Abstract
Objectives
To characterise the peritoneal and subcutaneous adipose penetration of alatrofloxacin. If the extent of penetration of this lipophilic fluoroquinolone is adequate in patients with extensive adipose layers, it may provide better antimicrobial coverage than more commonly used antibiotics that are less lipophilic.
Study Participants and Methods
Six morbidly obese individuals undergoing a Roux-Y gastric bypass procedure received single 1-hour infusions of alatrofloxacin equivalent to 300mg of its active metabolite, trovafloxacin. Blood samples were obtained over a 24-hour period and adipose tissue from subcutaneous and deep tissue sites were obtained approximately 3 hours post-infusion of alatrofloxacin. Plasma and adipose tissue concentrations of trovafloxacin were determined by high pressure liquid chromatography with fluorescence detection.
Results
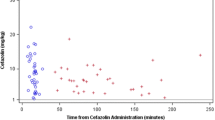
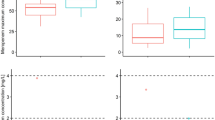
The mean maximum plasma concentration, area under the concentration-time curve, and elimination half-life of trovafloxacin were 3.6 mg/L, 37.4 mg/L·h, and 12.1h, respectively. The mean tissue concentrations at the subcutaneous and deep adipose sites were 0.43 and 0.41 μ/g, respectively.
Conclusions
These results indicated that the pharmacokinetics of trovafloxacin in morbidly obese individuals are similar to those in healthy control individuals. In addition, the concentrations of trovafloxacin achieved in the adipose tissue were above the minimum inhibitory concentration of most pathogens responsible for surgical and decubitus ulcer infections.


Similar content being viewed by others
References
Product information. Trovan® (trovafloxacin). New York, NY: Pfizer. 1998
Teng R, Vincent J, Baris B, et al. Pharmacokinetics of single and multiple doses of alatrofloxacin, the prodrug of CP-99,219, in healthy male volunteers [abstract F240]. In: Program and Abstracts of the Thirty-Fifth Interscience Conference on Antimicrobial Agents and Chemotherapy, San Francisco, CA. Washington, DC: American Society for Microbiology: 1995; 155
Brightly KE, Gootz TD. The chemistry and biological profile of trovafloxacin. J Antimicrob Chemother 1997; 39Suppl. B: 1–14
Nagachinta T, Stephens M, Reitz B, et al. Risk factors for surgical wound infection following cardiac surgery. J Infect Dis 1987; 156: 967–73
Nystrom PO, Jonstam A, Hojer H, et al. Incisional infection after colorectal surgery in obese patients. Acta Chir Scand 1987; 153: 225–7
Vincent J, Venitz J, Teng R, et al. Pharmacokinetics and safety of trovafloxacin in healthy male volunteers following administration of single intravenous doses of prodrug, alatrofloxacin. J Antimicrob Chemother 1997; 39Suppl. B: 75–80
Cockcroft DW, Gault MW. Prediction of creatnine clearance from serum creatnine. Nephron 1976; 16: 31–41
Kerndole DS, Kaiser AB. Postoperative infections and antimicrobial prophylaxis. In: Mandell GL, Bennett JE, Dolin R, editors. Mandell, Douglas and Bennett’s Principles and Practice of Infectious Diseases, 4th ed. New York: Churchill Livingstone, 1995: 2742–56
Nichols RL. Surgical infections: prevention and treatment-1965 to 1995. Am J Surg 1996; 172: 68–74
Melnik G, Schwesinger WH, Dogolo LC, et al. Concentrations of trovafloxacin in colonic tissue and peritoneal fluid after intravenous infusion of the prodrug alatrofloxacin in patients undergoing colorectal surgery. Am J Surg 1998; 176Suppl. 6A: 14S–7S
Garey KW, Amsden GW. Trovafloxacin: An Overview. Pharmacotherapy 1999; 19: 21–34
Acknowledgements
This study was supported by an unrestricted educational grant from the US Pharmaceuticals Group of Pfizer, Inc.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pai, M.P., Bordley, J. & Amsden, G.W. Plasma Pharmacokinetics and Tissue Penetration of Alatrofloxacin in Morbidly Obese Individuals. Clin. Drug Investig. 21, 219–224 (2001). https://doi.org/10.2165/00044011-200121030-00008
Published:
Issue Date:
DOI: https://doi.org/10.2165/00044011-200121030-00008