Abstract
Objective
This study aimed to compare the antihypertensive effect and tolerability of candesartan cilexetil, an angiotensin II type 1 (AT1) receptor blocker with a dose-dependent and long-lasting antihypertensive effect, with that of amlodipine, a widely used long-acting dihydropyridine calcium antagonist, and with that of a combination of candesartan cilexetil and amlodipine.
Design
Multicentre, randomised, double-blind, placebo-controlled parallel-group study.
Setting
42 general practice centres in France, Hungary, Poland, South Africa and the United Kingdom.
Patients
341 men and women, aged 21 to 81 years, with primary hypertension and a sitting diastolic blood pressure (DBP) of 95 to 114mm Hg.
Interventions
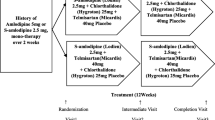
After a 4-week placebo run-in period, patients were randomised to once-daily treatment with candesartan cilexetil 8mg (n = 85), amlodipine 5mg (n = 84), candesartan cilexetil 8mg plus amlodipine 5mg (n = 89), or placebo (n = 83) for 8 weeks.
Main Outcome Measures
Differences between treatments in change in blood pressure from randomisation to the end of the study were evaluated using analysis of covariance.
Results
All active treatment regimens resulted in marked reductions in sitting and standing blood pressures compared with placebo (p < 0.001). Co-administration of candesartan cilexetil and amlodipine resulted in statistically significant and clinically important greater blood pressure reductions than either drug alone, with differences in adjusted mean systolic blood pressure (SBP)/DBP reductions of 5.6/2.0mm Hg (sitting) and 6.7/3.4mm Hg (standing) vs amlodipine; and of 5.8/0.8mm Hg (sitting) and 6.8/3.9mm Hg (standing) vs candesartan cilexetil (p < 0.05 for combination therapy vs both monotherapies, except for sitting DBP; not significant). All treatments were well tolerated.
Conclusions
This study not only confirms the efficacy and tolerability of candesartan cilexetil and amlodipine in patients with primary hypertension, but also shows that their combination results in a clinically important enhancement of antihypertensive effect while maintaining tolerability.



Similar content being viewed by others
References
Joint National Committee. The sixth report of the Joint National Committee on prevention, detection, evaluation, and treatment of high blood pressure. Arch Intern Med 1997; 157: 2413–46
Guidelines Subcommittee. 1999 World Health Organization —International Society of Hypertension Guidelines for the Management of Hypertension. J Hypertens 1999; 17: 151–83
Materson BJ, Reda DJ, Cushman WC. Department of Veterans Affairs single-drug therapy of hypertension study. Revised figures and new data. Department of Veterans Affairs Cooperative Study Group on Antihypertensive Agents. Am J Hypertens 1995; 8: 189–192
Neutel JM, Black HR, Weber MA. Combination therapy with diuretics: an evolution of understanding. Am J Med 1996; 101: 61S–70S
Moser M, Black HR. The role of combination therapy in the treatment of hypertension. Am J Hypertens 1998; 11: 73S–78S
Sever PS. Additional therapy versus monotherapy in the treatment of hypertension. Br J Cardiol 1999; 6: 24–34
Morsing P, Adler G, Brandt-Eliasson U, et al. Mechanistic differences of various AT1-receptor blockers in isolated vessels of different origin. Hypertension 1999; 33: 1406–13
Timmermans PB. Pharmacological properties of angiotensin II receptor antagonists. Can J Cardiol 1999; 15 Suppl. F: 26F–8F
Andersson OK, Neldam S. The antihypertensive effect and tolerability of candesartan cilexetil, a new generation angiotensin II antagonist, in comparison with losartan. Blood Pressure 1998; 7: 53–9
Burris JF, Ames RP, Applegate WB, et al. Double-blind comparison of amlodipine and hydrochlorothiazide in patients with mild to moderate hypertension. J Cardiovasc Pharmacol 1988; 12 Suppl. 7: S98–102
Julius S. Amlodipine in hypertension: an overview of the clinical dossier. J Cardiovasc Pharmacol 1988; 12 Suppl. 7: S27–33
Mroczek WJ, Burris JF, Allenby KS. A double-blind evaluation of the effect of amlodipine on ambulatory blood pressure in hypertensive patients. J Cardiovasc Pharmacol 1988; 12 Suppl. 7: S79–84
Malmqvist K, Kahan T, Dahl M. Angiotensin II type 1 (AT1) receptor blockade in hypertensive women: benefits of candesartan cilexetil versus enalapril and hydrochlorothiazide. Am JHypertens 2000; 13: 504–11
Lacourcière Y, Asmar R, for the Candesartan/Losartan study investigators. A comparison of the efficacy and duration of action of candesartan cilexetil and losartan as assessed by clinic and ambulatory blood pressure after a missed dose, in truly hypertensive patients. A placebo-controlled, forced titration study. Am J Hypertens 1999; 12: 1181–7
Belcher G, Hübner R, George M, et al. Candesartan cilexetil: safety and tolerability in health volunteers and patients with hypertension. J Hum Hypertens 1997; 11 Suppl. 2: S85–9
Bell TP, DeQuattro V, Lasseter KC, et al. for the Candesartan Cilexetil Study Investigators. Effective dose range of candesartan cilexetil for systemic hypertension. Am J Cardiol 1999; 15: 272–5
Philipp T, Letzel H, Arens H-J. Dose-finding study of candesartan cilexetil plus hydrochlorothiazide in patients with mild to moderate hypertension. J Hum Hypertens 1997; 11 Suppl. 2: S67–68
Lüscher TF, Vetter H, Siegenthaler W, et al. Compliance in hypertension: facts and concepts. J Hypertens 1985; 3 Suppl. 1:3–9
Ruschitzka FT, Lüscher TF. Is there a rationale for combining angiotensin-converting enzyme inhibitors and calcium antagonists in cardiovascular disease. Am Heart J 1997; 134: S31–47
Hansson L, Zanchetti A, Carruthers SG, et al. for the HOT Study Group. Effects of intensive blood-pressure lowering and low-dose aspirin in patients with hypertension: principal results of the Hypertension Optimal Treatment (HOT) randomised trial. Lancet 1998; 351: 1755–62
Acknowledgements
This study was supported by Astra Hässle AB, Mölndal, Sweden.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Farsang, C., Kawecka-Jaszcz, K., Langan, J. et al. Antihypertensive Effects and Tolerability of Candesartan Cilexetil Alone and in Combination with Amlodipine. Clin. Drug Investig. 21, 17–23 (2001). https://doi.org/10.2165/00044011-200121010-00003
Published:
Issue Date:
DOI: https://doi.org/10.2165/00044011-200121010-00003