Abstract
Objectives: To assess the tolerability, pharmacodynamic effects and pharmacokinetic parameters after repeated doses of clopidogrel (Plavix®) in patients with moderate or severe renal failure.
Patients: Eight patients with severe renal failure (endogenous creatinine clearance 5 to 15 ml/min) and eight patients with moderate renal impairment (endogenous creatinine clearance 30 to 60 ml/min) were included.
Study Design: An open, uncontrolled, parallel-group study over 8 days’ administration of 75mg once-daily clopidogrel.
Methods: Measurement of changes in ADP-induced platelet aggregation and skin bleeding time and of plasma concentrations and urinary excretion of clopidogrel and its main metabolite, SR 26334. Assessment of clinical tolerance and serial haematological and biochemical investigations.
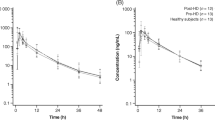
Results: At the end of the dosage period, platelet aggregation was equally inhibited, by about 25%, and bleeding time equally extended, by a factor of about 2, in the two groups. There were no tolerability concerns. Maximum plasma concentration (Cmax) and time to reach Cmax (tmax) for clopidogrel were not significantly different between the two groups. SR 26334 excreted into the urine and renal clearance rate were significantly lower in the severely impaired group, while plasma elimination half-lives were not significantly different. Cmax and tmax did not differ significantly between the two groups, but trough levels and area under the plasma concentration-time curve from zero to 24 hours (AUC0-24h) after the last dose were significantly higher in the moderately impaired group.
Conclusions: Clopidogrel 75mg once daily was well tolerated in patients with either moderate or severe renal failure, and provided good inhibition of ADP-induced platelet aggregation without excessive extension of bleeding time. Dose adjustment in such patients does not appear to be required.
Similar content being viewed by others
References
Harter HR, Burch JW, Majerus PW, et al. Prevention of thrombosis in patients on haemodialysis by low-dose aspirin. N Engl J Med 1979; 301: 577–9
Raine AEG. Hypertension, blood viscosity and cardiovascular morbidity in renal failure: implications of erythropoietin therapy. Lancet 1988; I: 97–100
Livio M, Benigni A, Begnis R, et al. Differential inhibition of platelet thromboxane and renal prostaglandins in the rat. J Pharmacol Exp Ther 1989; 248: 334–41
Steiner RW, Coggins C, Carvalho ACA. Bleeding time in uremia: a useful test to assess clinical bleeding. Am J Hematol 1979; 7: 107–17
di Minno G, Martinez J, McKean M-L, et al. Platelet dysfunction in uremia: multifaceted defect partially corrected by dialysis. Am J Med 1985; 79: 552–9
Gaspari F, Vigano G, Orisio S, et al. Aspirin prolongs bleeding time in uraemia by a mechanism distinct from platelet cyclo-oxygenease inhibition. J Clin Invest 1987; 79: 1788–95
Livio M, Benigni A, Vigano G, et al. Moderate dose of aspirin and risk of bleeding in renal failure. Lancet 1986; I: 414–6
Tabuchi N, Huet RCG, Sturk A, et al. Aprotinin preserves hemostasis in aspirin-treated patients undergoing cardiopulmonary bypass. Ann Thorac Surg 1994; 58: 1036–9
Herbert J-M, Frehel D, Vallée E, et al. Clopidogrel, a novel antiplatelet and antithrombotic agent. Cardiovasc Drug Rev 1993; 11: 180–98
Deray G, Brouard R, Bagnis C, et al. Safety and activity of clopidogrel, an antiplatelet agent, in chronic renal failure patients [abstract]. J Am Soc Nephrol 1996; 6: 384
Lowe GDO, Rumley A, Griffiths P, et al. Dose-dependent inhibition of platelet aggregation in healthy volunteers by a new agent, SR 25990c. Thromb Haemost 1989; 62: 414
Caplain H, Kieffer G, Thiercelin JF, et al. Tolerance and clinical pharmacology of repeated administration of clopidogrel (SR 25990c), a new antiplatelet agent, at three dose levels in normal healthy volunteers. Thromb Haemost 1989; 62: 410
Pharmacodynamics and pharmacokinetics of the novel anti-platelet agent, clopidogrel, in the young and the elderly with and without symptomatic atherosclerosis [abstract P916]. In: Guillin M-C, Bonnet G, Sissmann J, et al., editors. Eur Heart J 17: Abstract Supplement for the XVIIIth Congress of the European Society of Cardiology, Birmingham, UK, 25–29 August 1996; 161
CAPRIE Steering Committee. A randomised, blinded, trial of clopidogrel versus aspirin in patients at risk of ischaemic events (CAPRIE). Lancet 1996; 348: 1329–39
Born GVR. Aggregation of blood platelets by adenosine diphosphate and its reversal. Nature 1962; 194: 927–9
Cockcroft DW, Gault MH. Prediction of creatinine clearance from serum creatinine. Nephron 1976; 16: 31–41
Thébault JJ, et al. Mesure du temps de saignement par la méthode d’Ivy-Nelson: un moyen de surveillance des thérapeutiques antiaggrégantes. Feuillets Biol 1979; 20: 41–2
Hass WK, Easton JD, Adams HP, et al. for the Ticlopidine Aspirin Stroke Study Group. A randomised trial comparing ticlopidine hydrochloride with aspirin for the prevention of stroke in high-risk patients (TASS). N Engl J Med 1989; 321: 501–7
Roald HE, Barstad RM, Kieruff P, et al. Clopidogrel —a platelet inhibitor which inhibits thrombogenesis in non-anticoagulated human blood independently of the flow conditions. Thromb Haemost 1994; 71: 655–62
Clopidogrel compatibility with cardiac co-medication: studies of interactions with a beta-blocker, a calcium uptake antagonist and a cardiac glycoside. In: Cariou R, Peeters PAM, Forbes CD, et al., editors. Clin Pharmacol. In press
Necciari J. 1996. Internal report
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Deray, G., Bagnis, C., Brouard, R. et al. Clopidogrel Activities in Patients with Renal Function Impairment. Clin. Drug Investig. 16, 319–328 (1998). https://doi.org/10.2165/00044011-199816040-00006
Published:
Issue Date:
DOI: https://doi.org/10.2165/00044011-199816040-00006