Abstract
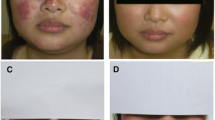
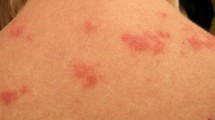
For nearly 50 years, thalidomide has struggled between success and controversy. After causing an epidemic of phocomelia and other birth defects during the 1960s, affecting thousands of neonates, thalidomide was used as a sedative in selective disorders including leprosy. The potent anti-inflammatory properties of thalidomide were serendipitously discovered while treating patients with erythema nodosum leprosum, and the drug is now approved by the US FDA for the treatment of this disease. Subsequently, the immunosuppressant effects of thalidomide, including the complex modulation of many cytokines, have been recognized. One promising application of thalidomide has been the treatment of cutaneous lupus erythematosus. Among the largest series reviewed, the drug has been found to ameliorate cutaneous lupus erythematosus in 90% of patients, on average. Remission is achieved in approximately 15–20% of patients with cutaneous lupus erythematosus at doses between 50–400mg daily. Contraceptive concerns and the recognized neuropathic effects of thalidomide limit the use of the drug in patients with cutaneous lupus. Physicians who prescribe thalidomide in the US must be registered with the drug manufacturer. With appropriate control of drug access and close physician monitoring, thalidomide provides a needed therapeutic option for the treatment of refractory cases of cutaneous lupus erythematosus.





Similar content being viewed by others
Notes
Use of tradenames is for product identification only and does not imply endorsement.
References
Mellin GW, Katzenstein M. The saga of thalidomide: neuropathy to embryopathy, with case reports and congenital abnormalities. N Engl J Med 1962; 23: 1184–1190
Burley DM, Dennison TC, Harrison W. Clinical experience with a new sedative drug. Practitioner 1959; 183: 57–61
Distaval. Lancet 1961; II: 1262
Mellin GW. The saga of thalidomide (concluded). N Engl J Med 1962; 267: 1238–1244
Wiedemann HR. Hinweis auf eine derzeitige Haufung hypound aplastischer fehlbildungen der gliedmassen. Med Welt 1961; 37: 1863–1866
Pfeiffer RA, Kosenow W. Thalidomide and congenital abnormalities [letter]. Lancet 1962; I: 45
Lenz W. Kindliche Missbildungen nach Medikament-Einnahme wahrend der Graviditat? [letter]. Deutsche Med Wchnschr 1961; 86: 2555
Lenz W. Thalidomide and congenital abnormalities [letter]. Lancet 1962; I: 45
Sheskin J. Thalidomide in the treatment of lepra reactions. Clin Pharm Ther 1965; 6: 303–306
Tseng S, Pak G, Washenik K, et al. Rediscovering thalidomide: a review of its mechanism of action, side effects and potential uses. J Am Acad Dermatol 1996; 35: 969–979
Sheskin J. The treatment of lepra reaction in lepromatous leprosy: fifteen years’ experience with thalidomide. Int J Dermatol 1980; 19: 318–322
Londono F. Thalidomide in the treatment of actinic prurigo. Int J Dermatol 1973; 12: 326–328
Mattos O. Prurigo nodular de hyde tratado com talidomida [letter]. Bol Div Nac Lepra 1973; 32: 71
Torras H, Mascaro JM, Lecha M. Thalidomide treatment of recurrent necrotic giant mucocutaneous aphthae and aphthosis. Arch Dermatol 1982; 118 (11): 875
Barba-Rubio J, Franco-Gonzalez F. Lupus eritematoso discoide y talidomida. Dermatol Rev Mex 1975; 19: 131–139
Grosshans E, Illy G. Thalidomide therapy for inflammatory dermatoses. Int J Dermatol 1984; 23: 598–602
Barnhill RL, McDougall AC. Thalidomide: use and possible mode of action in reactional lepromatous leprosy and in various other conditions. J Am Acad Dermatol 1982; 7: 317–323
Roath S, Elves MW, Israel MGC. Effect of thalidomide on human leukocyte cultures [letter]. Lancet 1962; II: 812
