Abstract
Primary open-angle glaucoma (POAG) is a chronic progressive disease characterised by damage to the optic nervehead (optic disc) which leads to visual field loss and may eventually result in blindness if left untreated. Intraocular pressure (IOP) is elevated in the majority of patients. Early intervention, based on reduction of IOP, can prevent or delay visual impairment and topical ocular hypotensive therapy is the mainstay of management. Argon laser trabeculoplasty or filtration surgery are most often used after failure of topical therapy but are also used as the initial intervention in some cases.
Latanoprost is an ester prodrug analogue of prostaglandin F2a available for the topical management of open-angle glaucoma and ocular hypertension. It lowers diurnal IOP by approximately 25 to 35%, by enhancing uveoscleral outflow of aqueous humour. This mechanism differs from that of other ocular hypotensive agents. Because its ocular hypotensive effects last for at least 24 hours after a single dose, latanoprost is administered once daily.
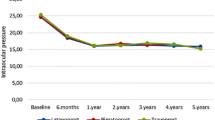
Comparative studies have shown that latanoprost 0.005% once daily is more effective than timolol 0.5% twice daily in lowering IOP. Additional reductions in IOP are achieved when latanoprost is used in combination with topical timolol or dipivefrin, or with oral acetazolamide.
The most common local adverse effects associated with latanoprost are eye irritation (burning, stinging, tearing and/or itching), mild conjunctival hyperaemia, superficial punctate keratopathy and foreign body sensation.
Increased pigmentation of the iris occurs in a variable proportion of patients treated with latanoprost. This phenomenon appears to be permanent, is most likely to occur in eyes with mixed iris colour and its long term clinical significance is unknown.
In contrast with timolol, latanoprost is not associated with systemic adverse effects, having essentially no effect on heart rate or systemic blood pressure.
Latanoprost is currently recommended for use in patients who respond inadequately to, or are unable to tolerate, other ocular hypotensive agents, or as firstline therapy, depending on the market. However, it has a number of favourable properties, which suggests that it has considerable potential for much wider use. Although further long term tolerability data are required, it appears possible that latanoprost and other topical prostaglandin analogues might largely replace β-adrenergic antagonists as first-line therapy for management of POAG and ocular hypertension in the future.
Similar content being viewed by others
References
Patel SS, Spencer CM. Latanoprost: a review of its pharmacological properties, clinical efficacy and tolerability in the management of primary open-angle glaucoma and ocular hypertension. Drugs Aging 1996 Nov; 9: 363–78.
David R, Livingston DG, Luntz MH. Ocular hypertension- a long-term follow-up of treated and untreated patients. Br J Ophthalmol 1977; 61: 668–74.
Hart Jr WM, Yablonski M, Kass MA, et al. Multivariate analysis of the risk of glaucomatous visual field loss. Arch Ophthalmol 1979; 97: 1455–8.
Kolker AE, Hetherington Jr J. Becker-Schaffer’s diagnosis and therapy of the glaucomas. 4th ed. St Louis: The C.V. Mosby Company, 1976.
Anonymous. Guidelines for the management of ocular hypertension and primary open angle glaucoma. Royal College of Ophthalmologists, London.
Quigley HA. Open-angle glaucoma [see comments]. N Engl J Med 1993 Apr 15; 328: 1097–106.
Rosenberg LF. Glaucoma: early detection and therapy for prevention of vision loss. Am Fam Physician 1995 Dec; 52: 2289–98.
Capino D, Leibowitz HM. Glaucoma: screening, diagnosis and therapy. Hosp Pract 1990 May 30: 73–91.
Nilsson SFE. The uveoscleral outflow route. Eye 1997; 11: 149–54.
Kini MM, Leibowitz HM, Colton T, et al. Prevalence of senile cataract, diabetic retinopathy, senile macular degeneration, and open-angle glaucoma in the Framingham Eye Study. Am J Ophthalmol 1978 Jan; 85: 28–34.
Leske MC, Connell AM, Wu S Y, et al. Risk factors for openangle glaucoma. The Barbados Eye Study [see comments]. Arch Ophthalmol 1995 Jul; 113: 918–24.
Sommer A. Intraocular pressure and glaucoma. Am J Ophthalmol 1989; 107: 186–8.
Quigley HA, Tielsch JM, Katz J, et al. Rate of progression in open-angle glaucoma estimated from cross-sectional prevalence of visual field damage. Am J Ophthalmol 1996 Sep; 122: 355–63.
