Abstract
Background
Colon cancer is the third and fourth most prevalent cancer among Iranian men and women, respectively. Suicide gene therapy is one of the alternative therapeutic modalities for cancer. The application of specific promoters for therapeutic genes should decrease the adverse effects of this modality.
Objectives
The combined aims of this study were to design a specific suicide gene therapy construct for colon cancer and study its effect in distinct representatives of transformed and nontransformed cells.
Study Design
The KRAS oncogene signaling pathway is one of the most important signaling pathways activated in colon cancer; therefore, we inserted the urokinase plasminogen activator receptor (uPAR; PLAUR gene) promoter as one of the upregulated promoters by this pathway upstream of a suicide gene (thymidine kinase [TK]) and a reporter gene (β-galactosidase, β-gal [LacZ]). This promoter is a natural combination of different motifs responsive to the RAS signaling pathway, such as the transcription factors AP1 (FOS/JUN), SP1, SP3, and AP2α, and nuclear factor kappa B (NFκB).
Results
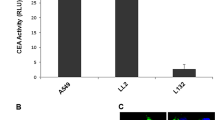
The reporter plasmid under the control of the uPAR promoter (PUCUPARLacZ) had the ability to express b-gal in colon cancer cells (human colon adenocarcinoma [SW480] and human colorectal carcinoma [HCT116] cell lines), while it could not express β-gal in nontransformed human umbilical vein endothelial cells (HUVEC) and normal colon cells. After confirming the ability of pUCUPARTK (suicide plasmid) to express TK in SW480 and HCT116 cells by real-time PCR, cytotoxicity assays showed that pUCUPARTK decreased the viability of these cells in the presence of ganciclovir 20 and 40 μg/mL (and higher), respectively. Although M30 CytoDEATH™ antibody could not detect a significant rate of apoptosis induced by ganciclovir in pUCUPARTK-transfected HCT116 cells, the percentage of stained cells was marked in comparison with untreated cells. While this antibody could detect apoptosis in HCT116 cell line transfected with positive control plasmid, it could not detect apoptosis in SW480 cells transfected with the same positive control. This discrepancy could be attributed to the different mechanisms of TK/ganciclovir-induced apoptosis in tumor protein p53 (TP53)-expressing (HCT116) and -deficient (SW480) cells. Annexin-propidium iodide staining could detect apoptosis in treated, pUCUPARTK-transfected SW480 and HCT116 cells.
Conclusion
This study showed that the uPAR promoter can be considered as a suitable candidate for specific suicide gene therapy of colon cancer and probably other cancers in which the RAS signaling pathway is involved in their carcinogenesis process.








Similar content being viewed by others
References
Sadjadi A, Nouraie M, Mohagheghi MA, et al. Cancer occurrence in Iran in 2002: an international perspective. Asian Pac J Cancer Prev 2005 Jul; 6(3): 359–63
Parker SL, Tong T, Bolden S, et al. Cancer statistics, 1996. CA Cancer J Clin 1996 Jan; 46(1): 5–27
Lipinski KS, Djeha AH, Ismail T, et al. High-level, beta-catenin/TCF-dependent transgene expression in secondary colorectal cancer tissue. Mol Ther 2001 Oct; 4(4): 365–71
Zeng ZJ, Li ZB, Luo SQ, et al. Retrovirus-mediated tk gene therapy of implanted human breast cancer in nude mice under the regulation of Tet-On. Cancer Gene Ther 2006 Mar; 13(3): 290–7
Wiewrodt R, Amin K, Kiefer M, et al. Adenovirus-mediated gene transfer of enhanced herpes simplex virus thymidine kinase mutants improves prodrug-mediated tumor cell killing. Cancer Gene Ther 2003 May; 10(5): 353–64
Sterman DH, Recio A, Vachani A, et al. Long-term follow-up of patients with malignant pleural mesothelioma receiving high-dose adenovirus herpes simplex thymidine kinase/ganciclovir suicide gene therapy. Clin Cancer Res 2005 Oct 15; 11(20): 7444–53
Konson A, Ben-Kasus T, Mahajna JA, et al. Herpes simplex virus thymidine kinase gene transduction enhances tumor growth rate and cyclooxygenase-2 expression in murine colon cancer cells. Cancer Gene Ther 2004 Dec; 11(12): 830–40
