Abstract
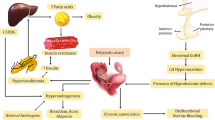
Polycystic ovary syndrome (PCOS) is a common heterogeneous disorder characterized by hyperandrogenism and chronic anovulation. The syndrome is frequently associated with an increased risk for insulin resistance and type 2 diabetes mellitus; obesity exacerbates insulin resistance and favors the progression from impaired glucose tolerance to diabetes in these patients. In young women, precocious pubarche and hyperinsulinemia are early manifestations of PCOS.
The familial clustering of women with PCOS suggests that heredity is implicated in the origin of the syndrome. However, genetic approaches to its pathogenesis have been hampered by the heterogeneity of phenotypic features within families, and the lack of uniform criteria for diagnosis.
Currently, PCOS is considered a polygenic trait that might result from the interaction of susceptibility and protective genomic variants under the influence of environmental factors. Both linkage analysis and association studies are valid tools for the study of the genetics of PCOS.
Candidate genes for PCOS include those related to androgenic pathways and metabolic associations of the syndrome. More recently, genes encoding inflammatory cytokines have been identified as target genes for PCOS, as proinflammatory genotypes and phenotypes are also associated with obesity, insulin resistance, type 2 diabetes, PCOS, and increased cardiovascular risk.
This paper reviews the candidate genes involved in the metabolic pathways that are altered in patients with PCOS. Despite a significant amount of research in this area, none of the genes studied so far has been identified as the PCOS susceptibility gene for the majority of cases. PCOS is the first component of the metabolic syndrome to be detected in many women, so the identification and correct diagnosis of PCOS has important preventive and therapeutic implications for the affected women and their families. In the future, new therapeutic approaches to PCOS will rely on knowing the genes, environmental influences, and etiologic mechanisms associated with the disorder.



Similar content being viewed by others
References
Carmina E, Lobo RA. Polycystic ovary syndrome (PCOS): arguably the most common endocrinopathy is associated with significant morbidity in women. J Clin Endocrinol Metab 1999; 84: 1897–9
Zawadski JK, Dunaif A. Diagnostic criteria for polycystic ovary syndrome: towards a rational approach. In: Dunaif A, Givens JR, Haseltime F, et al., editors. Polycystic ovary syndrome. Boston (MA): Blackwell, 1992: 377–84
Homburg R. What is polycystic ovarian syndrome? A proposal for a consensus on the definition and diagnosis of polycystic ovarian syndrome. Hum Reprod 2002; 17: 2495–9
Adams J, Poison DW, Abdulwahid N, et al. Multifollicular ovaries: clinical and endocrine features and response to pulsatile gonadotrophin releasing hormone. Lancet 1985; II: 1375–8
Adams J, Poison DW, Franks S. Prevalence of polycystic ovaries in women with anovulation and idiopathic hirsutism. BMJ Clin Res Ed 1986; 293: 355–9
Gadir AA, Khatim MS, Mowafi RS, et al. Implications of ultrasonically diagnosed polycystic ovaries: I. Correlations with basal hormonal profiles. Hum Reprod 1992; 7: 453–7
O’Driscoll JB, Mamtora H, Higginson J, et al. A prospective study of the prevalence of clear-cut endocrine disorders and polycystic ovaries in 350 patients presenting with hirsutism or androgenic alopecia. Clin Endocrinol (Oxf) 1994; 41: 231–6
Bunker CB, Newton J, Kilborn J, et al. Most women with acne have polycystic ovaries. Br J Dermatol 1989; 121: 675–80
Ehrmann DA, Rosenfield RL, Barnes RB, et al. Detection of functional ovarian hyperandrogenism in women with androgen excess. N Engl J Med 1992; 327: 157–62
