Abstract
Abstract
Eplerenone is a selective aldosterone blocker (SAB) approved for the treatment of essential hypertension. Oral eplerenone reduced BP effectively in patients with essential hypertension, both as monotherapy and in combination with other agents. The drug is generally well tolerated; the risk of hyperkalemia can be managed through careful selection and monitoring of patients. Preliminary data suggest that treatment of hypertension with eplerenone may provide protective effects against end-organ disease; further work is needed to elucidate the clinical significance of these findings, and to evaluate the outcome of treatment of hypertension in terms of cardiovascular morbidity and mortality and quality of life. In the meantime, as the first SAB to become available, eplerenone is an interesting addition to the drugs currently available for the treatment of hypertension.
Pharmacologic Properties
Eplerenone is an SAB with up to 1000-fold higher binding affinity for the mineralocorticoid receptor than for other steroid receptors, such as those for progesterone and androgens. Eplerenone competitively inhibits aldosterone binding to mineralocorticoid receptors and blocks its effects in both epithelial and nonepithelial tissues, including the kidney, heart, and vasculature. In animal models, in addition to causing natriuresis, eplerenone reduced vascular and myocardial fibrosis, attenuated the effects of vasoactive mediators on the vascular endothelium, and reduced cardiac hypertrophy.
After oral administration, eplerenone is rapidly absorbed with a mean peak plasma concentration (Cmax) of 1.72 μg/mL achieved approximately 1.2 hours after administration of a single 100mg dose; the mean area under the plasma concentration-time curve (AUC) was 9.54 μg · h/mL. Steady-state concentrations are reached within 2 days. Eplerenone is approximately 33–66% protein bound, and the apparent volume of distribution at steady state was 43–90L. Eplerenone is extensively metabolized, predominantly by cytochrome P450 (CYP) 3A4; no active metabolites have been identified. The apparent plasma clearance of eplerenone was approximately 10 L/h and the elimination half-life was 3–6 hours. Following a single oral dose of eplerenone, 67% was eliminated in the urine and 32% in the feces.
Although Cmax and AUC increased in patients with severe renal impairment, patients with moderate hepatic impairment, and elderly volunteers, the extent of the increased exposure does not necessitate dose adjustments in these patients. Nonetheless, eplerenone is not recommmended for use in patients with severe renal impairment (see Tolerability Summary). Cmax and AUC were lower in Black patients than White patients. Coadministered inhibitors of CYP3A4 increased blood levels of eplerenone; strong inhibitors (e.g. ketoconazole) increased systemic exposure to eplerenone 5-fold and concomitant administration is contraindicated.
Therapeutic Efficacy
Eplerenone 25–400mg daily reduced BP significantly compared with placebo in patients with hypertension, with a clear dose response up to 100mg daily, and no effect on heart rate. At a dosage of 100mg daily, DBP was reduced by approximately 4–8mm Hg and SBP by 8–12mm Hg. Based on preliminary data, eplerenone 50–200mg once daily demonstrated similar antihypertensive activity to enalapril 10–40mg once daily in patients with mild-to-moderate hypertension and amlodipine 2.5–10mg once daily in patients with systolic hypertension. Eplerenone 50–200mg once daily showed at least as good efficacy as losartan 50–100mg once daily in patients with low-renin hypertension and greater efficacy in Black patients.
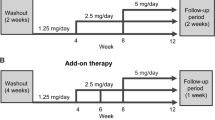
Eplerenone add-on therapy improved control of SBP and DBP with angiotensin II receptor antagonists (and, based on preliminary data, β-adrenoceptor antagonists), and control of SBP with ACE inhibitors (and calcium channel antagonists, based on preliminary data), with tolerability similar to monotherapy. In placebo-controlled trials, BP control was maintained for 24 hours with once daily dosages, although in one study 50mg twice daily reduced mean trough BP more than 100mg once daily.
Preliminary data also indicate that eplerenone decreased surrogate markers of end-organ protection in patients with hypertension, including microalbuminuria (at dosages of 50–200mg once daily) and left ventricular hypertrophy (200mg once daily). The combination of eplerenone with enalapril was more effective than eplerenone alone.
