Abstract
Objective: To assess the impact of a comprehensive migraine disease management program, as measured by humanistic outcomes measures, conducted in a managed care setting.
Design: A prospective comparative study comprised of an intervention and a control group to evaluate the impact of the disease management initiative.
Setting: Independent Practice Association (IPA)-type managed care organization.
Study participants and main outcomes measures: Study participants resided in adjacent regions (intervention region included Minneapolis, MN, USA and the usual care region included St Paul, MN, USA and adjacent areas in MN, USA) separated by natural geographic barriers. Eligible patients were identified through a review of the Medica plan’s administrative claims database and were asked to complete the Migraine Therapy Assessment Questionnaire (MTAQ), a tool to assess the presence of migraine management issues. Responders (patients completing the MTAQ) who reported ≥2 migraine care risk indicators in the intervention region received the disease management intervention, while all responders in the control region received usual care. Responders in both regions with ≥2 migraine care risk indicators were also asked to complete the Migraine Disability Assessment (MIDAS) questionnaire and the 12-item Short Form Health Survey (SF-12) at baseline. All participants were asked to complete the same instruments at the end of the program as they did at baseline. Interventions: Disease management intervention activities included patient and physician education, as well as a patient feedback report containing migraine care risk indicators, which was sent to the treating physicians. All patients were encouraged to contact their physicians if they reported two or more migraine management issues.
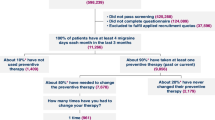
Results: A total of 2232 patients with migraine were enrolled in the study (1373 from the intervention region and 859 in the control region). Compared with patients in the control region, patients in the intervention region reported, as measured by MTAQ, significant improvement in migraine symptom relief, more knowledge about potential migraine triggers, a decrease in economic burden, and more satisfaction with migraine treatment. In addition, the change in MIDAS scores from baseline showed a greater shift towards decreasing disability in the intervention group compared with the control group. However, no statistically significant improvement was detected in terms of health status as measured by SF-12. Of the participants in the intervention region who completed the program evaluation survey, 40% indicated that they called or visited their physicians regarding their migraine survey results if it was recommended. For those contacting their physicians, 76% had their medications changed and 75% noted an improvement in relief due to a change in medication.
Conclusions: Considering the significant toll of migraine on patients, employers, and the healthcare system, healthcare plans should consider implementing migraine disease management programs to improve migraine care.





Similar content being viewed by others
Notes
MTAQ® is a registered trademark of and copyrighted by Merck & Co., Inc.
References
Saper JR. Diagnosis and symptomatic treatment of migraine. Headache 1997; 37 Suppl. 1: S1–S14
Lipton RB, Stewart WF, Diamond S, et al. Prevalence and burden of migraine in the United States: data from the American Migraine Study II. Headache 2001; 41(7): 646–57
Osterhaus JT, Townsend RJ, Gandek B, et al. Measuring the functional status and well being of patients with migraine headache. Headache 1994; 34: 337–43
Solomon GD, Price KL. Burden of migraine: areview of its socioeconomic impact. Pharmacoeconomics 1997; 11 Suppl. 1: 1–10
Clouse JC, Osterhaus JT. Healthcare resource use and costs associated with migraine in a managed healthcare setting. Ann Pharmacother 1994; 28: 659–64
Hu X, Markson LE, Lipton RB, et al. Burden of migraine in the United States: disability and economic costs. Arch Intern Med 1999; 159: 813–8
Lipton RB, Silberstein SD, Stewart WF. An update on the epidemiology of migraine. Headache 1994; 34: 319–28
Edmeads J, Findlay H, Tugwell P, et al. Impact of migraine and tension-type headache on lifestyle, consulting behaviour and medication use: a Canadian population survey. Can J Neural Sci 1993; 20: 131–7
Hu X, O’Donnell F, Kunkel RS, et al. Survey of migraineurs referred to headache specialists: care, satisfaction, and outcomes. Neurology 2000; 55: 141–3
Lipton RB, Amatniek J, Gross M, et al. Identifying and removing barriers to care. Neurology 1994; 44 Suppl. 4: 63–8
Pryse-Phillips W, Dodic DW, Edmeads JG, et al. Guidelines for the diagnosis and management of migraine in clinical practice. CMAJ 1997; 156: 1273–87
Solomon GD, Cady R, Klapper J, et al. National Headache Foundation: standards of care for treating headache in primary care practice. Cleve Clin J Med 1997; 64: 373–83
Silberstein SD. Practice parameter: evidence-based guidelines for migraine headache (an evidence-based review). Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2000; 55: 754–62
Dowson AJ, Lipscombe S, Sender J, et al. New guidelines for the management of migraine in primary care. Curr Med Res Opin 2002; 18(7): 414–39
Epstein RS, Sherwood LM. From outcomes research to disease management: a guide for the perplexed. Ann Intern Med 1996; 124: 832–7
Data on file, Merck & Co., Inc, 1999
Solomon G, Hu X, Simmons R, et al. Development of migraine disease management initiative in a managed care plan. Dis Manage Health Outcomes 2002; 10: 167–73
Chatterton ML, Lofland JH, Shechter A, et al. Reliability and validity of the migraine therapy assessment questionnaire (MTAQ). Headache 2002; 42: 1006–15
Stewart WF, Lipton RB, Whyte J, et al. An international study to assess reliability of the Migraine Disability Assessment (MIDAS) score. Neurology 1999; 53(5): 988–94
Ware JE, Kosinski M, Keller SD. SF-12®: how to score the SF-12® physical and metal health summary scales. 3rd ed. Lincoln (RI): Quality Metric Incorporated, 1998
International classification of diseases 9th revision, clinical modification. Salt Lake City (UT): Utah Medicode Incorporated, 1997
Acknowledgments
This study was sponsored by Merck & Co., Inc.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hu, H.X., Solomon, G.D., Conboy, K. et al. Impact of a Migraine Disease Management Program. Dis-Manage-Health-Outcomes 12, 273–280 (2004). https://doi.org/10.2165/00115677-200412040-00007
Published:
Issue Date:
DOI: https://doi.org/10.2165/00115677-200412040-00007