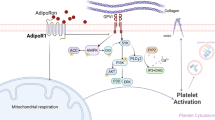
Abstract
Peroxisome proliferator-activated receptor γ (PPARγ) is an important transcription factor for lipid and glucose metabolism. Currently, the PPARγ ligands rosiglitazone and pioglitazone are used for the treatment of type 2 diabetes mellitus because they are potent insulin sensitizers. Recently, PPARγ has emerged as an important anti-inflammatory factor. Platelets, anucleate cells involved in hemostasis, have also been implicated as key contributors to inflammation, because they produce many pro-inflammatory and pro-atherogenic mediators when activated. Surprisingly, it was discovered recently that platelets contain PPARγ and that PPARγ ligands, both natural and synthetic, inhibit platelet activation and release of bioactive mediators. In particular, release of soluble CD40 ligand (sCD40L) and thromboxane (TXA2) was inhibited by PPARγ ligands in thrombin-activated platelets. CD40L signaling induces pro-inflammatory processes in many cell types, and increased blood levels of sCD40L are closely associated with inflammation, diabetes, and cardiovascular disease. Targeting platelet PPARγ will, therefore, be an important treatment strategy for the attenuation of chronic inflammatory processes and prevention of thrombus formation.




Similar content being viewed by others
References
von Daniken A, Lutz WK, Schlatter C. Lack of covalent binding to rat liver DNA of the hypolipidemic drugs clofibrate and fenofibrate. Toxicol Lett 1981; 7: 305–10
Issemann I, Green S. Cloning of novel members of the steroid hormone receptor superfamily. J Steroid Biochem Mol Biol 1991; 40: 263–9
Braissant O, Foufelle F, Scotto C, et al. Differential expression of peroxisome proliferator-activated receptors (PPARs): tissue distribution of PPAR-alpha, — beta, and -gamma in the adult rat. Endocrinology 1996; 137: 354–66
Kliewer SA, Umesono K, Noonan DJ, et al. Convergence of 9-cis retinoic acid and peroxisome proliferator signalling pathways through heterodimer formation of their receptors. Nature 1992; 358: 771–4
Barish GD, Narkar VA, Evans RM. PPARδ: a dagger in the heart of the metabolic syndrome. J Clin Invest 2006; 116: 590–7
Chung SW, Kang BY, Kim SH, et al. Oxidized low density lipoprotein inhibits interleukin-12 production in lipopolysaccharide-activated mouse macrophages via direct interactions between peroxisome proliferator-activated receptor-γ and nuclear factor-κ B. J Biol Chem 2000; 275: 32681–7
Kelly D, Campbell JI, King TP, et al. Commensal anaerobic gut bacteria attenuate inflammation by regulating nuclear-cytoplasmic shuttling of PPAR-γ and RelA. Nat Immunol 2004; 5: 104–12
Zhu Y, Qi C, Korenberg JR, et al. Structural organization of mouse peroxisome proliferator-activated receptor gamma (mPPAR gamma) gene: alternative promoter use and different splicing yield two mPPAR gamma isoforms. Proc Natl Acad Sci U S A 1995; 92: 7921–5
Fajas L, Auboeuf D, Raspe E, et al. The organization, promoter analysis, and expression of the human PPARgamma gene. J Biol Chem 1997; 272: 18779–89
Ricote M, Huang J, Fajas L, et al. Expression of the peroxisome proliferator-activated receptor gamma (PPARgamma) in human atherosclerosis and regulation in macrophages by colony stimulating factors and oxidized low density lipoprotein. Proc Natl Acad Sci U S A 1998; 95: 7614–9