Tolerability
Based on the available data, eplerenone was generally well tolerated in patients with hypertension, with an overall incidence of adverse events similar to placebo; the most common adverse events in the hypertension studies were dizziness, fatigue, influenza-like symptoms, cough, and diarrhea, all with a low incidence (≤3%). The incidence of antiandrogenic and progestogenic adverse effects with eplerenone was similar to that for placebo. The incidence of hyperkalemia increased at a dosage of 400mg daily compared with lower dosages; the risk of hyperkalemia also increased in patients with renal insufficiency.












Similar content being viewed by others
Notes
The use of trade names is for product identification purposes only and does not imply endorsement.
References
Alderman M, Arakawa K, Beilin L, et al. 1999 World Health Organization —International Society of Hypertension Guidelines for the Management of Hypertension. Blood Press 1999; (8 Suppl. 1): 9–43
Burl VL, Whelton P, Roccella EJ, et al. Prevalence of hypertension in the US adult population: results from the Third National Health and Nutrition Survey, 1988–1991. Hypertension 1995; 25: 305–13
Chobanian AV, Bakris GL, Black HR, et al. The Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure: the JNC 7 Report. JAMA 2003 May 21; 289(19): 2560–72
American Heart Association. Heart disease and stroke statistics: 2004 update [online]. Available from URL: http://www.americanheart.org [Accessed 2004 Oct 21]
Cha AJ, Malecha SE, Judge KW. Aldosterone: a new appreciation of its role in heart failure. Pharmacotherapy 2000; 20(9): 1107–15
Rocha R, Funder JW. The pathophysiology of aldosterone in the cardiovascular system. Ann N Y Acad Sci 2002 Sep; 970: 89–100
Struthers AD. Aldosterone: cardiovascular assault. Am Heart J 2002 Nov; 144 (5 Suppl.): S2–7
Givertz MM. Manipulation of the renin-angiotensin system. Circulation 2001; 104: e14–18
Epstein M. Aldosterone as a mediator of progressive renal disease: pathogenetic and clinical implications. Am J Kidney Dis 2001 Apr; 37(4): 677–88
Pitt B, Zannad F, Remme WJ, et al. The effect of Spironolactone on morbidity and mortality in patients with severe heart failure. N Engl J Med 1999; 341(10): 709–17
McKelvie RS, Yusuf S, Pericak D, et al. Comparison of candesartan, enalapril and their combination in congestive heart failure: Randomized Evaluation of Strategies for Left Ventricular Dysfunction (RESOLVD) pilot study. RESOLVD Pilot Study Investigators. Circulation 1999; 100: 1056–64
de Gasparo M, Whitebread SE, Preiswerk G, et al. Antialdosterones: incidence and prevention of sexual side effects. J Steroid Biochem 1989 Jan; 32(1B): 223–7
Jeunemaitre X, Chatellier G, Kreft-Jais C, et al. Efficacy and tolerance of Spironolactone in essential hypertension. Am J Cardiol 1987; 60: 820–5
G.D. Searle LLC. Inspra™ (eplerenone) tablets. Prescribing information (US) [online]. Available from URL: http://www.inspra.com [Accessed 2004 Feb 25]
Quaschning T, Ruschitzka F, Shaw S, et al. Aldosterone receptor antagonism normalizes vascular function in liquorice-induced hypertension. Hypertension 2001 Feb; 37 (2 Pt 2): 801–5
Rocba R, Stier Jr CT, Kifor I, et al. Aldosterone: a mediator of myocardial necrosis and renal arteriopathy. Endocrinology 2000 Oct; 141(10): 3871–8
Delyani JA, Robinson EL, Rudolph AE, Effect of a selective aldosterone receptor antagonist in myocardial infarction. Am J Physiol Heart Circ Physiol 2001; 50: H647–54
de Gasparo M, Joss U, Ramjoué HP, et al. Three new epoxy-spironolactoue derivatives: characterization in vivo and in vitro. J Pharmacol Exp Ther 1987; 240(2): 650–6
