Abstract
Multiple sclerosis (MS), a chronic inflammatory demyelinating disease of the central nervous system, is the most common crippling neurological disease of young adults in the US. The 2 basic clinical forms of the disease (relapsing and progressive), which can occur singly or in combination, encompass a wide range of clinical severities are usually established between 18 and 35 years of age and can persist an entire lifetime. Life expectancy of 20 years is 85% of normal.
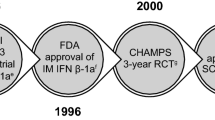
Historically, the standard proven and generally accepted clinical treatment of the disease has been corticotropin (ACTH) and methylprednisolone, which benefited clinical relapses but had no effect on clinical disability (the most important factor influencing the lives of individual MS patients) or other aspects of the chronic course of the disease. The most important new development in the treatment of MS has been the introduction of interferon beta into the clinic. Two forms of recombinant interferon beta have been approved by the FDA for use in relapsing MS: interferon beta-1b (IFN-β-1b) and interferon beta-1a (IFN-β-1a). The efficacy of IFN-β-1b in the treatment of relapsing-remitting MS was established first but no effect on physical disability progression was discerned.
In contrast, well designed trials of intramuscular IFN-β-1a (Avonex®) 6.0 MIU (30μ) weekly and subcutaneous IFN-α-1a (Rebif®) 6 MIU (22μg) or 12 MIU (44μg) 3 times weekly produced a significant delay in the time to sustained progression in physical disability, the first MS treatment to exert such a prophylactic effect. Additionally, IFN-β-1a significantly reduced clinical relapses and acute and chronic brain lesions revealed by MRI examinations. It is currently believed that IFN-β-1a treatment alters the fundamental course of relapsing MS.
The mechanisms of the therapeutic benefit of recombinant interferon betas are incompletely understood but may include augmentation of suppressor T cell function, inhibition of interferon gamma actions, inhibition of T cell activation, or induction of interleukin-10 gene transcription.
Similar content being viewed by others
References
Hohlfeld R. Biotechnological agents for the immunotherapy of multiple sclerosis. Principles, problems and perspectives. Brain 1997; 120: 865–916
Dhib-Jalbut S, McFarlin DE. Immunology of multiple sclerosis. Ann Allergy 1990; 64: 433–44
Julien J, Ferrer X. Multiple sclerosis: an overview. Biomed Pharmacother 1989; 43: 335–46
Traggs BD, Peterson J, Rarnsohoff RM, et al. Axonal transection in the lesions of multiple sclerosis. N Engl J Med 1998; 338: 278–85
Becker CC, Gidal BE, Fleming JO. Immunotherapy in multiple sclerosis. Pt 1. Am J Health Syst Pharm 1995; 52: 1985–2000
Hartung H-P. Immune-mediated demyelination. Ann Neurol 1993; 33: 563–7
Lucchinetti CF, Brück W, Rodriguez M, et al. Distinct patterns of multiple sclerosis pathology indicates heterogeneity in pathogenesis [review]. Brain Pathol 1996; 6: 259–74
Sercarz EE, Lehmann PV, Ametani A, et al. Dominance and crypticity of T cell antigenic determinants [review]. Annu Rev Immunol 1993; 11: 729–66
Miller SD, McRae BL, Vanderlugt CL, et al. Evolution of the T-cell repertoire during the course of experimental immune-mediated demyelinating diseases [review]. Immunol Rev 1995 a; 144: 225–44
Genain CP, Nguyen M-H, Letvin NL, et al. Antibody facilitation of multiple sclerosis-like lesions in a nonhuman primate. J Clin Invest 1995 a; 96: 2966–74
Wekerle H, Kojima K, Lannes-Vieira J, et al. Animal models [review]. Ann Neurol 1994; 36 Suppl.: S47–53
Cutler RWP. Demyelinating disease. In: Dale DC, Federman DD, Cassel CK, et al., editors. Scientific American medicine. New York, NY: Scientific American Inc., 1993; sect 11, chap IX: 1-8
Minden SL, Marder WD, Harrold LN, et al. Multiple sclerosis: a statistical portrait. Cambridge, (MA): ABT Associates, Inc., 1993
Weinshenker BG. Natural history of multiple sclerosis. Ann Neurol 1994; 36: S–6–11
Mathews WB. Course and prognosis. In: Mathews WB, Compston A, Allen IV, et al., editors. McAlpine’s multiple sclerosis. 2nd ed. New York, NY: Churchill Livingstone, 1991: 139–63
Lublin FD, Reingold SC. Defining the clinical course of multiple sclerosis. Neurology 1996; 46: 907–11
Kinkel RP, Rudick RA. Multiple sclerosis. In: Conn HF, editor. Current therapy. Philadelphia, (PA): WB Saunders Company, 1996: 883–93
Martyn CN. The epidemiology of multiple sclerosis. In: Mathews WB, Compston A, Allen IV, et al., editors. McAlpine’s multiple sclerosis. 2nd ed. New York, NY: Churchill Livingstone, 1991: 3–40
Compston A. Genetic epidemiology of multiple sclerosis. J Neurol Neurosurg Psychiat 1997; 62: 553–61
Kurtzke JF. Epidemiology of multiple sclerosis. In: Hallpike JF, Adams CW, Tourtellotte WW, editors. Multiple sclerosis. Baltimore, (MD): Williams & Wilkins, 1983: 47–96
Calder V, Owen S, Watson C, et al. MS: a localized immune disease of the central nervous system. In: Kelly J, Gallagher R, editors. Immunology today. New York, NY: Elsevier Science Publishers Ltd, 1989; 10: 99–103