Poison DW, Adams J, Wadsworth J, et al. Polycystic ovaries: a common finding in normal women. Lancet 1988; I: 870–2
Azziz R, Bradley ELJ, Potter HD, et al. Adrenal androgen excess in women: lack of a role for 17-hydroxylase and 17,20-lyase dysregulation. J Clin Endocrinol Metab 1995; 80: 400–5
Dewailly D. Definition and significance of polycystic ovaries. Baillière’s Clin Obstetr Gynaecol 1997; 11: 349–67
Koivunen R, Laatikainen T, Tomas C, et al. The prevalence of polycystic ovaries in healthy women. Acta Obstet Gynecol Scand 1999; 78: 137–41
Parker CRJ, Slayden SM, Azziz R, et al. Effects of aging on adrenal function in the human: responsiveness and sensitivity of adrenal androgens and cortisol to adrenocorticotropin in premenopausal and postmenopausal women. J Clin Endocrinol Metab 2000; 85: 48–54
Franks S. Polycystic ovary syndrome. N Engl J Med 1995; 333: 853–61
Knochenhauer ES, Key TJ, Kahsar-Miller M, et al. Prevalence of the polycystic ovary syndrome in unselected black and white women of the southeastern United States: a prospective study. J Clin Endocrinol Metab 1998; 83: 3078–82
Asunción M, Calvo RM, San Millán JL, et al. A prospective study of the prevalence of the polycystic ovary syndrome in unselected Caucasian women from Spain. J Clin Endocrinol Metab 2000; 85: 2434–8
Diamanti-Kandarakis E, Kouli CR, Bergiele AT, et al. A survey of the polycystic ovary syndrome in the Greek island of Lesbos: hormonal and metabolic profile. J Clin Endocrinol Metab 1999; 84: 4006–11
Ehrmann DA, Barnes RB, Rosenfield RL. Polycystic ovary syndrome as a form of functional hyperandrogenism due to dysregulation of androgen secretion. Endocr Rev 1995; 16: 322–53
Ibañez L, Potau N, Virdis R, et al. Postpubertal outcome in girls diagnosed of premature pubarche during childhood: increased frequency of functional ovarian hyperandrogenism. J Clin Endocrinol Metab 1993; 76: 1599–603
Lewy VD, Danadian K, Witchel SF, et al. Early metabolic abnormalities in adolescent girls with polycystic ovarian syndrome. J Pediatr 2001; 138: 38–44
Palmert MR, Gordon CM, Kartashov AI, et al. Screening for abnormal glucose tolerance in adolescents with polycystic ovary syndrome. J Clin Endocrinol Metab 2002; 87: 1017–23
Lapillonne A, Braillon P, Chatelain PG, et al. Body composition in appropriate and small for gestational age infants. Acta Paediatr 1997; 86: 196–200
McGarry JD. Disordered metabolism in diabetes: have we underemphasized the fat component? J Cell Biochem 1994; 55S: 29–38
Ibañez L, Potau N, Francois I, et al. Precocious pubarche, hyperinsulinism and ovarian hyperandrogenism in girls: relation to reduced fetal growth. J Clin Endocrinol Metab 1998; 83: 3558–62
Jaquet D, Leger J, Chevenne D, et al. Intrauterine growth retardation predisposes to insulin resistance but not to hyperandrogenism in young women. J Clin Endocrinol Metab 1999; 84: 3945–9
Rosenfield RL, Barnes RB, Cara JF, et al. Dysregulation of P450c17α as a cause of polycystic ovarian syndrome. Fertil Steril 1990; 53: 785–91
Moghetti P, Castello R, Negri C, et al. Insulin infusion amplifies 17 alpha-hydroxycorticosteroid intermediates response to adrenocorticotropin in hyperandrogenic women: apparent relative impairment of 17,20-lyase activity. J Clin Endocrinol Metab 1996; 81: 881–6
Adashi EY, Hsueh AJ, Yen SS. Insulin enhancement of luteinizing hormone and follicle-stimulating release by cultured pituitary cells. Endocrinology 1981; 108: 1441–9
Adashi EY, Resnick CE, D’Ercole AJ, et al. Insulin-like growth factors as intraovarian regulators of granulosa cell growth and function. Endocr Rev 1985; 6: 400–20