Freeman SM, Abboud CN, Whartenby KA, et al. The ‘bystander effect’: tumor regression when a fraction of the tumor mass is genetically modified. Cancer Res 1993 Nov 1; 53(21): 5274–83
Freytag SO, Stricker H, Peabody J, et al. Five-year follow-up of trial of replication-competent adenovirus-mediated suicide gene therapy for treatment of prostate cancer. Mol Ther 2007 Mar; 15(3): 636–42
Rainov NG. A phase III clinical evaluation of herpes simplex virus type 1 thymidine kinase and ganciclovir gene therapy as an adjuvant to surgical resection and radiation in adults with previously untreated glioblastoma multiforme. Hum Gene Ther 2000 Nov 20; 11(17): 2389–401
Zabala M, Wang L, Hernandez-Alcoceba R, et al. Optimization of the Tet-on system to regulate interleukin 12 expression in the liver for the treatment of hepatic tumors. Cancer Res 2004 Apr 15; 64(8): 2799–804
Ikegami S, Tadakuma T, Yamakami K, et al. Selective gene therapy for prostate cancer cells using liposomes conjugated with IgM type monoclonal antibody against prostate-specific membrane antigen. Hum Cell 2005 Mar; 18(1): 17–23
Nettelbeck DM, Jerome V, Muller R. Gene therapy: designer promoters for tumour targeting. Trends Genet 2000 Apr; 16(4): 174–81
Pang S, Taneja S, Dardashti K, et al. Prostate tissue specificity of the prostate-specific antigen promoter isolated from a patient with prostate cancer. Hum Gene Ther 1995 Nov; 6(11): 1417–26
Richards CA, Austin EA, Huber BE. Transcriptional regulatory sequences of carcinoembryonic antigen: identification and use with cytosine deaminase for tumor-specific gene therapy. Hum Gene Ther 1995 Jul; 6(7): 881–93
Adjei AA. Blocking oncogenic Ras signaling for cancer therapy. J Natl Cancer Inst 2001 Jul 18; 93(14): 1062–74
Arber N, Shapira I, Ratan J, et al. Activation of c-K-ras mutations in human gastrointestinal tumors. Gastroenterology 2000 Jun; 118(6): 1045–50
Duursma AM, Agami R. Ras interference as cancer therapy. Semin Cancer Biol 2003 Aug; 13(4): 267–73
Bos JL. The ras gene family and human carcinogenesis. Mutat Res 1988 May; 195(3): 255–71
Sutter T, Arber N, Moss SF, et al. Frequent K-ras mutations in small bowel adenocarcinomas. Dig Dis Sci 1996 Jan; 41(1): 115–8
Bos JL, Fearon ER, Hamilton SR, et al. Prevalence of ras gene mutations in human colorectal cancers. Nature 1987 May 28; 327(6120): 293–7
Gao HG, Chen JK, Stewart J, et al. Distribution of p53 and K-ras mutations in human lung cancer tissues. Carcinogenesis 1997 Mar; 18(3): 473–8
Beck SE, Jung BH, Del RE, et al. BMP-induced growth suppression in colon cancer cells is mediated by p21WAF1 stabilization and modulated by RAS/ERK. Cell Signal 2007 Jul; 19(7): 1465–72
Zuber J, Tchernitsa OI, Hinzmann B, et al. A genome-wide survey of RAS transformation targets. Nat Genet 2000 Feb; 24(2): 144–52
Pollock CB, Shirasawa S, Sasazuki T, et al. Oncogenic K-RAS is required to maintain changes in cytoskeletal organization, adhesion, and motility in colon cancer cells. Cancer Res 2005 Feb 15; 65(4): 1244–50
Wu M, Huang C, Li X, et al. LRRC4 inhibits glioblastoma cell proliferation, migration, and angiogenesis by downregulating pleiotropic cytokine expression and responses. J Cell Physiol 2008 Jan; 214(1): 65–74
Mesa Jr C, Mirza M, Mitsutake N, et al. Conditional activation of RET/PTC3 and BRAFV600E in thyroid cells is associated with gene expression profiles that predict a preferential role of BRAF in extracellular matrix remodeling. Cancer Res 2006 Jul 1; 66(13): 6521–9
Backlund MG, Mann JR, Wang D, et al. Ras up-regulation of cyclooxygenase-2. Methods Enzymol 2005; 407: 401–10
Gum R, Juarez J, Allgayer H, et al. Stimulation of urokinase-type plasminogen activator receptor expression by PMA requires JNK1-dependent and -independent signaling modules. Oncogene 1998 Jul 16; 17(2): 213–25
Campbell SL, Khosravi-Far R, Rossman KL, et al. Increasing complexity of Ras signaling. Oncogene 1998 Sep 17; 17: 1395–413
