Abstract
Background:
Roflumilast is an oral, selective phosphodiesterase (PDE)-4 inhibitor in development for the treatment of chronic obstructive pulmonary disease (COPD). Both roflumilast and its metabolite roflumilast N-oxide have anti-inflammatory properties that contribute to overall pharmacological activity.
Objectives:
To model the pharmacokinetics of roflumilast and roflumilast N-oxide, evaluate the influence of potential covariates, use the total PDE4 inhibitory activity (tPDE4i) concept to estimate the combined inhibition of PDE4 by roflumilast and roflumilast N-oxide, and use individual estimates of tPDE4i to predict the occurrence of adverse events (AEs) in patients with moderate-to-severe COPD.
Methods:
We modelled exposure to roflumilast and roflumilast N-oxide (21 studies provided the index dataset and five separate studies provided the validation dataset), extended the models to COPD (using data from two studies) and assessed the robustness of the parameter estimates. A parametric bootstrap estimation was used to quantify tPDE4i in subpopulations. We established logistic regression models for each AE occurring in >2% of patients in a placebo-controlled trial that achieved a p-value of <0.2 in a permutation test. The exposure variables were the area under the plasma concentration-time curve (AUC) of roflumilast, the AUC of roflumilast N-oxide and tPDE4i. Individual AUC values were estimated from population models.
Results:
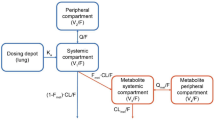
Roflumilast pharmacokinetics were modelled with a two-compartment model with first-order absorption including a lag time. A one-compartment model with zero-order absorption was used for roflumilast N-oxide. The final models displayed good descriptive and predictive performance with no appreciable systematic trends versus time, dose or study. Posterior predictive checks and robustness analysis showed that the models adequately described the pharmacokinetic parameters and the covariate effects on disposition. For roflumilast, the covariates of sex, smoking and race influenced clearance; and food influenced the absorption rate constant and lag time. For roflumilast N-oxide, age, sex and smoking influenced clearance; age, sex and race influenced the fraction metabolized; bodyweight influenced the apparent volume of distribution; and food influenced the apparent duration of formation. The COPD covariate increased the central volume of distribution of roflumilast by 184% and reduced its clearance by 39%; it also reduced the estimated volume of distribution of roflumilast N-oxide by 21% and reduced its clearance by 7.9%. Compared with the reference population (male, non-smoking, White, healthy, 40-year-old subjects), the relative geometric mean [95%CI] tPDE4i was higher in patients withCOPD(12.6%[−6.6, 35.6]), women (19.3%[8.2, 31.6]), Black subjects (42.1% [16.4, 73.4]), Hispanic subjects (28.2% [4.1, 57.9]) and older subjects (e.g. 8.3% [−11.2, 32.2] in 60-year-olds), and was lower in smokers (−19.1% [−34.0, −0.7]). Among all possible subgroups in this analysis, the subgroup with maximal tPDE4i comprised elderly, Black, female, non-smoking, COPD patients (tPDE4i 217% [95% CI 107, 437] compared with the value in the reference population). The probability of a patient with tPDE4i at the population geometric mean [95% CI] was 13.0% [7.5, 18.5] for developing diarrhoea, 6.0% [2.6, 9.4] for nausea and 5.1% [1.9, 8.6] for headache.
Conclusions:
Covariate effects have a limited impact on tPDE4i. There was a general association between tPDE4i and the occurrence of common AEs in patients with COPD.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Hatzelmann A, Schudt C. Anti-inflammatory and immunomodulatory potential of the novel PDE4 inhibitor roflumilast in vitro. J Pharmacol Exp Ther 2001; 297 (1): 267–79
Jones NA, Boswell-Smith V, Lever R, et al. The effect of selective phosphodiesterase isoenzyme inhibition on neutrophil function in vitro. Pulm Pharmacol Ther 2005; 18 (2): 93–101
Bundschuh DS, Eltze M, Barsig J, et al. In vivo efficacy in airway disease models of roflumilast, a novel orally active PDE4 inhibitor. J Pharmacol Exp Ther 2001; 297 (1): 280–90
Grootendorst DC, Gauw SA, Verhoosel RM, et al. Reduction in sputum neutrophil and eosinophil numbers by the PDE4 inhibitor roflumilast in patients with COPD. Thorax 2007; 62 (12): 1081–7
Calverley PM, Sanchez-Toril F, McIvor A, et al. Effect of 1-year treatment with roflumilast in severe chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2007; 176 (2): 154–61