Kiddy DS, Sharp PS, White DM, et al. Differences in clinical and endocrine features between obese and non-obese subjects with polycystic ovary syndrome: an analysis of 263 consecutive cases. Clin Endocrinol (Oxf) 1990; 32: 213–20
Nestler JE, Powers LP, Matt DW, et al. A direct effect of hyperinsulinemia on serum sex hormone-binding globulin levels in obese women with the polycystic ovary syndrome. J Clin Endocrinol Metab 1991; 72: 83–9
Reaven GM. Banting lecture: role of insulin resistance in human disease. Diabetes 1988; 37: 1595–607
Legro RS. Polycystic ovary syndrome and cardiovascular disease: a premature association? Endocr Rev 2003; 24: 302–12
Nestler JE. Should patients with polycystic ovarian syndrome be treated with metformin? Hum Reprod 2002; 17: 1950–3
Legro RS, Kunselman AR, Dunaif A. Prevalence and predictors of dyslipidemia in women with polycystic ovary syndrome. Am J Med 2001; 111: 607–13
Ehrmann DA, Barnes RB, Rosenfield RL, et al. Prevalence of impaired glucose tolerance and diabetes in women with polycystic ovary syndrome. Diabetes Care 1999; 22: 141–6
Dunaif A, Graf M, Mandeli J, et al. Characterization of groups of hyperandrogenic women with acanthosis nigricans, impaired glucose tolerance and/or hyperinsulinemia. J Clin Endocrinol Metab 1987; 65: 499–507
Norman RJ, Masters L, Milner CR, et al. Relative risk of conversion from normoglycaemia to impaired glucose tolerance or non-insulin dependent diabetes mellitus in polycystic ovarian syndrome. Hum Reprod 2001; 16: 1995–8
Legro RS, Kunselman AR, Dodson WC, et al. Prevalence and predictors of risk for type 2 diabetes mellitus and impaired glucose tolerance in polycystic ovary syndrome: a prospective, controlled study in 254 affected women. J Clin Endocrinol Metab 1999; 84: 165–9
Balen AH, Conway GS, Kaltsas G, et al. Polycystic ovary syndrome: the spectrum of the disorder in 1741 patients. Hum Reprod 1995; 10: 2107–11
Conway GS, Agrawal R, Betteridge DJ, et al. Dyslipidaemia is associated with insulin resistance in women with polycystic ovaries. Clin Endocrinol (Oxf) 1992; 37: 119–25
Holte J, Bergh T, Berne C, et al. Serum lipoprotein lipid profile in women with the polycystic ovary syndrome: relation to anthropometric, endocrine and metabolic variables. Clin Endocrinol (Oxf) 1994; 41: 463–71
Cooper DN, Clayton JF. DNA polymorphism and the study of disease associations. Hum Genet 1988; 78: 299–312
Ferriman D, Purdie AW. The inheritance of polycystic ovarian disease and a possible relationship to premature balding. Clin Endocrinol 1979; 11: 291–300
Lunde O, Magnus P, Sandvik L, et al. Familial clustering in the polycystic ovarian syndrome. Gynecol Obstet Invest 1989; 28: 23–30
Hague WM, Adams J, Reeders ST, et al. Familial polycystic ovaries: a genetic disease? Clin Endocrinol 1988; 29: 593–605
Carey AH, Chan KL, Short F, et al. Evidence for a single gene effect in polycystic ovaries an male pattern baldness. Clin Endocrinol 1993; 38: 653–8
Norman RJ, Masters S, Hague W. Hyperinsulinemia is common in family members of women with polycystic ovary syndrome. Fertil Steril 1996; 66: 942–7
Govind A, Obhrai MS, Clayton RN. Polycystic ovaries are inherited as an autosomal dominant trait: analysis of 29 polycystic ovary syndrome and 10 control families. J Clin Endocrinol Metab 1999; 84: 38–43
Givens JR. Familial polycystic ovarian disease. Endocrinol Metab Clin North Am 1988; 17: 771–83
Legro RS, Driscoll D, Strauss JFI, et al. Evidence for a genetic basis for hyperadrogenemia in polycystic ovary syndrome. Proc Natl Acad Sci U S A 1998; 95: 14956–60
Kahsar-Miller MD, Nixon C, Boots LR, et al. Prevalence of polycystic ovary syndrome (PCOS) in first-degree relatives of patients with PCOS. Fertil Steril 2001; 75: 53–8
Wright AF, Carothers AD, Pirastu M. Population choice in mapping genes for complex disease. Nat Genet 1999; 23: 397–404