Akbiyik F, Ray DM, Gettings KF, et al. Human bone marrow megakaryocytes and platelets express PPARγ, and PPARγ agonists blunt platelet release of CD40 ligand and thromboxanes. Blood 2004; 104: 1361–8
Harris SG, Phipps RP. The nuclear receptor PPAR γ is expressed by mouse T lymphocytes and PPAR γ agonists induce apoptosis. Eur J Immunol 2001; 31: 1098–105
Iijima K, Yoshizumi M, Ako J, et al. Expression of peroxisome proliferator-activated receptor γ (PPARγ) in rat aortic smooth muscle cells. Biochem Biophys Res Commun 1998; 247: 353–6
Nencioni A, Grunebach F, Zobywlaski A, et al. Dendritic cell immunogenicity is regulated by peroxisome proliferator-activated receptor γ. J Immunol 2002; 169: 1228–35
Padilla J, Leung E, Phipps P. Human B lymphocytes and B lymphomas express PPAR-γ and are killed by PPAR-γ agonists. Clin Immunol 2002; 103: 22–33
Ricote M, Li AC, Willson M, et al. The peroxisome proliferator-activated receptor-γ is a negative regulator of macrophage activation. Nature 1998; 391: 79–82
Su CG, Wen X, Bailey T, et al. A novel therapy for colitis utilizing PPAR-γ ligands to inhibit the epithelial inflammatory response. J Clin Invest 1999; 104: 383–9
Lehmann JM, Moore LB, Smith-Oliver A, et al. An antidiabetic thiazolidinedione is a high affinity ligand for peroxisome proliferator-activated receptor γ (PPAR γ). J Biol Chem 1995; 270: 12953–6
Forman BM, Tontonoz P, Chen, et al. 15-Deoxy-delta 12, 14-prostaglandin J2 is a ligand for the adipocyte determination factor PPAR γ. Cell 1995; 83: 803–12
Kliewer SA, Lenhard JM, Willson M, et al. A prostaglandin J2 metabolite binds peroxisome proliferator-activated receptor γ and promotes adipocyte differentiation. Cell 1995; 83: 813–9
McIntyre TM, Pontsler AV, Silva AR, et al. Identification of an intracellular receptor for lysophosphatidic acid (LPA): LPA is a transcellular PPARγ agonist. Proc Natl Acad Sci U S A 2003; 100: 131–6
Schopfer FJ, Lin Y, Baker PR, et al. Nitrolinoleic acid: an endogenous peroxisome proliferator-activated receptor γ ligand. Proc Natl Acad Sci U S A 2005; 102: 2340–5
Guan Y, Hao C, Cha DR, et al. Thiazolidinediones expand body fluid volume through PPARγ stimulation of ENaC-mediated renal salt absorption. Nat Med 2005; 11: 861–6
Fagerberg B, Edwards S, Halmos T, et al. Tesaglitazar, a novel dual peroxisome proliferator-activated receptor α/γ agonist, dose-dependently improves the metabolic abnormalities associated with insulin resistance in a non-diabetic population. Diabetologia 2005; 48: 1716–25
Skrumsager BK, Nielsen KK, Muller M, et al. Ragaglitazar: the pharmacokinetics, pharmacodynamics, and tolerability of a novel dual PPAR α and γ agonist in healthy subjects and patients with type 2 diabetes. J Clin Pharmacol 2003; 43: 1244–56
Brand CL, Sturis J, Gotfredsen CF, et al. Dual PPARα/γ activation provides enhanced improvement of insulin sensitivity and glycemie control in ZDF rats. Am J Physiol Endocrinol Metab 2003; 284: E841–54
Nissen SE, Wolski K, Topol EJ. Effect of muraglitazar on death and major adverse cardiovascular events in patients with type 2 diabetes mellitus. JAMA 2005; 294: 2581–6
Liu H, Zang C, Fenner MH, et al. Growth inhibition and apoptosis in human Philadelphia chromosome positive lymphoblastic leukemia cell lines by treatment with the dual PPARα/γ ligand TZD18. Blood 2006 May 1; 107(9): 3683–92
Rangwala SM, Lazar MA. The dawn of the SPPARMs? Sci STKE 2002; 2002: PE9
