Abstract
Anaemia, a major feature of chronic renal failure, is responsible for a significant proportion of the multisystemic disabling symptoms in patients with chronic renal failure (CRF). Anaemia correction with recombinant human erythropoietin (epoetin-α) often leads to significant amelioration of these symptoms.
Starting dose should be 50 U/kg of bodyweight given subcutaneously or intravenously 1, 2 or 3 times a week. The subcutaneous route is more effective, resulting in use of less epoetin-α and subsequent cost-saving. Adverse effects of epoetin-α tend to be more severe and more likely following intravenous administration.
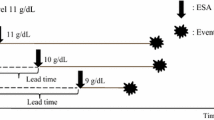
Once the target haematocrit is achieved, the epoetin-α dose should be reduced by 50 to 80%, but should not be stopped, since an abrupt fall in haematocrit may ensue if epoetin-α is completely discontinued. There is an emerging consensus that it is deleterious, and not necessary, to allow patients to become severely anaemic before starting epoetin-α. Thus, the current trend is to start epoetin-α once the haematocrit is 30 to 35%. On the other hand, what the target haematocrit should be is controversial. The threshold haematocrit beyond which the risks of anaemia correction outweigh the benefits in patients with CRF is not established. Studies that assessed quality of life outcomes as well as clinical experience in Europe clearly demonstrate better outcomes without adverse effects in patients with a haematocrit between 36 to 40%, compared to those with haematocrit of 30%.
Prior to starting epoetin-α in any patient with CRF, a methodological evalution should be performed to exclude other major causes of anaemia and to identify any comorbid conditions that may impair response to epoetin-α. Response to epoetin-α may be impaired by loss of residual renal function in patients with progressive CRF or kidney transplant recipients. Other major factors that impair response to epoetin-α include impaired utilisation of iron, absolute iron deficiency, inadequate dialysis, infection and bone marrow fibrosis.
Similar content being viewed by others
References
Eschbach JW. The anaemia of chronic renal failure: pathophysiology and the effects of recombinant erythropoietin. Kidney Inter 1989; 35: 134–48
Lundin AP. Quality of life: subjective and objective improvements with recombinant human erythropoietin therapy. Seminars in Nephrology 1989; 9: 22–9
Braumann KM, Nonnast-Daniel B, Boning D, et al. Improved physical performance after treatment of renal anaemia with recombinant human erythropoietin. Nephron 1991; 58: 129–34
Ifudu O, Mayer J, Matthew J, et al. Dismal rehabilitation in geriatric inner-city hemodialysis patients. JAMA 1994; 271: 29–33
Ifudu O, Dawood M, Homel P, et al. Excess morbidity in patients starting uremia therapy without prior care by a nephrologist. Am J Kidney Dis 1996; 28: 841–5
Lowrie EG, Huang WH, Lew NL, et al. The relative contribution of measured variables to death risk among hemodialysis patients. In: Death on hemodialysis: preventable or inevitable. Friedman EA, editor. Dordrecht: Kluwer Academic Publishers, 1994: 121–4
Boddy K, Lawson DH, Linton AL, et al. Iron metabolism in patients with chronic renal failure. Clin Sci 1970; 39: 115–21
Akmal M, Sawelson S, Karubian F, et al. The prevalence and significance of occult blood loss in patients with predialysis advanced chronic renal failure or receiving dialytic therapy. Clin Nephrol 1994; 42: 198–202
Zuckerman GR, Cornette GL, Clouse RE, et al. Upper gastrointestinal bleeding in patients with chronic renal failure. Ann Intern Med 1985; 102: 588–92
Silverberg DS, Iaina A, Peer G, et al. Intravenous iron supplementation for the treatment of the anaemia of moderate to severe chronic renal failure patients not receiving dialysis. Am J Kidney Dis 1996; 27: 234–8
Ifudu O, Dawood M, Friedman EA. Relative contributions of body iron status and uremia severity to anaemia in patients with advanced chronic renal failure. Nephron 1997; 77: 315–8
Beamish MR, Davies AG, Eakins JD, et al. The measurement of reticuloendothelial iron release using iron-dextran. Br J Haem 1971; 21: 617–22
Parker KP, Mitch WE, Stivelman JC, et al. Safety and efficacy of low-dose subcutaneous erythropoietin in hemodialysis patients. J Am Soc Nephrol 1997; 8: 288–93
Albitar S, Meulders Q, Hammoud H, et al. Subcutaneous versus intravenous administration of erythropoietin improves efficiency for the treatment of anaemia in hemodialysis patients. Nephrol Dial Transpl 1995; 6: 40–3