Vogel R, Newson RB, Shipley M, et al. Association between intraocular pressure and loss of visual field in chronic simple glaucoma. Br J Ophthalmol 1990 Jan; 74: 3–6.
Johnson DH, Brubaker RF. Glaucoma: an overview. Mayo Clin Proc 1986 Jan; 61: 59–67.
Leibowitz HM, Krueger DE, Maunder DE, et al. The Framingham Eye Study monograph: an ophthalmological and epidemiological study of cataract, glaucoma, diabetic retinopathy, macular degeneration, and visual acuity in a general population of 2631 adults, 1973–1975. Surv Ophthalmol 1980; 24: 335–610.
Tielsch JM, Sommer A, Katz J, et al. Racial variations in the prevalence of primary open-angle glaucoma. The Baltimore Eye Survey. JAMA 1991 Jul 17; 266: 369–74.
Skuta GL, Morgan RK. Corticosteroid-induced glaucomas. In: Ritch R, Shields MB, Krupin T, editors. St Louis: Mosby-Year Book Inc, 1996: 1177–88.
Garbe E, LeLorier J, Boivin J-F, et al. Inhaled and nasal glucocorticoids and the risks of ocular hypertension or open-angle glaucoma. JAMA 1997 Mar 5; 277: 722–7.
Sommer A, Tielsch JM, Katz J, et al. Racial differences in the cause-specific prevalence of blindness in East Baltimore. N Engl J Med 1991; 325: 1412–7.
Bengtsson B. The incidence of manifest glaucoma. Br J Ophthalmol 1989; 73: 483–7.
Goddard M. Sight testing and the role of optometry. Centre For Health Economics Discussion Paper 64. University of York.
Quigley HA, Vitale S. Models of open-angle glaucoma prevalence and incidence in the United States. Invest Ophthalmol Vis Sci 1997; 38: 83–91.
Mason RP, Kosoko O, Wilson MR, et al. National survey of the prevalence and risk factors of glaucoma in St. Lucia, West Indies. Ophthalmology 1989 Sep; 96: 1363–8.
National Center for Health Statistics, Ganley J P, Roberts J. Eye conditions and related need for medical care among persons 1–74 years of age: United States 1971–72. Vital and health statistics. Series 11. No. 228. Government Printing Office, Washington, DC. DHHS publication no. (PHS) 83–1678.
Dreyer EB. Preserving eyesight with foresight. Harvard Health Lett 1994; 19: 4–6.
Coyle D, Drummond M. The economic burden of glaucoma in the UK: the need for a far-sighted policy. Pharmacoeconomics 1995 Jun; 7: 484–9.
Migdal C, Gregory W, Hitchings R. Long-term functional outcome after early surgery compared with laser and medicine in open-angle glaucoma. Ophthalmology 1994 Oct; 101: 1651–6. discussion 1657.
Taniguchi T, Kitazawa Y. A risk-benefit assessment of drugs used in the management of glaucoma. Drug Saf 1994 Jul; 11: 6-;74.
Jampel HD. Target pressure in glaucoma therapy. J Glaucoma 1997 Apr; 6: 133–8.
Sorensen SJ, Abel SR. Comparison of the occular beta-blockers. Ann Pharmacother 1996 Jan; 30: 43–54.
Balfour JA, Wilde MI. Dorzolamide: a review of its pharmacology and therapeutic potential in the management of glaucoma and ocular hypertension. Drugs Aging 1997 May; 10: 384–403.
Adkins JA, Balfour JA. Brimonidine: a review of its pharmacology and clinical potential in the management of glaucoma and ocular hypertension. Drugs Aging 1998; 12(3): 225–41.
Diamond JP. Systemic adverse effects of topical ophthalmic agents: implications for older patients. Drugs Aging 1997 Nov; 11: 352–60.
Collum LMT. Lung-function and glaucoma treatment — a cause for concern. Ir Med J 1995; 88(1): 16.
Diggory P, Franks WA. Glaucoma therapy may take your breath away. Age Ageing 1997 Mar; 26: 63–7.
Van Buskirk EM. Adverse reactions from timolol administration. Ophthalmology 1980; 87: 447–50.
Anonymous. Alcon Azopt for glaucoma claims reduced burning versus Trusopt. The Pink Sheet 1998 Apr 6: 4.
Serle JB. Pharmacological advances in the treatment of glaucoma. Drugs Aging 1994 Sep; 5: 156–70.