Seminara SB, Crowley WF. Genetic approaches to unraveling reproductive disorders: example of bedside to bench research in the genomic era. Endocr Rev 2002; 23: 382–92
Simpson JL. Molecular approach to common causes of female infertility. Best Pract Res Clin Obstet Gynaecol 2002; 16: 685–702
Arnold A. Molecular human genetics: introduction to molecular & cellular research course [online]. Washington: MeetingCast. Available from URL: http://www.meetingcast.com/bassci/ [Accessed 2004 Mar 8]
Spielman RS, McGinnis RE, Ewens WJ. Transmission test for linkage disequilibrium: the insulin gene region and insulin-dependent diabetes mellitus (IDDM). Am J Hum Genet 1993; 52: 506–16
Greenberg DA. Linkage analysis of “necessary” disease loci versus “susceptibility” loci. Am J Hum Genet 1993; 52: 135–43
Franks S, Gharani N, McCarthy M. Candidate genes in polycystic ovary syndrome. Hum Repr Update 2001; 7: 405–10
Legro RS, Bentley-Lewis R, Driscoll D, et al. Insulin resistance in the sisters of women with polycystic ovary syndrome: association with hyperandrogenemia rather than menstrual irregularity. J Clin Endocrinol Metab 2002; 87: 2128–33
Yildiz BO, Yarali H, Oguz H, et al. Glucose intolerance, insulin resistance, and hyperandrogenemia in first degree relatives of women with polycystic ovary syndrome. J Clin Endocrinol Metab 2003; 88: 2031–6
Legro RS, Kunselman AR, Demers L, et al. Elevated dehydroepiandrosterone sulfate levels as the reproductive phenotype in the brothers of women with polycystic ovary syndrome. J Clin Endocrinol Metab 2002; 87: 2134–8
Witchel SF, Smith R, Tomboc M, et al. Candidate gene analysis in premature pubarche and adolescent hyperandrogenism. Fertil Steril 2001; 75: 724–30
Urbanek M, Legro RS, Driscoll DA, et al. Thirty-seven candidate genes for polycystic ovary syndrome: strongest evidence for linkage is with follistatin. Proc Natl Acad Sci U S A 1999; 96: 8573–8
Urbanek M, Legro RS, Driscoll D, et al. Searching for the polycystic ovary syndrome genes. J Pediatr Endocrinol Metab 2000; 13Suppl. 5: 1311–3
Urbanek M, Wu X, Vickery KR, et al. Allelic variants of the follistatin gene in polycystic ovary syndrome. J Clin Endocrinol Metab 2000; 85: 4455–61
Calvo RM, Villuendas G, Sancho J, et al. Role of the follistatin gene in women with polycystic ovary syndrome. Fertil Steril 2001; 75: 1020–3
Dunaif A, Segal KR, Futterweit W, et al. Profound peripheral insulin resistance, independent of obesity, in polycystic ovary syndrome. Diabetes 1989; 38: 1165–74
Sorbara L, Weiss D, Delson R, et al. Polycystic ovary syndrome, ethnicity and insulin action [abstract]. 14th International Diabetes Federation Congress Satellite on Epidemiology of Diabetes; 1991; Williamsburg (VA)
Legro RS. Polycystic ovary syndrome: phenotype to genotype. Endocrinol Metab Clin North Am 1999; 28: 379–96
Urbanek M, Spielman RS. Genetic analysis of candidate genes for the polycystic ovary syndrome. Curr Opin Endocrinol Metab 2002; 9: 492–501
Legro RS, Strauss JF. Molecular progress in infertility: polycystic ovary syndrome. Fertil Steril 2002; 78: 569–76
Xita N, Georgiou I, Tsatsoulis A. The genetic basis of polycystic ovary syndrome. Eur J Endocrinol 2002; 147: 717–25
Franks S, Gharani N, Waterworth D, et al. Genetics of polycystic ovary syndrome. Mol Cell Endocrinol 1998; 145: 123–8
HUGO Gene Nomenclature Committee (HGNC). Genew: the Human Gene Nomenclature Database [online]. Available from URL: http://www.gene.ucl.ac.uk/cgi-bin/nomenclature/searchgenes.pl [Accessed 2004 Mar 16]
Waterworth DM, Bennett ST, Gharani N, et al. Linkage and association of insulin gene VNTR polymorphism with polycystic ovary syndrome. Lancet 1997; 349: 986–90