Nolte RT, Wisely GB, Westin S, et al. Ligand binding and co-activator assembly of the peroxisome proliferator-activated receptor-γ. Nature 1998; 395: 137–43
Cock TA, Houten SM, Auwerx J. Peroxisome proliferator-activated receptor-γ. too much of a good thing causes harm. EMBO Rep 2004; 5: 142–7
Rocchi S, Picard F, Vamecq J, et al. A unique PPARγ ligand with potent insulin-sensitizing yet weak adipogenic activity. Mol Cell 2001; 8: 737–47
Wang Y, Porter WW, Suh N, et al. A synthetic triterpenoid, 2-cyano-3,12-dioxooleana-1,9-dien-28-oic acid (CDDO), is a ligand for the peroxisome proliferator-activated receptor γ. Mol Endocrinol 2000; 14: 1550–6
Misra P, Chakrabarti R, Vikramadithyan K, et al. PAT5A: a partial agonist of peroxisome proliferator-activated receptor γ is a potent antidiabetic thiazolidinedione yet weakly adipogenic. J Pharmacol Exp Ther 2003; 306: 763–71
Palakurthi SS, Aktas H, Grubissich LM, et al. Anticancer effects of thiazolidinediones are independent of peroxisome proliferator-activated receptor gamma and mediated by inhibition of translation initiation. Cancer Res 2001; 61: 6213–8
Chawla A, Barak Y, Nagy L, et al. PPAR-γ dependent and independent effects on macrophage-gene expression in lipid metabolism and inflammation. Nat Med 2001; 7: 48–52
Moore KJ, Rosen ED, Fitzgerald ML, et al. The role of PPAR-γ in macrophage differentiation and cholesterol uptake. Nat Med 2001; 7: 41–7
Clay CE, Monjazeb A, Thorburn J, et al. 15-Deoxy-delta 12, 14-prostaglandin J2-induced apoptosis does not require PPARγ in breast cancer cells. J Lipid Res 2002; 43: 1818–28
Shiau CW, Yang CC, Kulp SK, et al. Thiazolidenediones mediate apoptosis in prostate cancer cells in part through inhibition of Bcl-xL/Bcl-2 functions independently of PPARγ. Cancer Res 2005; 65: 1561–9
Han S, Roman J. Rosiglitazone suppresses human lung carcinoma cell growth through PPARγ-dependent and PPARγ-independent signal pathways. Mol Cancer Ther 2006; 5: 430–7
Fukami MH, Salganicoff L. Human platelet storage organelles: a review. Thromb Haemost 1977; 38: 963–70
Harrison P, Cramer EM. Platelet alpha-granules. Blood Rev 1993; 7: 52–62
Denis MM, Tolley ND, Bunting M, et al. Escaping the nuclear confines: signal-dependent pre-mRNA splicing in anucleate platelets. Cell 2005; 122: 379–91
Gnatenko DV, Dunn JJ, McCorkle SR, et al. Transcript profiling of human platelets using microarray and serial analysis of gene expression. Blood 2003; 101: 2285–93
Kieffer N, Guichard J, Farcet JP, et al. Biosynthesis of major platelet proteins in human blood platelets. Eur J Biochem 1987; 164: 189–95
Henn V, Slupsky JR, Grafe M, et al. CD40 ligand on activated platelets triggers an inflammatory reaction of endothelial cells. Nature 1998; 391: 591–4
Chapelsky MC, Thompson-Culkin K, Miller AK, et al. Pharmacokinetics of rosiglitazone in patients with varying degrees of renal insufficiency. J Clin Pharmacol 2003; 43: 252–9
Thompson-Culkin K, Zussman B, Miller AK, et al. Pharmacokinetics of rosiglitazone in patients with end-stage renal disease. J Int Med Res 2002; 30: 391–9
Bodary PF, Vargas FB, King SA, et al. Pioglitazone protects against thrombosis in a mouse model of obesity and insulin resistance. J Thromb Haemost 2005; 3: 2149–53
Li D, Chen K, Sinha N, et al. The effects of PPAR-γ ligand pioglitazone on platelet aggregation and arterial thrombus formation. Cardiovasc Res 2005; 65: 907–12