Haria M, Spencer CM. Unoprostone (isopropyl unoprostone). Drugs Aging 1996 Sep; 9: 213–8.
Haria M, Spencer CM. Unoprostone (isopropyl unoprostone). Erratum. Drugs Aging 1996 Nov; 9: 351.
Glaucoma Laser Trial Research Group. The Glaucoma Laser Trial. 1. Acute effects of argon laser trabeculoplasty on intraocular pressure. Arch Ophthalmol 1989; 107: 1135–42.
The Glaucoma Laser Trial Research Group. The Glaucoma Laser Trial (GLT). 2. Results of argon laser trabeculoplasty versus topical medicines. Ophthalmology 1990; 97: 1403–13.
Spaeth GL, Baez KA. Argon laser trabeculoplasty controls one third of cases of progressive, uncontrolled, open angle glaucoma for 5 years. Arch Ophthalmol 1992 Apr; 110: 491–4.
Brooks AM, Gillies WE. Laser trabeculoplasty——argon or diode? Aust N Z J Ophthalmol 1993 Aug; 21: 161–4.
Anonymous. Laser trabeculoplasty for primary open-angle glaucoma. Ophthalmology 1996 Oct; 103: 1706–12.
The Advanced Glaucoma Intervention Study Investigators. The Advanced Glaucoma Intervention Study (AGIS): 1. Study design and methods and baseline characteristics of study patients. Control Clin Trials 1994 Aug; 15: 299–325.
Hollo G. Argon and low energy, pulsed Nd:YAG laser trabeculoplasty. A prospective, comparative clinical and morphological study. Acta Ophthalmol Scand 1996 Apr; 74: 126–31.
Hamard P, Gayraud GM, Kopel J, et al. Treatment of refractory glaucoma by transcleral cyclophotocoagulation using semiconducter diode laser. Analysis of 50 patients followed-up for 19 months [in French]. J Fr Ophtalmol 1997; 20: 125–33.
Brooks AM, Gillies WE. The use of YAG cyclophotocoagulation to lower pressure in advanced glaucoma. Aust N Z J Ophthalmol 1991 Aug; 19: 207–10.
Molteno ACB, Van Rooyen MMB, Bartholomew RS. Implants for draining neovascular glaucoma. Br J Ophthalmol 1977; 61: 120–5.
Toris CB, Camras CB, Yablonski ME. Effects of PhXA41, a new prostaglandin Fd2α analog, on aqueous humor dynamics in human eyes. Ophthalmology 1993 Sep; 100: 1297–304.
Mishima HK, Kiuchi Y, Takamatsu M, et al. Circadian intraocular pressure management with latanoprost: diurnal and nocturnal intraocular pressure reduction and increased uveoscleral outflow. Surv Ophthalmol 1997 Feb; 41 Suppl. 2: S139–44.
Ziai N, Dolan JW, Kacere RD, et al. The effect on aqueous dynamics of PhXA41, a new prostaglandin Fd2α analogue, after topical application in normal and ocular hypertensive human eyes. Arch Ophthalmol 1993 Oct; 111: 1351–8.
Alm A, Villumsen J. PhXA34, a new potent ocular hypotensive drug. A study on dose-response relationship and on aqueous humor dynamics in healthy volunteers [see comments]. Arch Ophthalmol 1991 Nov; 109: 1564–8.
Lindén C, Alm A. Effects on intraocular pressure and aqueous flow of various dose regimens of latanoprost in human eyes. Acta Ophthalmol Scand 1997 Aug; 75: 412–5.
Rácz P, Ruzsonyi MR, Nagy ZT, et al. Maintained intraocular pressure reduction with once-a-day application of a new prostaglandin F2α analogue (PhXA41). An in-hospital, placebocontrolled study. Arch Ophthalmol 1993 May; 111: 657–61.
Nagasubramanian S, Sheth GP, Hitchings RA, et al. Intraocular pressure-reducing effect of PhXA41 in ocular hypertension: comparison of dose regimens. Ophthalmology 1993 Sep; 100: 1305–11.
Hotehama Y, Mishima HK, Kitazawa Y, et al. Ocular hypotensive effect of PhXA41 in patients with ocular hypertension or primary open-angle glaucoma. Jpn J Ophthalmol 1993; 37(3): 270–4.
Rácz P, Ruzsonyi MR, Nagy ZT, et al. Around-the-clock intraocular pressure reduction with once-daily application of latanoprost by itself or in combination with timolol. Arch Ophthalmol 1996 Mar; 114: 268–73.