Ibañez L, Ong K, Potau N, et al. Insulin gene variable number of tandem repeat genotype and the low birth weight, precocious pubarche and hyperinsulinism sequence. J Clin Endocrinol Metab 2001; 86: 5788–93
Calvo RM, Teilería D, Sancho J, et al. Insulin gene variable number of tandem repeats (VNTR) regulatory polymorphism is not associated with hyperandrogenism in Spanish women. Fertil Steril 2002; 77: 666–8
Siegel S, Futterweit W, Davies TF, et al. A C/T single nucleotide polymorphism at the tyrosine kinase domain of the insulin receptor gene is associated with polycystic ovary syndrome. Fertil Steril 2002; 78: 1240–3
Tucci S, Futterweit W, Concepcion ES, et al. Evidence for association of polycystic ovary syndrome in Caucasian women with a marker at the insulin receptor locus. J Clin Endocrinol Metab 2001; 86: 446–9
Villuendas G, Escobar-Morreale HF, Tosi F, et al. Association between the D19S884 marker at the insulin receptor gene locus and polycystic ovary syndrome. Fertil Steril 2003; 79: 219–20
El Mkadem SA, Lautier C, Macari F, et al. Role of allelic variants Gly972Arg of IRS-1 and Gly1057Asp of IRS-2 in moderate-to-severe insulin resistance of women with polycystic ovary syndrome. Diabetes 2001; 50: 2164–8
Ehrmann DA, Tang X, Yoshiuchi I, et al. Relationship of insulin receptor substrate-1 and -2 genotypes to phenotypic features of polycystic ovary syndrome. J Clin Endocrinol Metab 2002; 87: 4297–300
San Millán J, Corton M, Villuendas G, et al. Association of the polycystic ovary syndrome (PCOS) with genomic variants related to insulin resistance, diabetes mellitus and obesity. J Clin Endocrinol Metab. In press
Ehrmann DA, Schwarz PE, Hara M, et al. Relation of calpain-10 genotype to phenotypic features of polycystic ovary syndrome. J Clin Endocrinol Metab 2002; 87: 1669–73
Gonzalez A, Abril E, Roca A, et al. Comment: CAPN10 alleles are associated with polycystic ovary syndrome. J Clin Endocrinol Metab 2002; 87: 3971–6
Escobar-Morreale HF, Peral B, Villuendas G, et al. Common single nucleotide polymorphisms in intron 3 of the calpain-10 gene influence hirsutism. Fertil Steril 2002; 77: 581–7
Haddad L, Evans JC, Gharani N, et al. Variation within the type 2 diabetes susceptibility gene calpain-10 and polycystic ovary syndrome. J Clin Endocrinol Metab 2002; 87: 2606–10
Hara M, Alcoser SY, Qaadir A, et al. Insulin resistance is attenuated in women with polycystic ovary syndrome with the Pro(12)Ala polymorphism in the PPARgamma gene. J Clin Endocrinol Metab 2002; 87: 772–5
Korhonen S, Heinonen S, Hiltunen M, et al. Polymorphism in the peroxisome proliferator-activated receptor-gamma gene in women with polycystic ovary syndrome. Hum Reprod 2003; 18: 540–3
Witchel SF, White C, Siegel ME, et al. Inconsistent effects of the proline12 —> alanine variant of the peroxisome proliferator-activated receptor-gamma2 gene on body mass index in children and adolescent girls. Fertil Steril 2001; 76:741–7
Witchel SF, Trivedi RN, Kammerer C. Frequency of the T228A polymorphism in the SORBS1 gene in children with premature pubarche and in adolescent girls with hyperandrogenism. Fertil Steril 2003; 80: 128–32
Oksanen L, Tiitinen A, Kaprio J, et al. No evidence for mutations of the leptin or leptin receptor genes in women with polycystic ovary syndrome. Mol Hum Reprod 2000; 6: 873–6
Rajkhowa M, Talbot JA, Jones PW, et al. Polymorphism of glycogen synthetase gene in polycystic ovary syndrome. Clin Endocrinol (Oxf) 1996; 44: 85–90
Urbanek M, Du Y, Silander K, et al. Variation in resistin gene promoter not associated with polycystic ovary syndrome. Diabetes 2003; 52: 214–7
Milner CR, Craig JE, Hussey ND, et al. No association between the −308 polymorphism in the tumor necrosis factor alpha (TNF-alpha) promoter region and polycystic ovaries. Mol Hum Reprod 1999; 5: 5–9
