al. Inhibition of human immunodeficiency virus type 1 replication by siRNA targeted to the highly conserved primer binding site. Virology 2004 Dec 5; 330(1): 221–32
Novina CD, Murray MF, Dykxhoorn DM, et al. siRNA-directed inhibition of HIV-1 infection. Nat Med 2002 Jul; 8(7): 681–6
Nishitsuji H, Kohara M, Kannagi M, et al. Effective suppression of human immunodeficiency virus type 1 through a combination of short- or long-hairpin RNAs targeting essential sequences for retroviral integration. J Virol 2006 Aug; 80(15): 7658–66
Pomerantz RJ. RNA interference meets HIV-1: will silence be golden? Nat Med 2002 Jul; 8(7): 659–60
Liu YP, Haasnoot J, ter Brake O, et al. Inhibition of HIV-1 by multiple siRNAs expressed from a single microRNA polycistron. Nucleic Acids Res 2008 May; 36(9): 2811–24
Kumar P, Ban HS, Kim SS, et al. T cell-specific siRNA delivery suppresses HIV-1 infection in humanized mice. Cell 2008 Aug 22; 134(4): 577–86
Song E, Zhu P, Lee SK, et al. Antibody mediated in vivo delivery of small interfering RNAs via cell-surface receptors. Nat Biotechnol 2005 Jun; 23(6): 709–17
Mintzer MA, Simanek EE. Nonviral vectors for gene delivery. Chem Rev 2009 Feb; 109(2): 259–302
Whitehead KA, Langer R, Anderson DG. Knocking down barriers: advances in siRNA delivery. Nat Rev Drug Discov 2009 Feb; 8(2): 129–38
Fischer D, Li Y, Ahlemeyer B, et al. In vitro cytotoxicity testing of polycations: influence of polymer structure on cell viability and hemolysis. Biomaterials 2003 Mar; 24(7): 1121–31
Kurreck J. RNA interference: from basic research to therapeutic applications. Angew Chem Int Ed Engl 2009; 48(8): 1378–98
Ortega P, Bermejo JF, Chonco L, et al. Novel water-soluble carbosilane dendrimers: synthesis and biocompatibility. Eur J Inorg Chem 2006; (7): 1388–96
Weber N, Ortega P, Clemente MI, et al. Characterization of carbosilane dendrimers as effective carriers of siRNA to HIV-infected lymphocytes. J Control Release 2008 Nov 24; 132(1): 55–64
Fréchet JMJ. Dendrimers and other dendritic macromolecules: from building blocks to functional assemblies in nanoscience and nanotechnology. J Polymer Sci Part A: Polymer Chem 2003 Mar; 1758(3): 290–300
Loscher W, Potschka H. Role of drug efflux transporters in the brain for drug disposition and treatment of brain diseases. Prog Neurobiol 2005 May; 76(1): 22–76
Bermejo JF, Ortega P, Chonco L, et al. Water-soluble carbosilane dendrimers: synthesis biocompatibility and complexation with oligonucleotides; evaluation for medical applications. Chemistry 2007; 13(2): 483–95
Chonco L, Bermejo-Martin JF, Ortega P, et al. Water-soluble carbosilane dendrimers protect phosphorothioate oligonucleotides from binding to serum proteins. Org Biomol Chem 2007 Jun 21; 5(12): 1886–93
Song E, Lee SK, Dykxhoorn DM, et al. Sustained small interfering RNA-mediated human immunodeficiency virus type 1 inhibition in primary macrophages. J Virol 2003 Jul; 77(13): 7174–81
Capodici J, Kariko K, Weissman D. Inhibition of HIV-1 infection by small interfering RNA-mediated RNA interference. J Immunol 2002 Nov 1; 169(9): 5196–201
Das AT, Brummelkamp TR, Westerhout EM, et al. Human immunodeficiency virus type 1 escapes from RNA interference-mediated inhibition. J Virol 2004 Mar; 78(5): 2601–5
Toborek M, Lee YW, Flora G, et al. Mechanisms of the blood-brain barrier disruption in HIV-1 infection. Cell Mol Neurobiol 2005 Feb; 25(1): 181–99
Kuo YC, Chen HH. Effect of nanoparticulate polybutylcyanoacrylate and methylmethacrylate-sulfopropylmethacrylate on the permeability of zidovudine and lamivudine across the in vitro blood-brain barrier. Int J Pharm 2006 Dec 11; 327(1–2): 160–9
Hartig PC, Hunter 3rd ES. Gene delivery to the neurulating embryo during culture. Teratology 1998 Sep–Oct; 58(3–4): 103–12
Sata M, Walsh K. Endothelial cell apoptosis induced by oxidized LDL is associated with the down-regulation of the cellular caspase inhibitor FLIP. J Biol Chem 1998 Dec 11; 273(50): 33103–6
Kolchinsky P, Kiprilov E, Sodroski J. Increased neutralization sensitivity of CD4-independent human immunodeficiency virus variants. J Virol 2001 Mar; 75(5): 2041–50
Trillo-Pazos G, Diamanturos A, Rislove L, et al. Detection of HIV-1 DNA in microglia/macrophages, astrocytes and neurons isolated from brain tissue with HIV-1 encephalitis by laser capture microdissection. Brain Pathol 2003 Apr; 13(2): 144–54
Alvarez Losada S, Canto-Nogues C, Munoz-Fernandez MA. A new possible mechanism of human immunodeficiency virus type 1 infection of neural cells. Neurobiol Dis 2002 Dec; 11(3): 469–78