Alm A, Stjernschantz J, Scandinavian Latanoprost Study Group. Effects on intraocular pressure and side effects of 0.005% latanoprost applied once daily, evening or morning: a comparison with timolol. Ophthalmology 1995 Dec; 102: 1743–52.
Watson P, Stjernschantz J, Latanoprost Study Group. A sixmonth, randomized, double masked study comparing latanoprost with timolol in open-angle glaucoma and ocular hypertertension. Ophthalmology 1996 Jan; 103: 126–37.
Rulo AH, Greve EL, Geijssen HC, et al. Reduction of intraocular pressure with treatment of latanoprost once daily in patients with normal-pressure glaucoma. Ophthalmology 1996 Aug; 103: 1276–82.
Camras CB, United States Latanoprost Study Group. Comparison of latanoprost and timolol in patients with ocular hypertension and glaucoma: a six-month, masked, multicenter trial in the United States. Ophthalmology 1996 Jan; 103: 138–47.
Mishima HK, Masuda K, Kitazawa Y, et al. A comparison of latanoprost and timolol in primary open-angle glaucoma and ocular hypertension: a 12-week study. Arch Ophthalmol 1996 Aug; 114: 929–32.
Camras CB, Alm A, Watson P, et al. Latanoprost, a prostaglandin analog, for glaucoma therapy: efficacy and safety after 1 year of treatment in 198 patients. Ophthalmology 1996 Nov; 103: 1916–24.
Watson PG, The Latanoprost Study Group. Latanoprost: two years’ experience of its use in the United Kingdom. Ophthalmology 1998; 105: 82–7.
Alm A, Widengård I, Kjellgren D, et al. Latanoprost administered once daily caused a maintained reduction of intraocular pressure in glaucoma patients treated concomitantly with timolol. Br J Ophthalmol 1995 Jan; 79: 12–6.
Rulo AH, Greve EL, Hoyng PF. Additive effect of latanoprost, a prostaglandin F-2 alpha analogue, and timolol in patients with elevated intraocular pressure. Br J Ophthalmol 1994 Dec; 78: 899–902.
Widengard I, Mäpea O, Alm A. Effects of latanoprost and dipivefrin, alone or combined, on intraocular pressure and on blood-aqueous barrier permeability. Br J Ophthalmol 1998; 82: 404–6.
Watson PG, Barnett F, Parker V. The additive effect of PhXA41 and propine in patients with primary open angle glaucoma [abstract no. 6a], Oxford, England, Jul 1992.
Rulo AH, Greve EL, Hoyng PFJ. Additive ocular hypotensive effect of latanoprost and acetazolamide. A short-term study in patients with elevated intraocular pressure. Ophthalmology 1997; 104: 1503–7.
Friström B, Nilsson SEG. Interaction of PhXA41, a new prostaglandin analogue, with pilocarpine. A study on patients with elevated intraocular pressure. Arch Ophthalmol 1993 May; 111: 662–5.
Lindén C, Alm A. Latanoprost and physostigmine have mostly additive ocular hypotensive effects in human eyes. Arch Ophthalmol 1997 Jul; 115: 857–61.
Alm A, Camras CB, Watson PG. Phase III latanoprost studies in Scandinavia, the United Kingdom and the United States. Surv Ophthalmol 1997 Feb; 41 Suppl. 2: S105–10.
Alm A, Villumsen J, Törnquist P, et al. Intraocular pressure-reducing effect of PhXA41 in patients with increased eye pressure: a one-month study. Ophthalmology 1993 Sep; 100: 1312–6.
Hotehama Y, Mishima HK. Clinical efficacy of PhXA34 and PhXA41, two novel prostaglandin-Fd2α-isopropyl ester analogues for glaucoma treatment. Jpn J Ophthalmol 1993; 37(3): 259–69.
Thygesen J, Aaen K, Theodorsen F, et al. Latanoprost and the ocular surface. XIth Congress of the European Society of Ophthalmology, Budapest, Hungary, 1997; Jun 1–5.
Kosaka T, Mishima HK, Kiuchi Y, et al. The effects of prostaglandins on the blood-ocular barrier. Jpn J Ophthalmol 1995; 39: 368–76.
Lindén C, Nuija E, Alm A. Effects on IOP restoration and bloodaqueous barrier after long term treatment with latanoprost in open angle glaucoma and ocular hypertension. Br J Ophthalmol 1997 May; 81: 370–2.