Escobar-Morreale HF, Calvo R, Sancho J, et al. TNF-α and hyperandrogenism: a clinical, biochemical, and molecular genetic study. J Clin Endocrinol Metab 2001; 86: 3761–7
Peral B, San Millán JL, Castello R, et al. The methionine 196 arginine polymorphism in exon 6 of the TNF receptor 2 gene (TNFRSF1B) is associated with the polycystic ovary syndrome and hyperandrogenism. J Clin Endocrinol Metab 2002; 87: 3977–83
Villuendas G, San Millán JL, Sancho J, et al. The −597 G right arrow A and −174 G right arrow C polymorphism in the promoter of the IL-6 gene are associated with hyperandrogenism. J Clin Endocrinol Metab 2002; 87: 1134–41
Escobar-Morreale HF, Calvo RM, Villuendas G, et al. Association of polymorphisms in the interleukin 6 receptor complex with obesity and hyperandrogenism. Obes Res 2003; 11: 987–96
Dunaif A. Insulin resistance and the polycystic ovary syndrome: mechanism and implications for pathogenesis. Endocr Rev 1997; 18: 774–800
Escobar-Morreale HF, Villuendas G, Botella-Carretero JI, et al. Obesity, and not insulin resistance, is the major determinant of serum inflammatory cardiovascular risk markers in pre-menopausal womem. Diabetologia 2003; 46: 625–33
Talbot JA, Bicknell EJ, Rajkhowa M, et al. Molecular scanning of the insulin receptor gene in women with polycystic ovary syndrome. J Clin Endocrinol Metab 1996; 81: 1979–83
Takata Y, Webster NJG, Olefsky JM. Mutation of the two carboxyl-terminal tyrosines results in an insulin receptor with normal metabolic signaling but enhanced mitogenic signaling properties. J Biol Chem 1991; 266: 9135–8
Moller DE. Potential role of TNF-alpha in the pathogenesis of insulin resistance and type 2 diabetes. Trends Endocrinol Metab 2000; 11: 212–7
Bennett ST, Lucassen AM, Gough SC, et al. Susceptibility to human type 1 diabetes at IDDM2 is determined by tandem repeat variation at the insulin gene minisatellite locus. Nat Genet 1995; 9: 284–92
Bennett ST, Todd JA, Waterworth DM, et al. Association of the insulin gene VNTR with polycystic ovary syndrome [letter]. Lancet 1997; 349: 1771–2
Dunger DB, Ong KKL, Huxtable SJ, et al. Association of the INS VNTR with size at birth. Nat Genet 1998; 19: 98–100
Ong KK, Phillips DI, Fall C, et al. The insulin gene VNTR, type 2 diabetes and birth weight. Nat Genet 1999; 21: 262–3
Weaver JU, Kopelman PG, Hitman GA. Central obesity and hyperinsulinaemia in women are associated with polymorphism in the 5′ flanking region of the human insulin gene. Eur J Clin Invest 1992; 22: 265–70
Dunaif A, Wu X, Lee A, et al. Defects in insulin receptor signaling in vivo in the polycystic ovary syndrome (PCOS). Am J Physiol 2001; 281: E392–9
Ibáñez L, Marcos MV, Potau N, et al. Increased frequency of the G972R variant of the insulin receptor substrate-1 (IRS-1) gene among girls with a history of precocious pubarche. Fert Steril 2002; 78: 1288–93
Vafiadis P, Bennett ST, Todd JA, et al. Divergence between genetic determinants of IGF2 transcription levels in leukocytes and of IDDM2-encoded susceptibility to type 1 diabetes. J Clin Endocrinol Metab 1998; 83: 2933–9
O’Dell SD, Miller GJ, Cooper JA, et al. Apal polymorphism in insulin-like growth factor II (IGF2) gene and weight in middle-aged males. Int J Obes Relat Metab Disord 1997; 21: 822–5
Mesiano S, Katz SL, Lee JY, et al. Insulin-like growth factors augment steroid production and expression of steroidogenic enzymes in human fetal adrenal cortical cells: implications for adrenal androgen regulation. J Clin Endocrinol Metab 1997; 82: 1390–6
l’Allemand D, Penhoat A, Lebrethon MC, et al. Insulin-like growth factors enhance steroidogenic enzyme and corticotropin receptor messenger ribonucleic acid levels and corticotropin steroidogenic responsiveness in cultured human adrenocortical cells. J Clin Endocrinol Metab 1996; 81: 3892–7