Haseruck N, Erl W, Pandey D, et al. The plaque lipid lysophosphatidic acid stimulates platelet activation and platelet-monocyte aggregate formation in whole blood: involvement of P2Y1 and P2Y12 receptors. Blood 2004; 103: 2585–92
Liu F, Morris S, Epps J, et al. Demonstration of an activation regulated NF-κB/I-κBα complex in human platelets. Thromb Res 2002; 106: 199–203
Gelman L, Michalik L, Desvergne B, et al. Kinase signaling cascades that modulate peroxisome proliferator-activated receptors. Curr Opin Cell Biol 2005; 17: 216–22
Pascual G, Fong AL, Ogawa S, et al. A SUMOylation-dependent pathway mediates transrepression of inflammatory response genes by PPAR-γ. Nature 2005; 437: 759–63
Ali FY, Davidson SJ, Moraes LA, et al. Role of nuclear receptor signaling in platelets: antithrombotic effects of PPARβ. Faseb J 2006; 20: 326–8
Barak Y, Nelson MC, Ong ES, et al. PPARγ is required for placental, cardiac, and adipose tissue development. Mol Cell 1999; 4: 585–95
Ryding AD, Sharp MG, Mullins JJ. Conditional transgenic technologies. J Endocrinol 2001; 171: 1–14
Setoguchi K, Misaki Y, Terauchi Y, et al. Peroxisome proliferator-activated receptor-γ haploinsufficiency enhances B cell proliferative responses and exacerbates experimentally induced arthritis. J Clin Invest 2001; 108: 1667–75
Danese S, Fiocchi C. Platelet activation and the CD40/CD40 ligand pathway: mechanisms and implications for human disease. Crit Rev Immunol 2005; 25: 103–21
Inwald DP, McDowall A, Peters MJ, et al. CD40 is constitutively expressed on platelets and provides a novel mechanism for platelet activation. Circ Res 2003; 92: 1041–8
Furman MI, Krueger LA, Linden MD, et al. Release of soluble CD40L from platelets is regulated by glycoprotein Ilb/IIIa and actin polymerization. J Am Coll Cardiol 2004; 43: 2319–25
Zhang Y, Cao HJ, Graf B, et al. CD40 engagement up-regulates cyclooxygenase-2 expression and prostaglandin E2 production in human lung fibroblasts. J Immunol 1998; 160: 1053–7
Andre P, Nannizzi-Alaimo L, Prasad SK, et al. Platelet-derived CD40L: the switch-hitting player of cardiovascular disease. Circulation 2002; 106: 896–9
Elzey BD, Tian J, Jensen RJ, et al. Platelet-mediated modulation of adaptive immunity: a communication link between innate and adaptive immune compartments. Immunity 2003; 19: 9–19
Sempowski GD, Chess PR, Moretti AJ, et al. CD40 mediated activation of gingival and periodontal ligament fibroblasts. J Periodontol 1997; 68: 284–92
Sempowski GD, Chess PR, Phipps RP. CD40 is a functional activation antigen and B7-independent T cell costimulatory molecule on normal human lung fibroblasts. J Immunol 1997; 158: 4670–7
Quera R, Shanahan F. Thromboembolism -an important manifestation of inflammatory bowel disease. Am J Gastroenterol 2004; 99: 1971–3
Danese S, de la Motte C, Sturm A, et al. Platelets trigger a CD40-dependent inflammatory response in the microvasculature of inflammatory bowel disease patients. Gastroenterology 2003; 124: 1249–64
Danese S, Katz JA, Saibeni S, et al. Activated platelets are the source of elevated levels of soluble CD40 ligand in the circulation of inflammatory bowel disease patients. Gut 2003; 52: 1435–41
Ludwiczek O, Kaser A, Tilg H. Plasma levels of soluble CD40 ligand are elevated in inflammatory bowel diseases. Int J Colorectal Dis 2003; 18: 142–7