Jacobs L, Goodkin DE, Rudick RA, et al. Advances in specific therapy for multiple sclerosis. Curr Opin Neurol 1994; 7: 250–4
Rose AS, Kuzma JW, Kurtzke JF, et al. Cooperative study in the evaluation of therapy in multiple sclerosis: ACTH vs placebo: final report. Neurology 1970; 20: 1–159
Myers LW. Treatment of multiple sclerosis with ACTH and corticosteroids. In: Rudick RA, Goodkin DE, editors. Treatment of multiple sclerosis: trial design, results and future perspectives. London, England: Springer-Verlag, 1992: 135
Jacobs LD, Brownscheidle CM. Making strides in therapy for multiple sclerosis. Contemp Int Med 1997; 9: 33–40
Miller H, Newell DJ, Ridley A. Multiple sclerosis: treatment of acute exacerbations with corticotropin (ACTH). Lancet 1961; II: 1120–22
Durelli L, Cocito D, Riccio A, et al. High-dose intravenous methylprednisolone in the treatment of multiple sclerosis: clinical immunologic correlations. Neurology 1986; 36: 238
Milligan NM, Newcombe R, Compston DA. A double-blind controlled trial of high-dose methylprednisolone in patients with multiple sclerosis. I. clinical effects. J Neurol Neurosurg Psychiat 1987; 50: 511
Hughes RAC. Use of azathioprine in multiple schlerosis. In: Cazzullo CL. Caputo D, Ghezzi A, et al., editors. Virology and immunology in multile schlerosis: rationale for therapy. Berlin: Springer-Verlag, 1988: 153–61
Goodkin DE, Rudick RA, Vanderbrug Medendorp S, et al. Low-dose (7.5mg) oral methotrexate reduces the rate of progression in chronic progressive multiple sclerosis. Ann Neurol 1995; 37: 30–41
Jacobs L, O’Malley J, Freeman A, et al. Intrathecal interferon reduces exacerbations of multiple sclerosis. Science 1981; 214: 1026–8
Jacobs L, Salazar AM, Herndon RM, et al. Multicenter doubleblind study of effect of intrathecally administered natural human fibroblast interferon on exacerbations of multiple sclerosis. Lancet 1986; II: 1411–3
Jacobs LD, Cookfair DL, Rudick RA, et al. Intramuscular interferon beta-1a for disease progression in relapsing multiple sclerosis. Ann Neurol 1996; 39: 285–94
The IFN-β Multiple Sclerosis Study Group. Interferon beta-1b is effective in relapsing-remitting multiple sclerosis. I. Clinical results of a multicenter, randomized, double-blind, placebo-controlled trial. Neurology 1993; 43: 655–61
Paty DB, Li DKB, the UBC MS/MRI Study Group, et al. Interferon beta-1b is effective in relapsing-remitting multiple sclerosis. II. MRI analysis results of a multicenter, randomized, double-blind, placebo-controlled trial. Neurology 1993; 43: 662–7
The IFN-β Multiple Sclerosis Study Group and the University of British Columbia MS/MRI Analysis Group. Interferon beta-1b in the treatment of multiple sclerosis: final outcome of the randomized controlled trial. Neurology 1995; 45: 1277–85
Rudick RA, Goodkin DE, Jacobs LD, et al. Impact of interferon beta-1a on neurologic disability in relapsing multiple sclerosis. Neurology 1997; 49: 358–63
National Multiple Sclerosis Society. Research Programs Department News. Positive results announced for interferon beta-1a (Rebif®, Ares-Serono) for relapsing-remitting MS; 1997 Oct 3; News No. RP01-98
Rudick RA, Simonian NA, Alam JD, et al. Incidence and significance of neutralizing antibodies to interferon beta-1a in multiple sclerosis. Neurology 1998; 50: 1266–72
The IFN-β Multiple Sclerosis Study Group and the University of British Columbia MS/MRI Analysis Group. Neutralizing antibodies during treatment of multiple sclerosis with interferon beta-1b: experience during the first three years. Neurology 1996; 47: 889–94
Hochuli E. Interferon immunogenecity: technical evaluation of interferon-alpha-2a. J Interferon Cytokine Res 1997; 17: S15–S21
Wang C, Eufemi M, Turano C, et al. Influence of the carbohydrate moiety on the stability of glycoproteins. Biochemistry 1996; 35: 7299–307
Keck K. Ir gene control of carrier recognition. III. Cooperative recognition of two or more carrier determinants on insulins of different species. Eur J Immunol 1997; 7: 811–6
Rosenwasser LJ, Barcinski MA, Schwartz RH, et al. Immune response gene control of determinant selection. II. Genetic control of the murine T lymphocyte proliferative response to insulin. J Immunol 1979; 123: 471–6
Cohen IR, Talmon J. H-2 genetic control of the response of T lymphocytes to insulins. Priming of nonresponder mice by forbidden variants of specific antigenic determinants. Eur J Immunol 1980; 10: 284–9
Holliday SM, Benfield P. Interferon-β-1a: a review of its pharmacological properties and therapeutic potential in multiple sclerosis. BioDrugs 1997; 4: 317–30
Avonex™ (Interferon beta-1a) IM injection. Product Monograph. 1996, Biogen, Inc.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jacobs, L., Brownscheidle, C.M. Appropriate Use of Interferon Beta-1a in Multiple Sclerosis. BioDrugs 11, 155–163 (1999). https://doi.org/10.2165/00063030-199911030-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00063030-199911030-00002