Roath S, Elves MW, Israel MCG. Effects of thalidomide on human leukocytes cultured in vitro. Lancet 1963; I: 249–250
Field EO, Gibbs JE, Tucker DF, et al. Effect of thalidomide on the graft versus host reaction. Nature 1966; 211: 1308–1310
Vogelsang GB, Hess AD, Santos GW. Thalidomide for treatment of graft-versus-host disease: a review. Bone Marrow Transplant 1988; 3: 393–398
Murphy GP, Brede RD, Weber HW, et al. Thalidomide immunosuppression in canine renal allotransplantation. J Med 1970; 1: 66–73
Faure M, Thivolet J, Gaucherand M. Inhibition of PMN leukocytes chemotaxis by thalidomide. Arch Dermatol Res 1980; 269: 275–280
Crain E, McIntosh KR, Gordon G, et al. The effect of thalidomide on experimental autoimmune myasthenia gravis. J Autoimmunity 1989; 2: 197–202
Moncada B, Baranda ML, Gonzalez-Amaro R, et al. Thalidomide effect on T cell subsets as a possible mechanism of action. Int J Lepr Other Mycobact Dis 1985; 53: 201–205
Sarno EN, Grau GE, Vieira LMM, et al. Serum levels of TNF-alpha and IL-1 beta during leprosy reactional states. Clin Exp Immunol 1991; 84: 103–108
Sampaio EP, Sarno EN, Galilly R, et al. Thalidomide selectively inhibits tumor necrosis factor-alpha production by stimulated human monocytes. J Exp Med 1991; 173: 699–703
Moreira AL, Sampaio EP, Zmuidzinas A, et al. Thalidomide exerts its inhibitory action on tumor necrosis factor alpha by enhancing mRNA degradation. J Exp Med 1993; 177: 1675–1680
McHugh SM, Rifkin IR, Deighton J, et al. The immunosuppressive drug thalidomide induces T helper cell type 2 (Th2) and concomitantly inhibits Thl production in mitogen- and antigen-stimulated human peripheral blood mononuclear cell cultures. Clin Exp Immunol 1995; 99: 160–167
Vernon A, Juffermans NP, Speelman P, et al. A single oral dose of thalidomide enhances the capacity of lymphocytes to secrete gamma interferon in healthy humans. Antimicrob Agents Chemother 2000; 44: 2286–2290
Moller DR, Wysocka M, Greenlee BM, et al. Inhibition of IL-12 production by thalidomide. J Immunol 1997; 159: 5157–5161
Corral LG, Haslett P A, Muner GW, et al. Differential cytokine modulation and T-cell activation by two distinct classes of thalidomide analogues that are potent inhibitors of TNF-alpha. J Immunol 1999; 163: 380–386
Davies FE, Raje N, Hideshima T, et al. Thalidomide and immunomodulatory derivatives aug:trent natural killer cell cytotoxicity I multiple myeloma. Blood 2001; 98: 210–216
Walchner M, Meurer M, Plewig G, et al. Clinical and immunologic parameters during thalidomide treatment of lupus erythematosus. Int J Dermatol 2000; 39: 383–388
D’Amato RJ, Loughnan MS, Flynn E, et al. Thalidomide is an inhibitor of angiogenesis. Proc Natl Acad Sci USA 1994; 91: 4082–4085
Duong DJ, Spigel GT, Moxley RT, et al. American experience with low-dose thalidomide therapy for severe cutaneous lupus erythematosus. Arch Dermatol 1999; 135: 1079–1087
Ordi-Ros J, Cortes F, Cucurull E, et al. Thalidomide in the treatment of cutaneous lupus refractory to conventional therapy. J Rheumatol 2000; 27: 1429–1433
Kyriakis KP, Kontochristopoulos GJ, Panteleos DN. Experience with low-dose thalidomide in chronic discoid lupus erythematosus. Int J Dermatol 2000; 39: 218–222
Naafs B, Faber WR. Thalidomide therapy: an open trial. Int J Dermatol 1985; 24: 131–134
Hasper MF. Chronic cutaneous lupus erythematosus: thalidomide treatment of 11 patients. Arch Dermatol 1983; 119: 812–815
Knop J, Bonsmann G, Happle R, et al. Thalidomide in the treatment of sixty cases of chronic DLE. Br J Dermatol 1983; 108: 461–466