Ghanim H, Garg R, Aljada A, et al. Suppression of nuclear factor-κB and stimulation of inhibitor κB by troglitazone: evidence for an anti-inflammatory effect and a potential antiatherosclerotic effect in the obese. J Clin Endocrinol Metab 2001;86: 1306–12
Mohanty P, Aljada A, Ghanim H, et al. Evidence for a potent antiinflammatory effect of rosiglitazone. J Clin Endocrinol Metab 2004; 89: 2728–35
Vinik AI, Erbas T, Park TS, et al. Platelet dysfunction in type 2 diabetes. Diabetes Care 2001; 24: 1476–85
Almer LO, Nilsson IM. On fibrinolysis in diabetes mellitus. Acta Med Scand 1975; 198: 101–6
Imperatore G, Riccardi G, Iovine C, et al. Plasma fibrinogen: a new factor of the metabolic syndrome: a population-based study. Diabetes Care 1998; 21: 649–54
Betteridge DJ, El Tahir KE, Reckless JP, et al. Platelets from diabetic subjects show diminished sensitivity to prostacyclin. Eur J Clin Invest 1982; 12: 395–8
Bucala R, Tracey KJ, Cerami A. Advanced glycosylation products quench nitric oxide and mediate defective endothelium-dependent vasodilatation in experimental diabetes. J Clin Invest 1991; 87: 432–8
Johnson M, Harrison HE, Raftery AT, et al. Vascular prostacyclin may be reduced in diabetes in man. Lancet 1979; 1: 325–6
Trovati M, Mularoni EM, Burzacca S, et al. Impaired insulin-induced platelet antiaggregating effect in obesity and in obese NIDDM patients. Diabetes 1995; 44: 1318–22
Zeng G, Quon MJ. Insulin-stimulated production of nitric oxide is inhibited by wortmannin: direct measurement in vascular endothelial cells. J Clin Invest 1996; 98: 894–8
Dandona P, Aljada A, Mohanty P, et al. Insulin inhibits intranuclear nuclear factor κB and stimulates IκB in mononuclear cells in obese subjects: evidence for an anti-inflammatory effect? J Clin Endocrinol Metab 2001; 86: 3257–65
Akai T, Naka K, Okuda K, et al. Decreased sensitivity of platelets to prostacyclin in patients with diabetes mellitus. Horm Metab Res 1983; 15: 523–6
Schafer A, Alp NJ, Cai S, et al. Reduced vascular NO bioavailability in diabetes increases platelet activation in vivo. Arterioscler Thromb Vasc Biol 2004; 24: 1720–6
Halushka PV, Rogers RC, Loadholt CB, et al. Increased platelet thromboxane synthesis in diabetes mellitus. J Lab Clin Med 1981; 97: 87–96
Dandona P, Aljada A, Bandyopadhyay A. Inflammation: the link between insulin resistance, obesity and diabetes. Trends Immunol 2004; 25: 4–7
Jinchuan Y, Zonggui W, Jinming C, et al. Upregulation of CD40-CD40 ligand system in patients with diabetes mellitus. Clin Chim Acta 2004; 339: 85–90
Varo N, Libby P, Nuzzo R, et al. Elevated release of sCD40L from platelets of diabetic patients by thrombin, glucose and advanced glycation end products. Diab Vasc Dis Res 2005; 2: 81–7
Vaidyula VR, Rao AK, Mozzoli M, et al. Effects of hyperglycemia and hyperinsulinemia on circulating tissue factor procoagulant activity and platelet CD40 ligand. Diabetes 2006; 55: 202–8
Santilli F, Davi G, Consoli A, et al. Thromboxane-dependent CD40 ligand release in type 2 diabetes mellitus. J Am Coll Cardiol 2006; 47: 391–7
Loe H. Periodontal disease: the sixth complication of diabetes mellitus. Diabetes Care 1993; 16: 329–34
Genco RJ, Grossi SG, Ho A, et al. A proposed model linking inflammation to obesity, diabetes, and periodontal infections. J Periodontol 2005; 76: 2075–84
Mackler BF, Waldrop TC, Schur P, et al. IgG subclasses in human periodontal disease. I. Distribution and incidence of IgG subclass bearing lymphocytes and plasma cells. J Periodontal Res 1978; 13: 109–19