Bos JL. Ras oncogenes in human cancer: a review. Cancer Res 1989 Sep 1; 49(17): 4682–9
Neibergs HL, Hein DW, Spratt JS. Genetic profiling of colon cancer. J Surg Oncol 2002 Aug; 80(4): 204–13
Andreyev HJ, Ross PJ, Cunningham D, et al. Antisense treatment directed against mutated Ki-ras in human colorectal adenocarcinoma. Gut 2001 Feb; 48(2): 230–7
Dvory-Sobol H, Kazanov D, Arber N. Gene targeting approach to selectively kill colon cancer cells, with hyperactive K-Ras pathway. Biomed Pharma-cother 2005 Oct; 59Suppl. 2: S370–4
Reddy MA, Langer SJ, Colman MS, et al. An enhancer element responsive to ras and fms signaling pathways is composed of two distinct nuclear factor binding sites. Mol Endocrinol 1992 Jul; 6(7): 1051–60
Rosenberg S. New developments in the urokinase-type plasminogen activator system. Expert Opin Ther Targets 2001 Dec; 5(6): 711–22
Hagiwara H, Sato H, Shirai S, et al. Connexin 32 down-regulates the fibrinolytic factors in metastatic renal cell carcinoma cells. Life Sci 2006 Apr 4; 78(19): 2249–54
Barinka C, Parry G, Callahan J, et al. Structural basis of interaction between urokinase-type plasminogen activator and its receptor. J Mol Biol 2006 Oct 20; 363(2): 482–95
D’Alessio S, Margheri F, Pucci M, et al. Antisense oligodeoxynucleotides for urokinase-plasminogen activator receptor have anti-invasive and anti-proliferative effects in vitro and inhibit spontaneous metastases of human melanoma in mice. Int J Cancer 2004 May 20; 110(1): 125–33
Begum FD, Hogdall CK, Kjaer SK, et al. The prognostic value of plasma soluble urokinase plasminogen activator receptor (suPAR) levels in stage III ovarian cancer patients. Anticancer Res 2004 May; 24(3b): 1981–5
Bagheri-Yarmand R, Mazumdar A, Sahin AA, et al. LIM kinase 1 increases tumor metastasis of human breast cancer cells via regulation of the urokinase-type plasminogen activator system. Int J Cancer 2006 Jun 1; 118(11): 2703–10
Akahane T, Ishii M, Ohtani H, et al. Stromal expression of urokinase-type plasminogen activator receptor (uPAR) is associated with invasive growth in primary liver cancer. Liver 1998 Dec; 18(6): 414–9
Cantero D, Friess H, Deflorin J, et al. Enhanced expression of urokinase plasminogen activator and its receptor in pancreatic carcinoma. Br J Cancer 1997; 75(3): 388–95
Albo D, Tuszynski GP. Thrombospondin-1 up-regulates tumor cell invasion through the urokinase plasminogen activator receptor in head and neck cancer cells. J Surg Res 2004 Jul; 120(1): 21–6
Ahmed N, Oliva K, Wang Y, et al. Proteomic profiling of proteins associated with urokinase plasminogen activator receptor in a colon cancer cell line using an antisense approach. Proteomics 2003 Mar; 3(3): 288–98
Bauer TW, Fan F, Liu W, et al. Insulinlike growth factor-I-mediated migration and invasion of human colon carcinoma cells requires activation of c-Met and urokinase plasminogen activator receptor. Ann Surg 2005 May; 241(5): 748–56
Dass K, Ahmad A, Azmi AS, et al. Evolving role of uPA/uPAR system in human cancers. Cancer Treat Rev 2008 Apr; 34(2): 122–36
Allgayer H, Wang H, Shirasawa S, et al. Targeted disruption of the K-ras oncogene in an invasive colon cancer cell line down-regulates urokinase receptor expression and plasminogen-dependent proteolysis. Br J Cancer 1999 Aug; 80(12): 1884–91
Lengyel E, Stepp E, Gum R, et al. Involvement of a mitogen-activated protein kinase signaling pathway in the regulation of urokinase promoter activity by c-Ha-ras. J Biol Chem 1995 Sep 29; 270(39): 23007–12
Muller SM, Okan E, Jones P. Regulation of urokinase receptor transcription by Ras- and Rho-family GTPases. Biochem Biophys Res Commun 2000 Apr 21; 270(3): 892–8
Schewe DM, Biller T, Maurer G, et al. Combination analysis of activator protein-1 family members, Sp 1 and an activator protein-2alpha-related factor binding to different regions of the urokinase receptor gene in resected colorectal cancers. Clin Cancer Res 2005 Dec 15; 11 (24 Pt 1): 8538–48
Sambrook J, Russell DW. Molecular cloninga laboratory manual. 3rd ed. Cold Spring Harbor (NY): Cold Spring Harbor Laboratory Press, 2001