McGonigle RJS, Husserl F, Wallin JD, et al. Hemodialysis and continuous ambulatory peritoneal dialysis effects on erythropoiesis in renal failure. Kidney Int 1984; 25: 430–6
Zappacosta AR, Caro J, Erlev A. Normalization of hematocrit in patients with end-stage renal disease on continuous ambulatory peritoneal dialysis. Am J Med 1982; 72: 53–7
Saito A, Suzuki I, Chung TG, et al. Separation of an inhibitor of erythropoiesis in ‘middle molecules’ from hemodialysate from patients with chronic renal failure. Clin Chem 1986; 32: 1938–41
Delano BG, Ifudu O, Joseph M, et al. Endogenous erythropoietin levels are elevated in patients on continuous ambulatory peritoneal dialysis [abstract]. J Am Soc Nephrol 1995; 6: 536
Ateshkadi A, Johnson CA, Oxton LL, et al. Pharmacokinetics of intraperitoneal, intravenous, and subcutaneous recombinant human erythropoietin in patients on continuous ambulatory peritoneal dialysis. Am J Kidney Dis 1993; 21: 635–42
MacDougall IC, Roberts DE, Neubert P, et al. Pharmacokinetics of recombinant human erythropoietin in patients on continuous ambulatory peritoneal dialysis. Lancet 1989; 17: 1388–9
Muirhead N, Cattran DC, Zaltzman J, et al. Safety and efficacy of recombinant human erythropoietin in correcting the anaemia of patients with chronic renal allograft dysfunction. J Am Soc Nephrol 1994; 5: 1216–22
Nampoory MR, Johny KV, al-Hilali N, et al. Erythropoietin deficiency and relative resistance cause anaemia in post-renal transplant recipients with normal renal function. Nephrol Dial Transplant 1996; 11: 177–81
Miles AM, Markell MS, Daskalakis P, et al. Anemia following renal transplantation: erythropoietin response and iron deficiency. Clin Transplant 1997; 11: 313–5
Van Loo A, Vanholder R, Bernaert P, et al. Recombinant human erythropoietin corrects anaemia during the first weeks after renal transplantation: a randomized prospective study. Nephrol Dial Transplant 1996; 11: 1815–21.
Almond MK, Tailor D, Marsh FP, et al. Increased erythropoietin requirements in patients with failed renal transplants returning to a dialysis programme. Nephrol Dial Transplant 1994; 9: 270–3
Sepandj F, Jindal K, West M, et al. Economic appraisal of maintenance parenteral iron administration in treatment of anaemia in chronic hemodialysis patients. Nephrol Dial Transplant 1996; 11: 319–22
Fishbane S, Ungureanu V-D, Maesaka J, et al. The safety of intravenous iron dextran. Am J Kidney Dis 1996; 28: 529–34.
Kalantar-Zadeh K, Hoffken B, Wunsch H, et al. Diagnosis of iron deficiency anaemia in renal failure patients during the post-erythropoietin era. Am J Kidney Dis 1995; 26: 292–9
Kooistra MP, van Es A, Struyvenberg A, et al. Iron metabolism in patients with the anaemia of end-stage renal disease during treatment with recombinant human erythropoietin. Br J Haem 1991; 79: 634–9
Baldus M, Salopek S, Moller M, et al. Experience with zinc protoporphyrin as a marker of endogenous iron availability in chronic haemodialysis. Nephrol Dial Transpl 1996; 11: 486–91
Fairbanks VF, Beutler E. Iron deficiency. In: Beutler E, Licht-man MA, Coller BS, et al., editors. Williams Hematology. New York: McGraw-Hill Inc., 1995: 490–509
Goch J, Birgegard G, Danielson BG, et al. Iron absorption in patients with chronic uremia on maintenance hemodialysis and in healthy volunteers measured with a simple oral iron load test. Nephron 1996; 73: 403–6
Grimes AJ, Hutt MSR. Metabolism of 59 Fe-dextran complex in human subjects. BMJ 1957; 8: 1074–7
Danielson BG, Salmonson T, Derendorf H, et al. Pharmacokinetics of iron(III)-hydroxidesucrose complex after a single intravenous dose in healthy volunteers. Arzneimittel-Forschung 1996; 46: 615–21
Reece WD, Brackelsberg PO, Hotchkiss DK. Erythrocyte changes, serum iron concentration and performance following iron injection in neonatal beef calves. J Animal Science 1985; 61: 1387–94
Henderson PA, Hillman RS. Characteristics of iron dextran utilization in man. Blood 1969; 34: 357–75
Becker CE, MacGregor RR, Walker KS, et al. Fatal anaphylaxis after intramuscular iron-dextran. Ann Intern Med 1966; 65: 745–8
Fleming LW, Hopwood D, Shepherd AN, et al. Hepatic iron in dialyzed patients given intravenous iron dextran. J Clin Path 1990; 43: 119–24
Vanholder R, Ringoir S. Infectious morbidity and defects of phagocytic function in end-stage renal disease: a review. J Am Soc Nephrol 1993; 3: 1541–54
Collins A, Ebben J, Ma J. Frequent intravenous iron dosing is associated with higher infectious deaths [Abstract]. J Am Soc Nephrol 1997; 8: 190A
Stevens RG, Jones DY, Micozzi MS, et al. Body iron stores and the risk of cancer. N Engl J Med 1988; 319: 1047–52.