Stevens RJ, Andujar C, Edwards CJ, et al. Thalidomide in the treatment of the cutaneous manifestations oflupus erythematosus: experience in sixteen consecutive patients. Br J Rheumatol 1997; 36: 353–359
Atra E, Sato EI. Treatment of the cutaneous lesions of systemic lupus erythematosus with thalidomide. Clin Exp Rheumatol 1993; 11: 487–493
Burrows NP, Walport MJ, Hammond AH, et al. Lupus erythematosus profundus with partial C4 deficiency responding to thalidomide. Br J Dermatol 1991; 125: 62–67
Holm AL, Bowers KE, McMeekin TO, et al. Chronic cutaneous lupus erythematosus treated with thalidomide. Arch Dermatol 1993; 129: 1548–1550
Warren KJ, Nopper AJ, Crosby DL. Thalidomide for recalcitrant discoid lesions in a patient with systemic lupus erythematosus. J Am Acad Dermatol 1998; 39: 293–295
Bessis D, Guillot B, Monpoint S, et al. Thalidomide for systemic lupus erythematosus. Lancet 1992; 339: 549–550
Lenz W, Knapp K. Die thalidomid-embropathie [letter]. Deutsche Med Wchnschr 1962; 87: 11232
Howe P. Neuropathy after thalidomide (“Distaval”) [letter]. BMJ 1961; 2: 1084
Heathfield KWG. Neuropathy after thalidomide (“Distaval”). BMJ 1961; 2: 1084
Wulff CH, Hoyer H, Asboe-Hansen G, et al. Development of polyneuropathy during thalidomide therapy. Br J Dermatol 1985; 112: 475–480
Clemmensen OJ, Olsen PZ, Andersen KE. Thalidomide neurotoxicity. Arch Dermatol 1984; 120: 338–341
Ochonisky S, Verroust J, Bastuji-Garin S, et al. Thalidomide neuropathy incidence and clinico-electrophysiologic findings in 42 patients. Arch Dermatol 1994; 130: 66–69
Chapon F. Lechevalier B, da Silva DC, Rivrain Y, Dupuy B, Deschamps P. Neuropathies caused by thalidomide. Rev Neurol (Paris) 1985; 141: 719–728
Ordi J, Cortes F, Martinez N, et al. Concise communication: thalidomide induces amenorrhea in patients with lupus disease. Arthritis Rheum 1998; 41: 2273–2275
Gompel A, Frances C, Piette JC, et al. Ovarian failure with thalidomide treatment in complex aphthosis: comment on the concise communication by Ordi et al. Arthritis Rheum 1999; 42: 2259–2260
Piette JC, Sbaj A, Frances C. Warning: thalidomide-related thrombotic risk potentially concerns patients with Lupus. Lupus 2002; 11: 67–70
Flageul B, Wallach D, Cavelier-Balloy B, et al. Thalidomide et thromboses. Ann Dermatol Venereol 2000; 127: 171–174
Zangari M, Anaissie E, Barlogie B, et al. Increased risk of deep-vein thrombosis in patients with multiple myeloma receiving thalidomide and chemotherapy. Blood 2001; 98: 1614–1615
Osman K, Comenzo R, Rajkumar SV. Deep venous thrombosis and thalidomide therapy for multiple myeloma. N Engl J Med 2001; 344: 1951–1952
Camba L, Peccatori J, Pescarollo A, et al. Thalidomide and thrombosis in patients with multiple myeloma. Haematologica 2001; 86: 1108–1109
Hamuryudan V, Mat C, Saip S, et al. Thalidomide in the treatment of mucocutaneous lesions of the Behcet’s syndrome: a randomized double blind placebo controlled trial. Ann Intern Med 1998; 128: 443–450
Powell RJ, Garder-Medwin JMM. Guideline for the clinical use and dispensing of thalidomide. Postgrad Med J 1994; 70: 901–904
System for thalidomide education and prescribing safety (STEPS™). Starter Kit and Prescriber Info. Warren (NJ): Celgene Corporation, 2001
Teo SK, Harden JL, Burke AB, et al. Thalidomide is distributed into human semen after oral dosing. Drug Metab Dispos 2001; 29: 1355–1357
Acknowledgements
The authors have no conflicts of interest regarding the preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pelle, M.T., Werth, V.P. Thalidomide in Cutaneous Lupus Erythematosus. Am J Clin Dermatol 4, 379–387 (2003). https://doi.org/10.2165/00128071-200304060-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00128071-200304060-00002