Abstract
Objective: To assess the effect of the proton pump inhibitor pantoprazole on the pharmacokinetics of ethanol.
Study Participants: Nonalcoholic, healthy volunteers (four female, 12 male, age range 23 to 30 years).
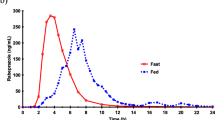
Study Design and Methods: This was a placebo-controlled, double-blind, randomised, crossover study. Participants received placebo and pantoprazole 40mg, respectively, for 7 days each, separated by a 14-day washout period. On day 7 of each period, a moderate dose of ethanol (0.5 g/kg) was consumed 2 hours after administration of the study medication. Blood ethanol levels were measured over a period of 8 hours and the following pharmacokinetic parameters determined: (i) area under the blood ethanol concentration versus time curve (AUC), (ii) maximum concentration of ethanol in blood (Cmax), and (iii) elimination rate of ethanol (kel).
Results: The mean values for the three pharmacokinetic parameters of ethanol were similar during the pantoprazole and placebo study periods: AUC 112.7 vs 111.3 mg/dl·h, Cmax 43.5 versus 42.7 mg/dl and kel 12.3 versus 12.5 dl/h. The 90% confidence intervals for the ratio of these values (ethanol + pantoprazole : ethanol + placebo) were completely within the predefined equivalence range.
Conclusions: A therapeutic dose of pantoprazole 40mg did not alter the absorption or the elimination rate of a moderate dose of ethanol in healthy individuals. Thus, it would be expected that in patients with gastric acid-related diseases such as peptic ulcer or gastro-oesophageal reflux disease, pantoprazole therapy would not cause elevation of blood ethanol concentration after moderate social drinking.




Similar content being viewed by others
References
Sanders SW. Pathogenesis and treatment of acid peptic disorders: comparison of proton pump inhibitors with other antiulcer agents. Clin Ther 1996; 18: 2–34
Ollyo J-B, Monnier P, Fontolliet C, et al. The natural history, prevalence and incidence of reflux oesophagitis. Gullet 1993; 3 Suppl.: 3–10
Petersen H. The prevalence of gastro-oesophageal reflux disease. Scand J Gastroenterol 1995; 211 Suppl.: 5–6
Penston JG, Pounder RE. A survey of dyspepsia in Great Britain. Aliment Pharmacol Ther 1996; 10: 83–9
Fennerty MB, Lieberman D. H2-receptor antagonists in the treatment of complicated gastroesophageal reflux disease: ‘for whom the bell tolls’. Gastroenterology 1994; 107: 1545–8
Richardson P, Hawkey CJ, Stack WA. Proton pump inhibitors. Pharmacology and rationale for use in gastrointestinal disorders. Drugs 1998; 56: 307–35
Moss SF, Arnold R, Tytgat GNJ, et al. Consensus statement for management of gastroesophageal reflux disease. J Clin Gastroenterol 1998; 27: 6–12
Lieber CS. Medical disorders of alcoholism. N Engl J Med 1995; 333: 1058–65
Bongers IM, van de Goor I, Garretsen HF. Social climate on alcohol in Rotterdam, The Netherlands: public opinion on drinking behaviour and alcohol control measures. Alcohol Alcohol 1998; 33: 141–50
Caballeria J, Baraona E, Rodamilans M, et al. Effects of cimetidine on gastric alcohol dehydrogenase activity and blood ethanol levels. Gastroenterology 1989; 96: 388–92
Frezza M, DiPadova C, Pozzato G, et al. High blood alcohol levels in women. The role of decreased gastric alcohol dehydrogenase activity and first-pass metabolism. N Engl J Med 1990; 322: 95–9
Hernandez MR, Caballeria J, Baraona E, et al. Human gastric alcohol dehydrogenase: its inhibition by H2-receptor antagonists, and its effect on the bioavailability of ethanol. Alcohol Clin Exp Res 1990; 14: 946–50
Caballeria J, Baraona E, Deulofeu R, et al. Effects of H2-receptor antagonists on gastric alcohol dehydrogenase activity. Dig Dis Sci 1991; 36: 1673–9
Palmer RH, Frank WO, Nambi P, et al. Effects of various concomitant medications on gastric alcohol dehydrogenase and the first-pass metabolism of ethanol. Am J Gastroenterol 1991; 86: 1749–55
DiPadova C, Roine R, Frezza M, et al. Effects of ranitidine on blood alcohol levels after ethanol ingestion. Comparison with other H2-receptor antagonists. JAMA 1992; 267: 83–6