Rabe KF, Bateman ED, O’Donnell D, et al. Roflumilast — an oral antiinflammatory treatment for chronic obstructive pulmonary disease: a randomised controlled trial. Lancet 2005; 366 (948): 563–71
Calverley PM, Rabe KF, Goehring UM, et al. Roflumilast in symptomatic chronic obstructive pulmonary disease: two randomised clinical trials. Lancet 2009; 374 (9691): 685–94
Fabbri LM, Calverley PM, Izquierdo-Alonso JL, et al. Roflumilast in moderateto- severe chronic obstructive pulmonary disease treated with longacting bronchodilators: two randomised clinical trials. Lancet 2009; 374 (9691): 695–703
Bethke TD, Böhmer GM, Hermann R, et al. Dose-proportional intraindividual single- and repeated-dose pharmacokinetics of roflumilast, an oral, once-daily phosphodiesterase 4 inhibitor. J Clin Pharmacol 2007; 47 (1): 26–36
Hauns B, Hermann R, Hünnemeyer A, et al. Investigation of a potential food effect on the pharmacokinetics of roflumilast, an oral, once-daily phosphodiesterase 4 inhibitor, in healthy subjects. J Clin Pharmacol 2006; 46 (10): 1146–53
Lahu G, Hü nnemeyer A, Herzog R, et al. Effect of repeated dose of erythromycin on the pharmacokinetics of roflumilast and roflumilast N-oxide. Int J Clin Pharmacol Ther 2009; 47 (4): 236–45
Lahu G, Hü nnemeyer A, von Richter O, et al. Effect of single and repeated doses of ketoconazole on the pharmacokinetics of roflumilast and roflumilast N-oxide. J Clin Pharmacol 2008; 48 (11): 1339–49
von Richter O, Lahu G, Hü nnemeyer A, et al. Effect of fluvoxamine on the pharmacokinetics of roflumilast and roflumilast N-oxide. Clin Pharmacokinet 2007; 46 (7): 613–22
Cotreau MM, von Moltke LL, Greenblatt DJ. The influence of age and sex on the clearance of cytochrome P450 3A substrates. Clin Pharmacokinet 2005; 44 (1): 33–60
Bebia Z, Buch SC, Wilson JW, et al. Bioequivalence revisited: influence of age and sex on CYP enzymes. Clin Pharmacol Ther 2004; 76 (6): 618–27
Mangoni AA, Jackson SH. Age-related changes in pharmacokinetics and pharmacodynamics: basic principles and practical applications. Br J Clin Pharmacol 2004; 57 (1): 6–14
Funck-Brentano C, Raphael M, Lafontaine M, et al. Effects of type of smoking (pipe, cigars or cigarettes) on biological indices of tobacco exposure and toxicity. Lung Cancer 2006; 54 (1): 11–8
Hermann R, Siegmund W, Giessmann T, et al. The oral, once-daily phosphodiesterase 4 inhibitor roflumilast lacks relevant pharmacokinetic interactions with inhaled budesonide. J Clin Pharmacol 2007; 47 (8): 1005–13
Hermann R, Nassr N, Lahu G, et al. Steady-state pharmacokinetics of roflumilast and roflumilast N-oxide in patients with mild and moderate liver cirrhosis. Clin Pharmacokinet 2007; 46 (5): 403–16
David M, Zech K, Seiberling M, et al. Roflumilast, a novel, oral, selective PDE4 inhibitor, shows high oral bioavailability. J Allergy Clin Immunol 2004; 113: S220–1
Nassr N, Lahu G, Hü nnemeyer A, et al. Magnesium hydroxide/aluminium hydroxide-containing antacid does not affect the pharmacokinetics of the targeted phosphodiesterase 4 inhibitor roflumilast. J Clin Pharmacol 2007; 47 (5): 660–6
Hermann R, Lahu G, Hauns B, et al. ‘Total PDE4 inhibitory activity’: a concept for evaluating pharmacokinetic alterations of roflumilast and roflumilast N-oxide in special populations and drug-drug interactions [abstract no. 2542]. Eur Respir J 2006; 28 Suppl. 50: 436s [online]. Available from URL: http://www.ers-education.org/pages/default.aspx?id=335&idBrowse=7697&det=1 [Accessed 2010 Jun 14]
Manning CD, Burman M, Christensen SB, et al. Suppression of human inflammatory cell function by subtype-selective PDE4 inhibitors correlates with inhibition of PDE4A and PDE4B. Br J Pharmacol 1999; 128 (7): 1393–8
Halpin DM. ABCD of the phosphodiesterase family: interaction and differential activity in COPD. Int J Chron Obstruct Pulmon Dis 2008; 3 (4): 543–61
Lipworth BJ. Phosphodiesterase-4 inhibitors for asthma and chronic obstructive pulmonary disease. Lancet 2005; 365 (9454): 167–75
Spina D. Phosphodiesterase-4 inhibitors in the treatment of inflammatory lung disease. Drugs 2003; 63 (23): 2575–94
Spina D. PDE4 inhibitors: current status. Br J Pharmacol 2008; 155 (3): 308–15
Kowalski KG, Hutmacher MM. Efficient screening of covariates in population models using Wald’s approximation to the likelihood ratio test. J Pharmacokinet Pharmacodyn 2001; 28 (3): 253–75