Weinberger MH, Roniker B, Krause SL, et al. Eplerenone, a selective aldosterone blocker, in mild-to-moderate hypertension. Am J Hypertens 2002 Aug; 15(8): 709–16
Krum H, Nolly H, Workman D, et al. Efficacy of eplerenone added to reninangiotensin blockade in hypertensive patients. Hypertension 2002 Aug; 40(2): 117–23
Weinberger M, MacDonald T, Conlin PR, et al. Comparison of eplerenone and losartan in patients with low-renin hypertension [abstract no. OR-55]. Am J Hypertens 2002 Apr; 15 (4 Pt 2): 24A
Van Mieghem W, von Behren V, Balazovjech I, et al. Eplerenone is safe and effective as add-on therapy in hypertensive patients uncontrolled with calcium channel blockers or beta blockers [abstract no. P1163]. Eur Heart J 2002; (23 Suppl.): 211
Roniker B. Eplerenone: a selective antagonist of the aldosterone receptor [abstract no. 67]. Hypertension 1997 Oct; 30(4): 995
Delyani JA, Rocha R, Cook CS, et al. Eplerenoue: a selective aldosterone receptor antagonist (SARA). Cardiovasc Drag Rev 2001 Fall; 19(3): 185–200
Frierdich G, Schuh J, Brown M, et al. Effects of the selective mineralocorticoid receptor antagonist, eplerenone, in a model of aldosterone-induced hypertension and cardiac fibrosis [abstract no. B004]. Am J Hypertens 1998 Apr; 11 (4 Pt 2): 94A
Rocha R, Frierdich GE, Nachowiak D, et al. Aldosterone induces a vascular inflammatory phenotype in the rat heart [abstract no. 40]. Hypertension 2001 Sep; 38(3): 479
Rajagopalan S, Duquaine D, King S, et al. Mineralocorticoid receptor antagonism in experimental atherosclerosis. Circulation 2002 May 7; 105(18): 2212–6
Hurlimann D, Lu:¢her TF, Shaw SG, et al. Eplerenone normalizes vascular function in liquorice-induced hypertension [abstract no. 3337]. Eur Heart J 2001 Sep; (22 Suppl.): 612
White WB, Duprez D, St Hillaire R, et al. Effects of the selective aklosterone blocker eplerenone versus the calcium antagonist amlodipine in systolic hypertension. Hypertension 2003 May; 41: 1021–6
Pitt B, Reichek N, Willenbrock R, et al. Effects of eplerenone, enalapril, and eplerenone/enalapril in patients with essential hypertension and left ventricular hypertrophy: the 4E-Left Ventricular Hypertrophy Study. Circulation 2003 Oct 14; 108: 1831–8
Williams GH, Burgess E, Kolloch RE, et al. Efficacy of eplerenone versus enalapril as monotherapy in systemic hypertension. Am J Cardiol 2004 Apr 15; 93(8): 990–6
Epstein M, Buckalew Jr V, Martinez F, et al. Antiproteinuric efficacy of eplerenone, enalapril, and eplerenone/enalapril combination therapy in diabetic hypertensives with microalbuminuria [abstract no. OR-54]. Am J Hypertens 2002 Apr; 15 (4 Pt 2): 24A
Flack JM, Oparil S, Pratt JH, et al. Efficacy and tolerability of eplerenone and losartan in hypertensive black and white patients. J Am Coll Cardiol 2003 Apr 2; 41(7): 1148–55
Delyani J, Myles K, Funder J. Eplerenone (SC 66110), a highly selective aldosterone antagonist [abstract no. B002]. Am J Hypertension 1998 Apr; 11 (4 Pt 2): 94A
Coleman CI, Reddy P, Song JC, et al. Eplerenone: the first selective aldosterone receptor antagonist for the treatment of hypertension. Formulary 2002 Oct; 37: 514–24
Blacher J, Amah G, Girerd X, et al. Association between increased plasma levels of aldosterone and decreased systemic arterial compliance in subjects with essential hypertension. Am J Hypertension 1997; 10: 1326–34
Rajagopalan S, Pitt B. Aldosterone antagonists in the treatment of hypertension and target organ damage. Curr Hypertens Rep 2001 Jun; 3(3): 240–8
Barton M, d’Uscio LV, Shaw S, et al. ETA receptor blockade prevents increased tissue endothelin-1, vascular hypertrophy, and endothelial dysfunction in salt-sensitive hypertension. Hypertension 1998; 31 (Pt 2): 499–504