Mallat A, Roudot TF, Bergmann JF, et al. Inhibition of gastric alcohol dehydrogenase activity by histamine H2-receptor antagonists has no influence on the pharmacokinetics of ethanol after a moderate dose. Br J Clin Pharmacol 1994; 37: 208–11
Teyssen S, Chari ST, Joos A, et al. Effect of a 28-day therapy with famotidine on blood levels of alcohol and gastrin and intragastric pH in healthy human subjects. Scand J Gastroenterol 1994; 29: 398–405
Girre C, Coutelle C, David P, et al. Lack of effect of lansoprazole on the pharmacokinetics of ethanol in male volunteers. Gastroenterology 1994; 106Suppl. 4: A504
Minocha A, Rahal PS, Brier ME, et al. Omeprazole therapy does not affect pharmacokinetics of orally administered ethanol in healthy male subjects. J Clin Gastroenterol 1995; 21: 107–9
Battiston L, Tulissi P, Moretti M, et al. Lansoprazole and ethanol metabolism: comparison with omeprazole and cimetidine. Pharmacol Toxicol 1997; 81: 247–52
Fitton A, Wiseman L. Pantoprazole. A review of its pharmacological properties and therapeutic use in acid-related disorders. Drugs 1996; 51: 460–82
Dettmer A, Vogt R, Sielaff F, et al. Low-dose pantoprazole (20 mg) is effective for relief of symptoms and healing of lesions in mild reflux esophagitis. A randomised, double-blind, parallel and multi-centre study. Aliment Pharmacol Ther 1998; 12: 865–72
Escourrou J, Deprez P, Saggioro A, et al. Maintenance therapy with pantoprazole 20 mg prevents relapse of reflux oesophagitis. Aliment Pharmacol Ther 1999; 13: 1481–91
Bardhan KD. Pantoprazole: a new proton pump inhibitor in the management of upper gastrointestinal disease. Drugs of Today 1999; 35: 773–808
Steinijans VW, Hartmann M, Huber R, et al. Lack of pharmacokinetic interaction as an equivalence problem. Int J Clin Pharmacol Ther Toxicol 1992; 30Suppl. 1: S63–8
Diletti E, Hauschke D, Steinijans VW. Sample size determination for bioequivalence assessment by means of confidence intervals. Int J Clin Pharmacol Ther Toxicol 1992; 30Suppl. 1: S51–8
Steinijans VW, Hauschke D. International harmonization of regulatory requirements for average bioequivalence and current issues in individual bioequivalence. Drug Inf J 1995; 29: 1055–62
Amir I, Anwar N, Baraona E, et al. Ranitidine increases the bioavailability of imbibed alcohol by accelerating gastric emptying. Life Sci 1996; 58: 511–8
Poli E, Coruzzi G, Bertaccini G. Ranitidine but not famotidine releases acetylcholine from the guinea pig myenteric plexus. Agents Actions 1990; 30: 191–4
Haber PS, Gentry RT, Mak KM, et al. Metabolism of alcohol by human gastric cells: relation to first-pass metabolism. Gastroenterology 1996; 111: 863–70
Gupta AM, Baraona E, Lieber CS. Significant increase of blood alcohol by cimetidine after repetitive drinking of small alcohol doses. Alcohol Clin Exp Res 1995; 19: 1083–7
Simon B, Müller P, Marinis E, et al. Effect of repeated oral administration of BY1023/SK&F96022 — a new substituted benzimidazole derivative — on pentagastrin-stimulated gastric acid secretion and pharmacokinetics in man. Aliment Pharmacol Ther 1990; 4: 373–9
Toon S, Khan AZ, Holt BI, et al. Absence of effect of ranitidine on blood alcohol concentrations when taken morning, midday, or evening with or without food. Clin Pharmacol Ther 1994; 55: 385–91
Steinijans VW, Huber R, Hartmann M, et al. Lack of pantoprazole drug interactions in man: an updated review. Int J Clin Pharmacol Ther 1996; 34: S31–50
Herberg KW, Hartmann M, Neukirchen B, et al. Pantoprazole does not affect performance in traffic-related safety tests: a double blind, randomised, placebo-controlled, crossover study in healthy volunteers. Clin Drug Invest 1998; 16: 63–70
Acknowledgements
The authors thank Dr Kathy B. Thomas, Byk Gulden, Konstanz, Germany for useful suggestions during the preparation and editing of the manuscript. This study was supported by a grant from Byk Gulden, Konstanz, Germany.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Heinze, H., Fischer, R., Pfützer, R. et al. Lack of Interaction between Pantoprazole and Ethanol. Clin. Drug Investig. 21, 345–351 (2001). https://doi.org/10.2165/00044011-200121050-00004
Published:
Issue Date:
DOI: https://doi.org/10.2165/00044011-200121050-00004