Azadmanesh K, Rohvand F, Amini S, et al. Evaluation of stimulatory effects of HTLV-I tax protein on CREB and NFKB related signaling pathways two B-glycosidase based reporter plasmids. Yakhteh Med J 2005; 6(24): 218–25
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using realtime quantitative PCR and the 2 CT method. Methods 2001; 25(4): 402–8
Bilbao R, Gerolami R, Bralet MP, et al. Transduction efficacy, antitumoral effect, and toxicity of adenovirus-mediated herpes simplex virus thymidine kinase/ganciclovir therapy of hepatocellular carcinoma: the woodchuck animal model. Cancer Gene Ther 2000 May; 7(5): 657–62
Hauck W, Stanners CP. Transcriptional regulation of the carcinoembryonic antigen gene. Identification of regulatory elements and multiple nuclear factors. J Biol Chem 1995 Feb 24; 270(8): 3602–10
Dabrowska A, Szary J, Kowalczuk M et al. CEA-negative glioblastoma and melanoma cells are sensitive to cytosine deaminase/5-fluorocytosine therapy directed by the carcinoembryonic antigen promoter. Acta Biochim Pol 2004; 51(3): 723–34
Ueda K, Iwahashi M, Nakamori M, et al. Improvement of carcinoembryonic antigen-specific prodrug gene therapy for experimental colon cancer. Surgery 2003 Mar; 133(3): 309–17
Block A, Milasinovic D, Mueller J et al. Amplified Muc1-specific gene expression in colon cancer cells utilizing a binary system in adenoviral vectors. Anticancer Res 2002 Nov–Dec; 22(6A): 3285–92
Yamamoto M, Alemany R, Adachi Y, et al. Characterization of the cyclooxy-genase-2 promoter in an adenoviral vector and its application for the mitigation of toxicity in suicide gene therapy of gastrointestinal cancers. Mol Ther 2001 Mar; 3(3): 385–94
Lehmann K, Janda E, Pierreux CE, et al. Raf induces TGFbeta production while blocking its apoptotic but not invasive responses: a mechanism leading to increased malignancy in epithelial cells. Genes Dev 2000 Oct 15; 14(20): 2610–22
Ahnen DJ, Feigl P, Quan G, et al. Ki-ras mutation and p53 overexpression predict the clinical behavior of colorectal cancer: a Southwest Oncology Group study. Cancer Res 1998 Mar 15; 58(6): 1149–58
Shirasawa S, Furuse M, Yokoyama N, et al. Altered growth of human colon cancer cell lines disrupted at activated Ki-ras. Science 1993 Apr 2; 260(5104): 85–8
Dumler I, Weis A, Mayboroda OA, et al. The Jak/Stat pathway and urokinase receptor signaling in human aortic vascular smooth muscle cells. J Biol Chem 1998 Jan 2; 273(1): 315–21
Koshelnick Y, Ehart M, Stockinger H, et al. Mechanisms of signaling through urokinase receptor and the cellular response. Thromb Haemost 1999 Aug; 82(2): 305–11
Tang H, Kerins DM, Hao Q, et al. The urokinase-type plasminogen activator receptor mediates tyrosine phosphorylation of focal adhesion proteins and activation of mitogen-activated protein kinase in cultured endothelial cells. J Biol Chem 1998 Jul 17; 273(29): 18268–72
Koch A, Waha A, Hartmann W, et al. Elevated expression of Wnt antagonists is a common event in hepatoblastomas. Clin Cancer Res 2005 Jun 15; 11(12): 4295–304
Wang Y, Dang J, Johnson LK, et al. Structure of the human urokinase receptor gene and its similarity to CD59 and the Ly-6 family. Eur J Biochem 1995 Jan 15; 227(1-2): 116–22
Li CY, Tan L, Zhang GJ, et al. Transcriptional regulation of urokinase receptor in high- (95D) and low-metastatic (95C) human lung cancer cells. Acta Biochim Biophys Sin (Shanghai) 2004 Jun; 36(6): 405–11
Allgayer H, Wang H, Wang Y, et al. Transactivation of the urokinase-type plasminogen activator receptor gene through a novel promoter motif bound with an activator protein-2alpha-related factor. J Biol Chem 1999 Feb 19; 274(8): 4702–14
Leupold JH, Asangani I, Maurer GD, et al. Src induces urokinase receptor gene expression and invasion/intravasation via activator protein-1/p-c-Jun in colorectal cancer. Mol Cancer Res 2007 May; 5(5): 485–96
Soravia E, Grebe A, De LP, et al. A conserved TATA-less proximal promoter drives basal transcription from the urokinase-type plasminogen activator receptor gene. Blood 1995 Jul 15; 86(2): 624–35