Liu Y, Liu H, Kim BO, et al. CD4-independent infection of astrocytes by human immunodeficiency virus type 1: requirement for the human mannose receptor. J Virol 2004 Apr; 78(8): 4120–33
Jevprasesphant R, Penny J, Jalal R, et al. The influence of surface modification on the cytotoxicity of PAMAM dendrimers. Int J Pharm 2003 Feb 18; 252(1–2): 263–6
Vogelbaum MA, Tong JX, Higashikubo R, et al. Transfection of C6 glioma cells with the bax gene and increased sensitivity to treatment with cytosine arabinoside. J Neurosurg 1998 Jan; 88(1): 99–105
Lampela P, Soininen P, Urtti A, et al. Synergism in gene delivery by small PEIs and three different nonviral vectors. Int J Pharm 2004 Feb 11; 270(1–2): 175–84
Alvarez S, Jimenez JL, Serramia MJ, et al. Lack of association of HIV-1 biological or molecular properties with neurotropism for brain cells. J Mol Neurosci 2006; 29(2): 131–44
Gorry PR, Bristol G, Zack JA, et al. Macrophage tropism of human immunodeficiency virus type 1 isolates from brain and lymphoid tissues predicts neurotropism independent of coreceptor specificity. J Virol 2001 Nov; 75(21): 10073–89
MacKay JA, Deen DF, Szoka Jr FC. Distribution in brain of liposomes after convection enhanced delivery: modulation by particle charge, particle diameter, and presence of steric coating. Brain Res 2005 Feb 28; 1035(2): 139–53
Lockman PR, Koziara JM, Mumper RJ, et al. Nanoparticle surface charges alter blood-brain barrier integrity and permeability. J Drug Target 2004; 12(9–10): 635–41
Fenart L, Casanova A, Dehouck B, et al. Evaluation of effect of charge and lipid coating on ability of 60-nm nanoparticles to cross an in vitro model of the blood-brain barrier. J Pharmacol Exp Ther 1999 Dec; 291(3): 1017–22
Huang RQ, Qu YH, Ke WL, et al. Efficient gene delivery targeted to the brain using a transferrin-conjugated polyethyleneglycol-modified polyamidoamine dendrimer. Faseb J 2007 Apr; 21(4): 1117–25
Dehouck B, Fenart L, Dehouck MP, et al. A new function for the LDL receptor: transcytosis of LDL across the blood-brain barrier. J Cell Biol 1997 Aug 25; 138(4): 877–89
Balazs Z, Panzenboeck U, Hammer A, et al. Uptake and transport of high-density lipoprotein (HDL) and HDL-associated alpha-tocopherol by an in vitro blood-brain barrier model. J Neurochem 2004 May; 89(4): 939–50
Yousif S, Marie-Claire C, Roux F, et al. Expression of drug transporters at the blood-brain barrier using an optimized isolated rat brain microvessel strategy. Brain Res 2007 Feb 23; 1134(1): 1–11
Giovannoni G, Miller RF, Heales SJ, et al. Elevated cerebrospinal fluid and serum nitrate and nitrite levels in patients with central nervous system complications of HIV-1 infection: a correlation with blood-brain-barrier dysfunction. J Neurol Sci 1998; 156(1): 53–8
Petito CK, Cash KS. Blood-brain barrier abnormalities in the acquired immunodeficiency syndrome: immunohistochemical localization of serum proteins in postmortem brain. Ann Neurol 1992 Nov; 32(5): 658–66
Acknowledgments
This work was supported by grants from Fundación para la Investigación y Prevención del SIDA en España, FIPSE (240800/09), Red Temática de Investigación Cooperativa Sanitaria ISCIII (RETIC RD06/0006/0035), MNT-ERA NET 2007 (NAN2007-31198-E), Fondos de Investigación Sanitaria (FIS PS09/02029; PS09/02669), Fundación Caja Navarra, Co-munidad Autónoma de Madrid (S-SAL-0159-2006), and COST Action (TD0802) to Dr Muñoz-Fernóndez. Authors from the Universidad de Alcaláwere supported by MNT-ERA NET 2007 (NAN2007-31135-E) and Fondo de Investigación Sanitaria (PI040993). Dr José; Luis Jiménez was supported by FIS PI081495 and Programa de Investigación de la Consejería Sanidad de la Comunidad de Madrid, and Dr López-Fernández was supported by FIS CP06/0267 Miguel Servert Program. Maria Isabel Clemente is the holder of a fellowship from Fondo de Investigaciones Sanitarias (FI0501093). We would like to thank Laura Diaz for her assistance with flow cytometry analysis.
The authors have no conflicts of interest that are directly relevant to the content of this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jiménez, J.L., Clemente, M.I., Weber, N.D. et al. Carbosilane Dendrimers to Transfect Human Astrocytes with Small Interfering RNA Targeting Human Immunodeficiency Virus. BioDrugs 24, 331–343 (2010). https://doi.org/10.2165/11538400-000000000-00000
Published:
Issue Date:
DOI: https://doi.org/10.2165/11538400-000000000-00000