Morton RS, Dongari-Bagtzoglou AI. Cyclooxygenase-2 is upregulated in inflamed gingival tissues. J Periodontol 2001; 72: 461–9
Offenbacher S, Salvi GE. Induction of prostaglandin release from macrophages by bacterial endotoxin. Clin Infect Dis 1999; 28: 505–13
Marx N, Imhof A, Froehlich J, et al. Effect of rosiglitazone treatment on soluble CD40L in patients with type 2 diabetes and coronary artery disease. Circulation 2003; 107: 1954–7
Varo N, Vicent D, Libby P, et al. Elevated plasma levels of the atherogenic mediator soluble CD40 ligand in diabetic patients: a novel target of thiazolidinediones. Circulation 2003; 107: 2664–9
Sidhu JS, Cowan D, Tooze JA, et al. Peroxisome proliferator-activated receptor-γ agonist rosiglitazone reduces circulating platelet activity in patients without diabetes mellitus who have coronary artery disease. Am Heart J 2004; 147: e25
Libby P, Ridker PM, Maseri A. Inflammation and atherosclerosis. Circulation 2002; 105: 1135–43
Lusis AJ. Atherosclerosis. Nature 2000; 407: 233–41
Ross R. Atherosclerosis -an inflammatory disease. N Engl J Med 1999; 340: 115–26
Gallino A, Haeberli A, Baur HR, et al. Fibrin formation and platelet aggregation in patients with severe coronary artery disease: relationship with the degree of myocardial ischemia. Circulation 1985; 72: 27–30
Fuster V, Badimon L, Badimon JJ, et al. The pathogenesis of coronary artery disease and the acute coronary syndromes (2). N Engl J Med 1992; 326: 310–8
Ross R. The pathogenesis of atherosclerosisan update. N Engl J Med 1986; 314: 488–500
Wagner DD, Burger PC. Platelets in inflammation and thrombosis. Arterioscler Thromb Vasc Biol 2003; 23: 2131–7
Badimon JJ, Zaman A, Helft G, et al. Acute coronary syndromes: pathophysiology and preventive priorities. Thromb Haemost 1999; 82: 997–1004
Fuster V, Badimon L, Badimon JJ, et al. The pathogenesis of coronary artery disease and the acute coronary syndromes (1). N Engl J Med 1992; 326: 242–50
Vorchheimer DA, Becker R. Platelets in atherothrombosis. Mayo Clin Proc 2006; 81: 59–68
Gawaz M, Langer H, May AE. Platelets in inflammation and atherogenesis. J Clin Invest 2005; 115: 3378–84
Dong ZM, Chapman SM, Brown AA, et al. The combined role of P- and E-selectins in atherosclerosis. J Clin Invest 1998; 102: 145–52
Huo Y, Schober A, Forlow SB, et al. Circulating activated platelets exacerbate atherosclerosis in mice deficient in apolipoprotein E. Nat Med 2003; 9: 61–7
Langer H, May AE, Daub K, et al. Adherent platelets recruit and induce differentiation of murine embryonic endothelial progenitor cells to mature endothelial cells in vitro. Circ Res 2006; 98: e2–10
Andre P, Prasad KS, Denis CV, et al. CD40L stabilizes arterial thrombi by a β3 integrin-dependent mechanism. Nat Med 2002; 8: 247–52
Lutgens E, Gorelik L, Daemen MJ, et al. Requirement for CD154 in the progression of atherosclerosis. Nat Med 1999; 5: 1313–6
Mach F, Schonbeck U, Sukhova GK, et al. Functional CD40 ligand is expressed on human vascular endothelial cells, smooth muscle cells, and macrophages: implications for CD40-CD40 ligand signaling in atherosclerosis. Proc Natl Acad Sci U S A 1997; 94: 1931–6
Phipps RP. Atherosclerosis: the emerging role of inflammation and the CD40-CD40 ligand system. Proc Natl Acad Sci U S A 2000; 97: 6930–2