Dang J, Boyd D, Wang H, et al. A region between -141 and -61 bp containing a proximal AP-1 is essential for constitutive expression of urokinase-type plasminogen activator receptor. Eur J Biochem 1999 Aug; 264(1): 92–9
Allgayer H, Wang H, Gallick GE, et al. Transcriptional induction of the urokinase receptor gene by a constitutively active Src: requirement of an upstream motif (—152/—13 5) bound with Sp1. J Biol Chem 1999 Jun 25; 274(26): 18428–37
Wang Y, Dang J, Wang H, et al. Identification of a novel nuclear factor-kappaB sequence involved in expression of urokinase-type plasminogen activator receptor. Eur J Biochem 2000 Jun; 267(11): 3248–54
Hapke S, Gawaz M, Dehne K, et al. beta(3)A-integrin downregulates the urokinase-type plasminogen activator receptor (u-PAR) through a PEA3/ets transcriptional silencing element in the u-PAR promoter. Mol Cell Biol 2001 Mar; 21(6): 2118–32
Schewe DM, Leupold JH, Boyd DD, et al. Tumor-specific transcription factor binding to an activator protein-2/Sp 1 element of the urokinase-type plasminogen activator receptor promoter in a first large series of resected gastrointestinal cancers. Clin Cancer Res 2003 Jun; 9(6): 2267–76
Malerba M, Nikolova D, Cornelis J, et al. Targeting of autonomous parvo-viruses to colon cancer by insertion of Tcf sites in the P4 promoter. Cancer Gene Ther 2006 Mar; 13(3): 273–80
Bienz M, Clevers H. Linking colorectal cancer to Wnt signaling. Cell 2000 Oct 13; 103(2): 311–20
Uch R, Gerolami R, Faivre J, et al. Hepatoma cell-specific ganciclovir-mediated toxicity of a lentivirally transduced HSV-TkEGFP fusion protein gene placed under the control of rat alpha-fetoprotein gene regulatory sequences. Cancer Gene Ther 2003 Sep; 10(9): 689–95
Thust R, Tomicic M, Klocking R, et al. Comparison of the genotoxic and apoptosis-inducing properties of ganciclovir and penciclovir in Chinese hamster ovary cells transfected with the thymidine kinase gene of herpes simplex virus-1: implications for gene therapeutic approaches. Cancer Gene Ther 2000 Jan; 7(1): 107–17
Caulin C, Salvesen GS, Oshima RG. Caspase cleavage of keratin 18 and reorganization of intermediate filaments during epithelial cell apoptosis. J Cell Biol 1997 Sep 22; 138(6): 1379–94
Beltinger C, Fulda S, Kammertoens T, et al. Herpes simplex virus thymidine kinase/ganciclovir-induced apoptosis involves ligand-independent death receptor aggregation and activation of caspases. Proc Natl Acad Sci U S A 1999 Jul 20; 96(15): 8699–704
Wei SJ, Chao Y, Shih YL, et al. Involvement of Fas (CD95/APO-1) and Fas ligand in apoptosis induced by ganciclovir treatment of tumor cells transduced with herpes simplex virus thymidine kinase. Gene Ther 1999 Mar; 6(3): 420–31
Tomicic MT, Thust R, Kaina B. Ganciclovir-induced apoptosis in HSV-1 thymidine kinase expressing cells: critical role of DNA breaks, Bcl-2 decline and caspase-9 activation. Oncogene 2002 Mar 28; 21(14): 2141–53
Acknowledgments
This research was funded by the Pasteur Institute of Iran. We hereby thank Mrs Maryam Noorayee Kia (Head Nurse of 1st Surgery Ward, Imam Khomeini Hospital, Tehran University of Medical Sciences, Tehran, Iran), Mrs Leyli Ghaffarpour (Head Nurse of 1st Operating Room, Imam Khomeini Hospital), and other nurses in these wards for their kind help in obtaining the normal colon tissue. We also thank Dr Ahmad Kaviani (1st Surgery Ward, Imam Khomeini Hospital) for his kind help in obtaining the colon tissue during an operation.
The authors have no conflicts of interest that are directly relevant to the content of this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Teimoori-Toolabi, L., Azadmanesh, K., Amanzadeh, A. et al. Selective Suicide Gene Therapy of Colon Cancer Exploiting the Urokinase Plasminogen Activator Receptor Promoter. BioDrugs 24, 131–146 (2010). https://doi.org/10.2165/11530840-000000000-00000
Published:
Issue Date:
DOI: https://doi.org/10.2165/11530840-000000000-00000