Altana Pharma. Effect of roflumilast on pulmonary function and respiratory symptoms in patients with chronic obstructive pulmonary disease (COPD) (BY217/M2-110) [ClinicalTrials.gov identifier NCT00062582]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://www.clinicaltrials.gov/ct2/show/NCT00062582?id=NCT00062582&rank=1 [Accessed 2010 Jun 14]
Data on file, Nycomed GmbH, 2009
Efron B, Tibshirani RJ. An introduction to the bootstrap. Boca Raton (FL): Chapman and Hall/CRC, 1993
Wang DD, Zhang S. Standardized visual predictive check: how and when to use it model validation [abstract no. 1501]. 18th Annual Meeting, Population Approach Group in Europe; 2009 Jun 23–26; St Petersburg [online]. Available from URL: http://www.page-meeting.org/default.asp?abstract=1501 [Accessed 2010 Jun 14]
Maintenance and Support Services Organization. The medical dictionary for regulatory activities. Chantilly (VA): Northrop Grumman Corporation, 2010
Egan J. Signal detection theory and ROCanalysis. New York: Academic Press, 2010
Chambers JM, Cleveland WS, Kleiner B, et al. Graphical methods for data analysis. Boston (MA): Wadsworth & Brooks/Cole, 1983: 62
Bonate P. Pharmacokinetic-pharmacodynamic modeling and simulation. New York: Springer, 2006
Harris RZ, Benet LZ, Schwartz JB. Gender effects in pharmacokinetics and pharmacodynamics. Drugs 1995; 50 (2): 222–39
McCune JS, Lindley C, Decker JL, et al. Lack of gender differences and large intrasubject variability in cytochrome P450 activity measured by phenotyping with dextromethorphan. J Clin Pharmacol 2001; 41 (7): 723–31
Hunt CM, Westerkam WR, Stave GM. Effect of age and gender on the activity of human hepatic CYP3A. Biochem Pharmacol 1992; 44 (2): 275–83
Parkinson A, Mudra DR, Johnson C, et al. The effects of gender, age, ethnicity, and liver cirrhosis on cytochrome P450 enzyme activity in human liver microsomes and inducibility in cultured human hepatocytes. Toxicol Appl Pharmacol 2004; 199 (3): 193–209
Strum DP, Eger EI, Unadkat JD, et al. Age affects the pharmacokinetics of inhaled anesthetics in humans. Anesth Analg 1991; 73 (3): 310–8
Wynne HA, Mutch E, James OF, et al. The effect of age upon the affinity of microsomal mono-oxygenase enzymes for substrate in human liver. Age Ageing 1988; 17 (6): 401–5
Wandel C, Witte JS, Hall JM, et al. CYP3A activity in African American and European American men: population differences and functional effect of the CYP3A4*1B50-promoter region polymorphism. Clin Pharmacol Ther 2000; 68 (1): 82–91
Fabbri LM, Rabe KF. From COPD to chronic systemic inflammatory syndrome? Lancet 2007; 370 (9589): 797–9
Bhowmik A, Seemungal TA, Sapsford RJ, et al. Relation of sputum inflammatory markers to symptoms and lung function changes in COPD exacerbations. Thorax 2000; 55 (2): 114–20
Tracey KJ, Wei H, Manogue KR, et al. Cachectin/tumor necrosis factor induces cachexia, anemia, and inflammation. J Exp Med 1988; 167 (3): 1211–27
Masubuchi Y, Enoki K, Horie T. Down-regulation of hepatic cytochrome P450 enzymes in rats with trinitrobenzene sulfonic acid-induced colitis. Drug Metab Dispos 2008; 36 (3): 597–603
Nassr N, Lahu G, von Richter O, et al. Lack of a pharmacokinetic interaction between steady-state roflumilast and single-dose midazolam in healthy subjects. Br J Clin Pharmacol 2007; 63 (3): 365–70
Data on file, Nycomed GmbH, 2007
Böhmer GM, Nassr N, Wenger M, et al. The targeted oral, once-daily phosphodiesterase 4 inhibitor roflumilast and the leukotriene receptor antagonist montelukast do not exhibit significant pharmacokinetic interactions. J Clin Pharmacol 2009; 49 (4): 389–97
Bethke TD, Giessmann T, Westphal K, et al. Roflumilast, a once-daily oral phosphodiesterase 4 inhibitor, lacks relevant pharmacokinetic interactions with inhaled salbutamol when co-administered in healthy subjects. Int J Clin Pharmacol Ther 2006; 44 (11): 572–9
Nassr N, Hü nnemeyer A, Herzog R, et al. Effects of rifampicin on the pharmacokinetics of roflumilast and roflumilast N-oxide in healthy subjects. Br J Clin Pharmacol 2009; 68 (4): 580–7
Data on file, Nycomed GmbH, 2005
Data on file, Nycomed GmbH, 2002
Data on file, Nycomed GmbH, 2001
Data on file, Nycomed GmbH, 2000
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lahu, G., Hünnemeyer, A., Diletti, E. et al. Population Pharmacokinetic Modelling of Roflumilast and Roflumilast N-Oxide by Total Phosphodiesterase-4 Inhibitory Activity and Development of a Population Pharmacodynamic-Adverse Event Model. Clin Pharmacokinet 49, 589–606 (2010). https://doi.org/10.2165/11536600-000000000-00000
Published:
Issue Date:
DOI: https://doi.org/10.2165/11536600-000000000-00000