Cara JF. Insulin-like growth factors, insulin-like growth factor binding proteins and ovarian androgen production. Horm Res 1994; 42: 49–54
Barreca A, Del Monte P, Ponzani P, et al. Intrafollicular insulin-like growth factor-II levels in normally ovulating women and in patients with polycystic ovary syndrome. Fertil Steril 1996; 65: 739–45
Sreenan SK, Zhou YP, Otani K, et al. Calpains play a role in insulin secretion and action. Diabetes 2001; 50: 2013–20
Horikawa Y, Oda N, Cox NJ, et al. Genetic variation in the gene encoding calpain-10 is associated with type 2 diabetes mellitus. Nat Genet 2000; 26: 163–75
Gonzalez A, Abril E, Roca A, et al. CAPN10 alleles are associated with polycystic ovary syndrome. J Clin Endocrinol Metab 2002; 87: 3971–6
Gonzalez A, Abril E, Roca A, et al. Specific CAPN10 gene haplotypes influence the clinical profile of polycystic ovary patients. J Clin Endocrinol Metab 2003; 88: 5529–36
Ek J, Urhammer SA, Sorensen TI, et al. Homozygosity of the Pro12Ala variant of the peroxisome proliferation-activated receptor-gamma2 (PPAR-gamma2): divergent modulating effects on body mass index in obese and lean Caucasian men. Diabetologia 1999; 42: 892–5
Ek J, Andersen G, Urhammer SA, et al. Studies of the Pro12Ala polymorphism of the peroxisome proliferator-activated receptor-gamma2 (PPAR-gamma2) gene in relation to insulin sensitivity among glucose tolerant Caucasians. Diabetologia 2001; 44: 1170–6
Lin WH, Chiu KC, Chang HM, et al. Molecular scanning of the human sorbin and SH3-domain-containing-1 (SORBS1) gene: positive association of the T228A polymorphism with obesity and type 2 diabetes. Hum Mol Genet 2001; 10: 1753–60
Nieters A, Becker N, Linseisen J. Polymorphisms in candidate obesity genes and their interaction with dietary intake of n-6 polyunsaturated fatty acids affect obesity risk in a sub-sample of the EPIC-Heidelberg cohort. Eur J Nutr 2002; 41: 210–21
Leviev I, Kalix B, Brulhart Meynet MC, et al. The paraoxonase PON1 promoter polymorphism C(−107)T is associated with increased serum glucose concentrations in non-diabetic patients. Diabetologia 2001; 44: 1177–83
Fernandez-Real JM, Ricart W. Insulin resistance and inflammation in an evolutionary perspective: the contribution of cytokine genotype/phenotype to thriftiness. Diabetologia 1999; 42: 1367–74
Fernandez-Real JM, Ricart W. Insulin resistance and chronic cardiovascular inflammatory syndrome. Endocr Rev 2003; 24: 278–301
Witchel SF, Lee PA, Suda-Hartman M, et al. Evidence for a heterozygote advantage in congenital adrenal hyperplasia due to 21-hydroxylase deficiency. J Clin Endocrinol Metab 1997; 82: 2097–101
Hotamisligil GS. The role of TNFalpha and TNF receptors in obesity and insulin resistance. J Intern Med 1999; 245: 621–5
Hotamisligil GS, Peraldi P, Budavari A, et al. IRS-1 mediated inhibition of insulin receptor tyrosine kinase activity in TNF-α and obesity-induced insulin resistance. Science 1996; 271: 665–8
Naz RK, Thurston D, Santoro N. Circulating tumor necrosis factor (TNF)-alpha in normally cycling women and patients with premature ovarian failure and polycystic ovaries. Am J Reprod Immunol 1995; 34: 170–5
Fernandez-Real JM, Broch M, Ricart W, et al. Plasma levels of the soluble fraction of tumor necrosis factor receptor 2 and insulin resistance. Diabetes 1998; 47: 1757–62
Fernandez-Real JM, Gutierrez C, Ricart W, et al. Plasma levels of the soluble fraction of tumor necrosis factor receptors 1 and 2 are independent determinants of plasma cholesterol and LDL cholesterol concentrations in healthy subjects. Atherosclerosis 1999; 146: 321–7