Nicolela MT, Buckley AR, Walman BE, et al. A comparative study of the effects of timolol and latanoprost on blood flow velocity of the retrobulbar vessels. Am J Ophthalmol 1996 Dec; 122: 784–9.
Rowe JA, Hattenhauer MG, Herman DC. Adverse side effects associated with latanoprost. Am J Ophthalmol 1997; 124: 683–5.
Selén G, Stjernschantz J, Resul B. Prostaglandin-induced iridial pigmentation in primates. Surv Ophthalmol 1997 Feb; 41 Suppl. 2: 125–8.
Wistrand PJ, Stjernschantz J, Olsson K. The incidence and timecourse of latanoprost-induced iridial pigmentation as a function of eye color. Surv Ophthalmol 1997 Feb; 41 Suppl. 2: 129–38.
Peak A, Sutton BM. Systemic adverse effects associated with topically applied latanoprost [letter]. Ann Pharmacother 1998; 32: 504–5.
Hedner J, Svedmyr N, Lunde H, et al. The lack of respiratory effects of the ocular hypotensive drug latanoprost in patients with moderate-steroid treated asthma. Surv Ophthalmol 1997 Feb; 41 Suppl. 2: S111–5.
Pharmacia & Upjohn Company. Xalatan prescribing information. Dec 1996, Kalamazoo, USA.
Brubaker RF. Flow of aqueous humor in humans. Inv Ophthalmol Vis Sci 1991; 32: 3145–66.
Topper JE, Brubaker RF. Effects of timolol, epinephrine, and acetazolamide on aqueous flow during sleep. Invest Ophthalmol Vis Sci 1985; 26: 1315–9.
Author information
Authors and Affiliations
Corresponding author
Additional information
Various sections of the manuscript reviewed by: A. Alm, Department of Ophthalmology, University Hospital, Uppsala, Sweden; C.B. Camras, Department of Ophthalmology, University of Nebraska Medical Center, Omaha, Nebraska, USA; J.P. Diamond, Directorate of Ophthalmology, Bristol Eye Hospital, Bristol, England; W.A. Franks, Glaucoma Unit, Moorfields Eye Hospital, London, England; P.F.J. Hoyng, The Netherlands Ophthalmic Research Institute, Amsterdam, The Netherlands; J.D.C. Macdiarmid, Hamilton Eye Clinic, Hamilton, New Zealand; H.K. Mishima, Department of Ophthalmology, Hiroshima University, School of Medicine, Hiroshima, Japan; H.A. Quigley, Johns Hopkins Hospital, Baltimore, Maryland, USA; P. Watson, Cambridge, England.
Data Selection
Sources: Medical literature published in any language since 1966 on latanoprost, identified using AdisBase (a proprietary database of Adis International, Auckland, New Zealand) and Medline. Additional references were identified from the reference lists of published articles. Bibliographical information, including contributory unpublished data, was also requested from the company developing the drug.
Search strategy: AdisBase search terms were ‘latanoprost’, ‘glaucoma’, ‘reviews on treatment’, ‘epidemiology’ and ‘cost of illness’. Medline search terms were ‘guidelines’, ‘decision-making’, ‘health policy’, ‘managed care programs’, ‘guideline’, ‘consensus’, ‘policy’, ‘disease management’, ‘algorithms’, ‘epidemiology’, ‘practice guideline’, ‘expert panel’, ‘utilization review’, ‘glaucoma’ and ‘epidemiology’, ‘etiology’, and ‘economics’. Searches were last updated 14 July 1998.
Selection: Studies in patients with glaucoma or ocular hypertension who received latanoprost. Inclusion of studies was based mainly on the methods section of the trials. When available, large, well controlled trials with appropriate statistical methodology were preferred. Relevant pharmacodynamic and pharmacokinetic data are also included, as well as background data on pathophysiology and epidemiology and management guidelines.
Index terms: Latanoprost, glaucoma, ocular hypertension, therapeutic use, disease management, epidemiology, reviews on treatment.
Rights and permissions
About this article
Cite this article
Balfour, J.A., Adkins, J.C. Management of Primary Open-Angle Glaucoma and Ocular Hypertension: The Potential Role of Latanoprost. Dis-Manage-Health-Outcomes 4, 101–112 (1998). https://doi.org/10.2165/00115677-199804020-00004
Published:
Issue Date:
DOI: https://doi.org/10.2165/00115677-199804020-00004