Schiffrin EL, Deng LY, Sventek P, et al. Enhanced expression of endothelin-1 gene in resistance arteries in severe human essential hypertension. J Hypertension 1997; 15(1): 57–63
van Zwieten PA. Drug treatment of isolated systolic hypertension. Nephrol Dial Transplant 2001; 16(6): 1095–7
Rocha R, Chander PN, Zuckerman A, et al. Role of mineralocorticoids in renal injury in stroke-prone hypertensive rats [abstract no. 49]. Hypertension 1998 Sep; 32(3): 598
Cook CS, Zhang L, Fischer JS. Absorption and disposition of a selective aldosterone receptor antagonist, eplerenone, in the dog. Pharm Res 2000 Nov; 17(11): 1426–31
Cook CS, Berry LM, Bible RH, et al. Pharmacokinetics and metabolism of [14C]eplerenone after oral administration to humans. Drug Metab Dispos 2003 Nov; 31(11): 1448–55
Tolbert DS, Reid SE, Roniker B. Safety, tolerability, and pharmacokinetics of eplerenone, a selective aldosterone blocker [abstract no. 45]. Pharmacotherapy 2002 Oct; 22: 1331–2
Tolbert DS, Reid SE, Roniker B. Pharmacokinetics of eplerenone in special populations [abstract no. 46]. Pharmacotherapy 2002 Oct; 22(10): 1332
Final report for evaluation of the single- and multiple-dose pharmacokinetics of eplerenone in subjects with and without renal impairment. Report no. NE3-01-06-034. US: G.D. Searle and Co., 2001 Oct 19. (Data on file)
Tolbert DS, Reid SE, Roniker B. Pharmacokinetics of eplerenone coadministered with other medications [abstract no. 44]. Pharmacotherapy 2002 Oct; 22(10): 1331
Tolbert DS, Reid SE, Qian J, et al. Effects of erythromycin on the pharmacokinetics of eplereuone [abstract no. T2264]. AAPS PhannSci 2002; 4 (4) [online]. Available from URL: www.aapspharmaceutica.com [Accessed 2004 Nov 30]
Tolbert DS, Reid SE, Qian J, et al. Effects of St. John’s Wort on the pharmacokinetics of eplerenone [abstract no. T2267]. AAPS PharmSci 2002; 4 (4) [online]. Available from URL: www.aapspharmaceutica.com [Accessed 2004 Nov 30]
Tolbert DS, Reid SE, Qian J, et al. Effects of eplerenone on the pharmacokinetics of cisapride [abstract no. T2266]. AAPS PharmSci 2002; 4 (4) [online]. Available from URL: www.aapspharmaceutica.com [Accessed 2004 Nov 30]
White WB, Carr AA, Krause S, et al. Assessment of the novel selective aldosterone blocker eplerenone using ambulatory and clinical blood pressure in patients with systemic hypertension. Am J Cardiol 2003 Jul 1; 92(1): 38–42
Saruta T, Kageyama S, Ogihara T, et al. Efficacy and safety of the selective aldosterone blocker eplerenone in Japanese patients with hypertension: a randomized, double-blind, placebo-controlled, dose-ranging study. J Clin Hypertens (Greenwich) 2004 Apr; 6(4): 175–85
Burgess ED, Lacourciere Y, Ruilope-Urioste LM, et al. Long-term safety and efficacy of the selective aldosterone blocker eplerenone in patients with essential hypertension. Clin Ther 2003 Sep; 25(9): 2388–404
Williams ES, Miller JM. Results from late-breaking clinical trial sessions at the American College of Cardiology 51st Annual Scientific Session. J Am Coll Cardiol 2002 Jul 3; 40(1): 1–18
Final report for the evaluation of eplerenone versus losartan and placebo in black and white patients. Report no.: IE3-01-06-020. US: G.D. Searle and Co., 2001 Sep 19. (Data on file)
Guidelines Committee. 2003 European Society of Hyperteusion-European Society of Cardiology guidelines for the management of arterial hypertension. J Hypertens 2003; 21(6): 1011–53
Williams B, Poulter NR, Brown MJ, et al. Guidelines for management of hypertension: report of the fourth working party of the British Hypertension Society, 2004 — BHS IV. British Hypertension Society. J Hum Flypertens 2004; 18: 139–85