Aukrust P, Muller F, Ueland T, et al. Enhanced levels of soluble and membranebound CD40 ligand in patients with unstable angina: possible reflection of T lymphocyte and platelet involvement in the pathogenesis of acute coronary syndromes. Circulation 1999; 100: 614–20
Garlichs CD, Eskafi S, Raaz D, et al. Patients with acute coronary syndromes express enhanced CD40 ligand/CD154 on platelets. Heart 2001; 86(6): 649–55
Garlichs CD, John S, Schmeisser A, et al. Upregulation of CD40 and CD40 ligand (CD154) in patients with moderate hypercholesterolemia. Circulation 2001; 104: 2395–400
Lee Y, Lee WH, Lee SC, et al. CD40L activation in circulating platelets in patients with acute coronary syndrome. Cardiology 1999; 92(1): 11–6
Slupsky JR, Kalbas M, Willuweit A, et al. Activated platelets induce tissue factor expression on human umbilical vein endothelial cells by ligation of CD40. Thromb Haemost 1998; 80: 1008–14
Lutgens E, Cleutjens KB, Heeneman S, et al. Both early and delayed anti-CD40L antibody treatment induces a stable plaque phenotype. Proc Natl Acad Sci U S A 2000; 97: 7464–9
Sukhova GK, Schonbeck U, Rabkin E, et al. Evidence for increased collagenolysis by interstitial collagenases-1 and -3 in vulnerable human atheromatous plaques. Circulation 1999; 99: 2503–9
Schonbeck U, Sukhova GK, Shimizu K, et al. Inhibition of CD40 signaling limits evolution of established atherosclerosis in mice. Proc Natl Acad Sci U S A 2000; 97: 7458–63
Vishnevetsky D, Kiyanista VA, Gandhi PJ. CD40 ligand: a novel target in the fight against cardiovascular disease. Ann Pharmacother 2004; 38: 1500–8
Duval C, Chinetti G, Trottein F, et al. The role of PPARs in atherosclerosis. Trends Mol Med 2002; 8: 422–30
Lehrke M, Lazar MA. The many faces of PPARγ. Cell 2005; 123: 993–9
Wang M, Tafuri S. Modulation of PPARγ activity with pharmaceutical agents: treatment of insulin resistance and atherosclerosis. J Cell Biochem 2003; 89: 38–47
Li AC, Binder CJ, Gutierrez A, et al. Differential inhibition of macrophage foam-cell formation and atherosclerosis in mice by PPARα, β/δ, and γ. J Clin Invest 2004; 114: 1564–76
Samaha FF, Szapary PO, Iqbal N, et al. Effects of rosiglitazone on lipids, adipokines, and inflammatory markers in nondiabetic patients with low high-density lipoprotein cholesterol and metabolic syndrome. Arterioscler Thromb Vasc Biol 2006; 26: 624–30
Azar RR, Kassab R, Zoghbi A, et al. Effects of clopidogrel on soluble CD40 ligand and on high-sensitivity C-reactive protein in patients with stable coronary artery disease. Am Heart J 2006; 151: 521e1–4
Xiao Z, Theroux P. Clopidogrel inhibits platelet-leukocyte interactions and thrombin receptor agonist peptide-induced platelet activation in patients with an acute coronary syndrome. J Am Coll Cardiol 2004; 43: 1982–8
Zondlo SC, Wang X, Feuerstein GZ. Effects of orally active glycoprotein IIb/IIIa antagonists on platelet CD40 ligand (CD154) expression and platelet-heteroag-gregate formation. Thromb Res 2002; 106: 35–40
Acknowledgements
This work was supported by T32-DE07165, T32-ES07026, DE011390, and HL078603. The authors declare that no conflicts of interest exist.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ray, D.M., Spinelli, S.L., O’Brien, J.J. et al. Platelets as a Novel Target for PPARγ Ligands. BioDrugs 20, 231–241 (2006). https://doi.org/10.2165/00063030-200620040-00004
Published:
Issue Date:
DOI: https://doi.org/10.2165/00063030-200620040-00004