Summary
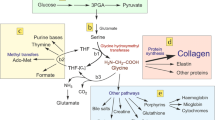
In terms of their effects on matrix synthesis by the indigenous chondrocytes of human articular cartilage, it has been suggested that NSAIDs fall into three categories: those exemplified by aceclofenac and tenidap that are capable of stimulating matrix synthesis, whilst others, which include piroxicam, aspirin and nabumetone, do not appear to have a major effect on matrix synthesis, and a third group, which includes ibuprofen, indomethacin, nimesulide and naproxen, that are markedly inhibitory in terms of matrix synthesis. Whilst the inhibitory effect of certain NSAIDs can be ascribed to toxic effects of the compounds or their metabolites, it has been difficult to understand the mechanism whereby some NSAIDs could stimulate matrix synthetic activity. This paper demonstrates the sensitivity of human cartilage synthetic activity to inhibition by the cytokine interleukin (IL)-1 and confirms that glycosaminoglycan synthesis is responsive to growth factors such as insulin-like growth factor (IGF)-1. The present work showed that the stimulatory effect of NSAIDs does not take place in the absence of serum. The serum, however, may be replaced by IGF-1 at 100 µg/L. In the presence of this growth factor, the inhibition by IL-1 may be partially reversed in the presence of aceclofenac. The recovery from an episode of IL-1 is also increased in the presence of aceclofenac. These experiments lead to the hypothesis that the stimulatory action of NSAIDs is due to the inhibition of locally produced IL-1 and the consequent expression of growth factor activity. Other NSAIDs that may also inhibit IL-1 synthesis or release probably do not have a marked effect as they have toxic effects on cartilage metabolism. The dose curve of tenidap is probably an expression of the balance between stimulation at low concentrations (by allowing growth factor activity to function), and inhibition due to toxic effects at higher concentrations. Aceclofenac does not appear to be toxic at the highest plasma concentrations, although there is a peak of activity around 2 mg/L. These experiments lead to the suggestion that NSAIDs such as aceclofenac would be appropriate for the long-term treatment of arthritic conditions provided that one is prepared to extrapolate between in vitro experiments on human cartilage and what may be happening in vivo.
Similar content being viewed by others
References
Dingle JT. Cartilage maintenance in osteoarthritis: interactions of cytokines, NSAID and prostaglandins in articular cartilage damage and repair. J Rheum 1991; 18 Suppl. 28: 30–7
Dingle JT. Cartilage damage and repair: the roles of IL1, NSAIDs and prostaglandins in osteoarthritis. In: New frontiers in prostaglandin therapeutics. Princetown, USA: Excerpa Medica, 1990: 1–15
Tyler JA. Insulin-like growth factor 1 can decrease degradation and promote synthesis of proteoglycan in cartilage exposed to cytokines. Biochem J 1989; 260: 543–8
Dingle JT. Prostaglandins in human cartilage metabolism. J Lipid Mediat 1993; 6: 303–12
Verbruggen G, Veys EM, Malfait A, et al. Proteoglycan metabolism in tissue cultured human articular cartilage: influence of piroxicam. J Rheumatol 1989; 16(3): 355–61
Rashad S, Revell P, Hemingway A, et al. Effect of non-steroid anti-inflammatory drugs on the course of osteoarthritis. Lancet 1989; 11: 519–22
Huskisson EC, Berry H, Gishen P, et al. On behalf of the LINK Study group. Effects of anti-inflammatory drugs on the progression of osteoarthritis of the knee. J Rheumatol 1995; 22: 1941–6
McKenzie LS, Horsburgh BA, Ghosh P, et al. Effect of anti-inflammatory drugs on sulphated glycosaminoglycan synthesis in aged human articular cartilage. Ann Rheum Dis 1976; 35: 487–95
Brandt KD. Effects of nonsteroidal anti-inflammatory drugs on chondrocyte metabolism in vitro and in vivo. Am J Med 1987; 83(5A): 29–34