Sempos CT, Looker AC, Gillum RF, et al. Body iron stores and the risk of coronary heart disease. N Engl J Med 1994: 330: 1119–24
Murray MJ, Stein N. The effect of injected iron on the absorption of iron in iron deficiency. Br J Haem 1972; 23: 13–6
Madore F, Lowrie E, Brugnara C, et al. Anaemia in hemodialysis patients: variables affecting this outcome predictor. J Am Soc Nephrol 1997; 8: 1921–9
Ifudu O, Matthew JJ, Mayers JD, et al. Severity of acquired immunodeficiency syndrome and the response to erythropoietin in uremia. Am J Kidney Dis 1997; 30: 28–35
Ifudu O, Feldman J, Friedman EA. The intensity of hemodialysis and the response to erythropoietin in patients with endstage renal disease. N Engl J Med 1996; 334: 420–5
Opatrna’ S, Opatrny’ Jr K, Cejkova’ P, et al. Relationship between anaemia and adequacy of continuous ambulatory peritoneal dialysis. Nephron 1997; 77: 359–60
Tzamaloukas AH, Murata GH, Sena P. When do plasma levels of azotemic indices indicate inadequacy of peritoneal dialysis? Nephron 1994; 67: 495–6
Macdougall IC, Allen D, Tucker B, et al. Serum interleukin-6 levels are a useful indicator of marrow suppression in patients with resistance to erythropoietin due to inflammatory disease [abstract]. J Am Soc Nephrol 1993; 4: 428
Ifudu O, Fowler A, Friedman EA. Hepatitis B virus infection does not abolish response to erythropoietin in end-stage renal disease [Abstract]. ASAIO Journal 1997; 43: 76
Ifudu O, Valcourt JC, Friedman EA. Sepsis provokes an acute drop in hematocrit in erythropoietin-treated hemodialysis patients [abstract]. J Am Soc Nephrol 1997; 8: 238A
Rao SD, Shih M, Mohini R. Effect of serum parathyroid hormone and bone marrow fibrosis on the response to erythropoietin in uremia. N Engl J Med 1993; 328: 171–5
Ifudu O, Homel P. Effect of serum parathyroid hormone and aluminum levels on the response to erythropoietin in uremia. Nephron 1998; 79: 477–8
Raine AEG, Roger SD. Effects of erythropoietin on blood pressure. Am J Kidney Dis 1991; 18: 76–83
Ifudu O, Dawood M, Homel P. Erythropoietin-induced elevation in blood pressure is immediate and dose-dependent. Nephron 1998; 79: 486–7
Eschbach JW, Kelly MR, Haley NR, et al. Treatment of the anaemia of progressive renal failure with recombinant human erythropoietin. N Engl J Med 1989; 321: 158–63
Nagai T, Akizawa T, Kohjiro S, et al. RhuEPO enhances the production of plasminogen activator inhibitor-1in culture endothelial cells. Kidney Inter 1996; 49: 102–7
De Marchi S, Cecchin E, Falleti E, et al. Long-term effects of erythropoietin therapy on fistula stenosis and plasma concentrations of PDGF and MCP-1 in hemodialysis patients. J Am Soc Nephrol 1997; 8: 1147–56
Goldwasser P, Avram MM, Collier JT, et al. Correlates of vascular access occlusion in hemodialysis. Am J Kidney Dis 1994; 24: 785–94
Churchill DN, Muirhead N, Goldstein M, et al. Probability of thrombosis of vascular access among hemodialysis patients treated with recombinant human erythropoietin. J Am Soc Nephrol 1994; 4: 1809–13
Ifudu O, Friedman EA. Economic implications of inadequate response to erythropoietin in end-stage renal disease. Dialysis Transpl 1997; 26: 664–98
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ifudu, O. Optimal Use of Epoetin-α in the Anaemia of Chronic Renal Failure. BioDrugs 11, 175–183 (1999). https://doi.org/10.2165/00063030-199911030-00004
Published:
Issue Date:
DOI: https://doi.org/10.2165/00063030-199911030-00004