UK Prospective Diabetes Study Group. Tight blood pressure control and risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 38. Br Med J 1998 Sep 12; 317: 703–13
Díez J, Gonzàlez A, Lopez B, et al. Effects of antihypertensive agents on the left ventricle: clinical implications. Am J Cardiovasc Drugs 2001; 1(4): 263–79
Hollenberg NK, Williams GH, Anderson R, et al. Symptoms and the distress they cause: comparison of an aldosterone antagonist and a calcium channel blocking agent in patients with systolic hypertension. Arch Intern Med 2003 Jul 14; 163(13): 1543–8
Pitt B, Remme W, Zannad F, et al. Eplerenone, a selective aldosterone blocker, in patients with left ventricular dysfunction after myocardial infarction. New Engl J Med 2003 Apr 3; 348(14): 1309–21
Sica DA. Eplerenone: a new aldosterone receptor antagonist — are the FDA’s restrictions appropriate? J Clin Hypertens 2002; 4(6): 441–5
MacMahon S, Rodgers A. The effects of antihypertensive treatment on vascular disease: reappraisal of the evidence in 1994. J Vasc Med Biol 1993; 4(5–6): 265–71
Psaty BM, Smith NL, Siscovick DS, et al. Health outcomes associated with antihypertensive therapies used as first-line agents: a systematic review and meta-analysis. JAMA 1997 Mar 5; 277(9): 739–45
Moser M, Hebert PR. Prevention of disease progression, left ventricular hypertrophy and congestive heart failure in hypertension treatment trials. J Am Coll Cardiol 1996; 27: 1214–8
Levy D, Larson MG, Vasan RS, et al. The progression from hypertension to congestive heart failure. JAMA 1996 May 22; 275(20): 1557–62
Blood pressure, cholesterol, and stroke in eastern Asia: Eastern Stroke and Coronary Heart Disease Collaborative Research Group. Lancet 1998 Dec 5; 352: 1801–7
Klag MJ, Whelton PK, Randall BL, et al. Blood pressure and end-stage renal disease in men. N Engl J Med 1996; 334(1): 13–8
Collins R, MacMahon S. Blood pressure, antihypertensive drug treatment and the risks of stroke and of coronary heart disease. Br Med Bull 1994 Apr; 50: 272–98
Whitworth JA. 2003 World Health Organization (WHO)/Intemational Society of Hypertension (ISH) statement on management of hypertension: World Health Organization International Society of Hypertension Writing Group. J Hypertens 2003; 21: 1983–92
Aruaz-Pacheco C, Parrott MA, Raskin P. Treatment of hypertension in adults with diabetes: American Diabetes Association. Diabetes Care 2003 Jan; (26 Suppl. 1): S80–2
Olivieri O, Ciacciarelli A, Signorelli D, et al. Aldosterone to renin ratio in a primary care setting: the Bussolengo Study. J Clin Endocrinol Metab 2004; 89(9): 4221–6
Epstein M. Aldosterone as a determinant of cardiovascular and renal dysfunction. J R Soc Med 2001 Aug; 94(8): 378–83
Rocha R, Williams GH. Rationale for the use of aldosterone antagonists in congestive heart failure. Drugs 2002; 62(5): 723–31
Delyani JA. Mineralocorticoid receptor antagonists: the evolution of utility and pharmacology [genetics, pathophysiology, and pharmacological aspects]. Kidney Int 2000 Apr; 57(4): 1408–11
Dahloöf B, Penneit K, Hansson L. Reversal of left ventricular hypertrophy in hypertensive patients: a metaanalysis of 109 treatment studies. Am J Hypertens 1992; 5: 95–110
Brown NJ, Vaughan DE. Angiotensin-converting enzyme inhibitors. Circulation 1998; 97: 1411–20
Toto R, Shultz P, Raij L, et al. Efficacy and tolerability of losartan in hypertensive patients with renal impairment. Hypertension 1998; 31: 684–91
Asmar R. Effect of antihypertensive agents on arterial stiffness as evaluated by pulse wave velocity: clinical implications. Am J Cardiovasc Drugs 2001; 1(5): 387–97