Willbrink B, Van der Veen MJ, Huber H, et al. In vitro influence of ketoprofen on proteoglycan metabolism of human normal and osteoarthritic cartilage. Agents Actions 1991; 32(2/4): 154–9
Roy WS. The metabolism of normal and osteoarthritic human articular cartilage and the effect of NSAID on this metabolism. MPhil Thesis, 1994: Cambridge
Pelletier JP, Cloutier JM, Martel-Pelletier J. In vitro effect of tiaprofenic acid, sodium salicylate and hydrocortisone on the proteoglycan metabolism of human osteoarthritic cartilage. J Rheumatol 1989; 165: 646–55
Dingle JT. The effect of NSAIDs in human articular cartilage GAG synthesis. Eur J Rheumatol Inflamm 1996; 16: 47–52
Gonzalez E, De La Cruz C, de Nicolas R, et al. Long term effects of NSAID on the production of cytokines and other inflammatory mediators by blood cells of patients with OA. Agents Actions 1994; 41: 171–8
Dingle JT, Saklatvala J, Hembry R, et al. A cartilage catabolic factor from synovium. Biochem J 1979; 184: 177–80
Saklatvala J, Sarsfield SJ. How do IL-1 and TNF induce degradation of proteoglycan in cartilages. In: Glauert AM, editor. The control of tissue damage. Amsterdam: Elsevier, 1988: 97–106
Arend WP, Dayer J-M. Cytokines and cytokine inhibitors or antagonists in rheumatoid arthritis. Arthritis Rheum 1990; 33: 305–15
Barrett AJ, Saklatvala J. Proteinases in joint disease. In: Kelly WN, Harris ED, Ruddy S, et al., editors. Textbook of rheumatology. 2nd ed. Philadelphia: WB Saunders, 1985: 182–96
Buttle DJ, Saklatvala J, Tamai M, et al. Inhibition of interleukin 1-stimulated cartilage proteoglycan degradation by a lipophilic inactivator of cysteine endopeptidase. Biochem J 1991; 280: 175–7
Martel-Pelletier J, McCollum R, Fujimoto N, et al. Excess of metalloproteases over tissue inhibitor or metalloprotease may contribute to cartilage degradation in osteoarthritis and rheumatoid arthritis. Lab Invest 1994; 70: 807–15
Shield MJ. Misoprostol: New Frontiers; benefits beyond the gastrointestinal tract. Scand J Rheumatol 1992; Suppl. 92: 31–52
Dingle JT, Horner A, Shield M. The sensitivity of synthesis of human cartilage matrix to inhibition by IL1 suggests a mechanism for the development of osteoarthritis. Cell Biochem Funct 1991; 9: 99–102
Dingle JT, Fell HB, Lucy JA. Synthesis of connective tissue components. The effect of retinol and hydrocortisone on cultured limb-bone rudiments. Biochem J 1996; 98: 173–81
Dingle JT. Heberden Oration 1978. Recent studies on the control of joint damage: the contribution of the Strangeways Research Laboratory. Ann Rheum Dis 1979; 38: 201–14
Dingle JT. The use of cytokines and NSAIDs in osteoarthritis; the use of human cartilage in drug assessment. Eur J Rheumatol Inflammation 1992; 12: 3–8
Dingle JT, Parker M. Chondroprotection and the role of NSAID: a study of human cartilage metabolism. Brazil J Rheum 1997; 37: 37–46
Pelletier JP, Cloutier JM, Martel-Pelletier J. Evidence for involvement of IL1 in OA cartilage degradation: protective effect of NSAID. J Rheumatol 1989; 16 Suppl. 18: 19–27
Cawston TE. Metalloproteinases and connective tissue breakdown. Rheumatol Rev 1994; 3: 147–54
Arend WP, Dayer JM. Naturally occurring inhibitors of cytokines in immunopharmacology of joints and connective tissue. In: Davies EM, Dingle JT, editors. London: Academic Press, 1994: 129–49
Bort R, Ponsoda X, Carrases E, et al. Comparative metabolism of the NSAID aceclofenac in the rat, monkey and man. Drug Metabolism 1997. In press
Dingle JT, Page Thomas DP, King B, et al. In vivo studies of articular tissue damage mediated by catabolin/interleukin 1. Ann Rheum Dis 1987; 46: 527–33
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dingle, J.T., Parker, M. NSAID Stimulation of Human Cartilage Matrix Synthesis. Clin. Drug Invest. 14, 353–362 (1997). https://doi.org/10.2165/00044011-199714050-00003
Published:
Issue Date:
DOI: https://doi.org/10.2165/00044011-199714050-00003