Ibañlez L, Potau N, Zamboni M, et al. Hyperinsulinemia in postpubertal girls with a history of premature pubarche and functional ovarian hyperandrogenism. J Clin Endocrinol Metab 1996; 81: 1237–43
Ibañez L, Valls C, Potau N, et al. Sensitization to insulin in adolescent girls to normalize hirsutism, hyperandrogenism, oligomenorrhea, dyslipidemia, and hyperinsulinism after precocious pubarche. J Clin Endocrinol Metab 2000; 85: 3526–30
Tuomilehto J, Lindstrom J, Eriksson JG, et al. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N Engl J Med 2001; 344: 1343–50
Chang RJ, Nakamura RM, Judd HL, et al. Insulin resistance in nonobese patients with polycystic ovarian disease. J Clin Endocrinol Metab 1983; 57: 356–9
Velázquez EM, Mendoza S, Hamer T, et al. Metformin therapy in polycystic ovary syndrome reduced hyperinsulinemia, insulin resistance, hyperandrogenemia, and systolic blood pressure, while facilitating normal menses and pregnancy. Metabolism 1994; 43: 647–54
Crave FJ, Fimbel S, Lejeune H, et al. Effects of diet and metformin administration on sex hormone-binding globulin, androgens, and insulin in hirsute obese women. J Clin Endocrinol Metab 1995; 80: 2057–62
Dunaif A, Scott D, Finegood D, et al. The insulin-sensitizing agent troglitazone improves metabolic and reproductive abnormalities in the polycystic ovary syndrome. J Clin Endocrinol Metab 1996; 81: 3299–306
Nestler JE, Jakubowicz DJ, Reamer P, et al. Ovulatory and metabolic effects of D-chiro-inositol in the polycystic ovary syndrome. N Engl J Med 1999; 340: 1314–20
Harborne L, Fleming R, Lyall H, et al. Descriptive review of the evidence for the use of metformin in polycystic ovary syndrome. Lancet 2003; 361: 1894–901
Stumvol M, Haring H. The peroxisome proliferator-activated receptor-gamma2 Pro12Ala polymorphism. Diabetes 2002; 51: 2341–7
Seely BL, Staubs PA, Reichart DR, et al. Protein tyrosine phosphatase 1B interacts with the activated insulin receptor. Diabetes 1996; 45: 1379–85
Goldstein BJ, Bittner-Kowalcyzk A, White MF, et al. Tyrosine dephosphorylation and deactivation of insulin receptor substrate-1 by protein-tyrosine phosphatase 1B: possible facilitation by the formation of a ternary complex with the Grb2 adaptor protein. J Biol Chem 2000; 275: 4283–9
Hundal RS, Petersen KF, Mayerson AB, et al. Mechanism by which high-dose aspirin improves glucose metabolism in type 2 diabetes. J Clin Invest 2002; 109: 1321–6
Jiang G, Dallas-Yang Q, Liu F, et al. Salicylic acid reverses phorbol 12-myristate-13-acetate (PMA)- and tumor necrosis factor alpha (TNFalpha)-induced insulin receptor substrate 1 (IRS1) serine 307 phosphorylation and insulin resistance in human embryonic kidney 293 (HEK293) cells. J Biol Chem 2003; 278: 180–6
Gao Z, Zuberi A, Quon MJ, et al. Aspirin inhibits serine phosphorylation of insulin receptor substrate 1 in tumor necrosis factor-treated cells through targeting multiple serine kinases. J Biol Chem 2003; 278: 24944–50
Acknowledg ments
Belén Roldán Martín is a postdoctoral fellow of the Fundación Ramón Areces, Madrid. This work was supported in part by Grants FIS 02/0741 and RGDM G03/212 from the Fondo de Investigación Sanitaria, Ministerio de Sanidad y Consumo, and Grant CAM 08.6/0010/2001 from the Consejería de Educación, Comunidad de Madrid, Spain. ## The authors have no conflicts of interest directly relevant to the content of this review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Roldán, B., Millán, J.L.S. & Escobar-Morreale, H.F. Genetic Basis of Metabolic Abnormalities in Polycystic Ovary Syndrome. Am J Pharmacogenomics 4, 93–107 (2004). https://doi.org/10.2165/00129785-200404020-00004
Published:
Issue Date:
DOI: https://doi.org/10.2165/00129785-200404020-00004