Schiffrin EL. Vascular remodeling and endothelial function in hypertensive patients: effects of antihypertensive therapy. Scand Cardiovasc J 1998; (Suppl. 47): 15-21
Van Bortel LMAB, Kool MJF, Spek JJ. Disparate effects of antihypertensive drugs on large artery distensibility and compliance in hypertension. Am J Cardiol 1995; 76: 46E–9E
Cushman WC, Ford CE, Cutler JA, et al. Success and predictors of blood pressure control in diverse North American settings: the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT). ALLHAT Collaborative Research Group. J Clin Hypertens 2002; 4(6): 393–404
Ram CVS. Antihypertensive drugs: an overview. Am J Cardiovasc Drugs 2002; 2(2): 77–89
Pitt OL, Young WE, MacDonald TM. A review of the medical treatment of primary aldosteronism. J Hypertension 2001; 19(3): 353–61
Calhoun DA, Nishizaka MK, Zaman MA, et al. Hyperaldosteronism among black and white subjects with resistant hypertension. Hypertension 2002; 40: 892–6
Hansson L, Zanchetti A, Carruthers SG, et al. Effects of intensive blood-pressure lowering and low-dose aspirin in patients with hypertension: principal results of the Hypertension Optimal Treatment (HOT) randomised trial. HOT Study-Group. Lancet 1998 Jun 13; 351: 1755–62
Author information
Authors and Affiliations
Corresponding author
Additional information
Various sections of the manuscript reviewed by:
M. Burnier, Polyclinique Medicale Universitaire, Lausanne, Switzerland; A.H. Gradman, Division of Cardiovascular Disease, Western Pennsylvania Hospital, Pittsburgh, Pennsylvania, USA; Y. Higashi, Department of Cardiovascular Physiology and Medicine, Hiroshima University, Hiroshima, Japan; N.M. Kaplan, Southwestern Medical Center at Dallas, University of Texas, Dallas, Texas, USA; H. Krum, Department of Epidemiology and Preventive Medicine, Monash University, Melbourne, Victoria, Australia; B. Pitt, Division of Cardiology, University of Michigan, Ann Arbor, Michigan, USA; J.H. Pratt, Department of Medicine, Indiana University School of Medicine, Indianapolis, Indiana, USA; C.V.S. Ram, Texas Blood Pressure Institute, University of Texas Southwestern Medical School, Dallas, Texas, USA; G.L. Schwartz, Department of Internal Medicine, Mayo Clinic, Rochester, Minnesota, USA.
Data Selection
Sources: Medical literature published in any language since 1980 on eplerenone, identified using Medline and EMBASE, supplemented by AdisBase (a proprietary database of Adis International). Additional references were identified from the reference lists of published articles. Bibliographical information, including contributory unpublished data, was also requested from the company developing the drug.
Search strategy: Medline search terms were ‘eplerenone’ or ‘CGP-30083’ or ‘epoxymexrenone’. EMBASE search terms were ‘eplerenone’ or ‘CGP-30083’ or ‘epoxymexrenone’ or ‘SC-66110’. AdisBase search terms were ‘eplerenone’ or ‘SC 66110’. Searches were last updated 3 November 2004.
Selection: Studies in patients with hypertension who received eplerenone. Inclusion of studies was based mainly on the methods section of the trials. When available, large, well controlled trials with appropriate statistical methodology were preferred. Relevant pharmacodynamic and pharmacokinetic data are also included.
Index terms: Eplerenone, selective aldosterone receptor antagonist, selective aldosterone blocker, hypertension, pharmacodynamics, pharmacokinetics, therapeutic use.
Rights and permissions
About this article
Cite this article
Croom, K.F., Perry, C.M. Eplerenone. Am J Cardiovasc Drugs 5, 51–69 (2005). https://doi.org/10.2165/00129784-200505010-00007
Published:
Issue Date:
DOI: https://doi.org/10.2165/00129784-200505010-00007