Summary
Information on the long term efficacy and tolerability of vigabatrin in children is scarce. This open, 4-year add-on study reports the effects of vigabatrin in 22 children aged 26 to 220 months with refractory epilepsy. The 22 children entered this 4-year long term study after 2 months of add-on vigabatrin at a fixed dose, and two 2-month periods of vigabatrin adjustment.
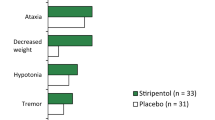
During those 4 years, 2 patients with complete response moved away, 3 with a seizure reduction of > 50% left the study at parental request, and 5 (23%) were withdrawn because of inefficacy or an increase in seizure frequency. Five patients (23%) reported adverse events such as drowsiness, fatigue, apathy, constipation and aggressiveness, although none resulted in vigabatrin withdrawal. In the 12 children completing the study, vigabatrin at a daily dose of 63.8 ± 19.0 mg/kg reduced the median number of seizures from 30 seizures/month at baseline to 0 seizures/month at the end of the 4 years (p < 0.05) — a seizure reduction of > 50% in 9 of the 12 patients (complete control in 7 patients).
It is concluded that add-on vigabatrin achieved a 4-year long term benefit in at least 40% of children with refractory partial epilepsy, and that the development of tolerance or adverse events were rarely observed in this period.
Similar content being viewed by others
References
Jung MJ, Lippert B, Metcalf BW, et al. Gamma-vinyl GABA (4-amino-hex-5-enoic acid), a new selective irreversible inhibitor of GABA-T: effects on brain GABA metabolism in mice. J Neurochem 1977; 29: 797–802
Metcalf BW. Inhibitors of GABA metabolism. Biochem Pharmacol 1979; 28: 1705–12
Duncan JS. Vigabatrin. In: Trimble MR, editor. New anticonvulsants: advances in the treatment of epilepsy. Chichester: John Wiley & Sons, 1994: 113–25
Dam M. Long-term evaluation of vigabatrin (gamma vinyl GABA) in epilepsy. Epilepsia 1989; 30 Suppl. 3: 26–30
Remy C, Beaumont D. Efficacy and safety of vigabatrin in the long-term treatment of refractory epilepsy. Br J Clin Pharmacol 1989; 27 Suppl. 1: 125–9
Cocito L, Maffini M, Perfumo P, et al. Vigabatrin in complex partial seizures: a long-term study. Epilep Res 1989; 3: 160–6
Tartara A, Manni R, Galimberti CA, et al. Vigabatrin in the treatment of epilepsy: a long-term follow-up study. J Neurol Neurosurg Psychiatry 1989; 52: 467–71
Grünewald RA, Thompson PJ, Corcoran R, et al. Effects of vigabatrin on partial seizures and cognitive function. J Neurol Neurosurg Psychiatry 1994; 57: 1057–63
Browne TR, Mattson RH, Penry JK, et al. Multicenter long-term safety and efficacy study of vigabatrin for refractory complex partial seizures: an update. Neurology 1991; 41: 363–4
Reynolds EH, Ring HA, Fair IN, et al. Open, double-blind and long-term study of vigabatrin in chronic epilepsy. Epilepsia 1991; 32: 530–8
Sivenius J, Ylinen A, Murros K, et al. Vigabatrin in drug-resistant partial epilepsy: a 5-year follow-up period. Neurology 1991; 41: 562–5
Pitkänen A, Ylinen A, Matilainen R, et al. Long-term antiepileptic efficacy of vigabatrin in drug-refractory epilepsy in mentally retarded patients. A 5-year follow-up study. Arch Neurol 1993; 50: 24–9
Livingston JH, Beaumont D, Arzimanoglou A, et al. Vigabatrin in the treatment of epilepsy in children. Br J Clin Pharmacol 1989; 27 Suppl. 1: 109–12
Luna D, Dulac O, Pajot N, et al. Vigabatrin in the treatment of childhood epilepsies: a single-blind placebo-controlled study. Epilepsia 1989; 30: 430–7
Dulac O, Chiron C, Luna D, et al. Vigabatrin in childhood epilepsy. J Child Neurol 1991; 6 Suppl. 2: 30–7
Herranz JL, Arteaga R, Farr IN, et al. Dose-response study of vigabatrin in children with refractory epilepsy. J Child Neurol 1991; 6 Suppl. 2: 45–51
Uldall P, Alving J, Gram L, et al. Vigabatrin in pediatric epilepsy — an open study. J Child Neurol 1991; 6 Suppl. 2: 38–44
Gibbs JM, Appleton RE, Rosenbloom L. Vigabatrin in intractable childhood epilepsy: a retrospective study. Pediatr Neurol 1992; 8: 338–40
Dalla Bernardina B, Simeone M, Fontana E, et al. Il vigabatrin nel trattamento delle epilessie infantili: a proposito di 49 osservazioni personali. Boll Lega It Epil 1992; 79/80: 311–2
Schmitz-Moormann W, Kruse R. Vigabatrin-langzeittherapie bei symptomatischen epilepsien mit komplex-fokalen anfallen im kindesalter. Akt Neurol 1992; 19 Suppl. 1: 23–5
Curatolo R. Vigabatrin for refractory partial seizures in children with tuberous sclerosis. Neuropediatrics 1994; 25: 55
Feucht M, Brantner-Inthaler S. Gamma-vinyl-GABA (vigabatrin) in the therapy of Lennox-Gastaut syndrome: an open study. Epilepsia 1994; 35: 993–8
Uldall P, Alving J, Gram L, et al. Vigabatrin in childhood epilepsy: a 5-year follow-up study. Neuropediatrics 1995; 26: 253–6
Laub MC. Vigabatrin zur behandlung therapieresistenter epilepsien im kindes- und Jugendalter. Monatsschr Kinderheilkd 1995; 143: 496–9
Dalla Bernardina B, Fontana E, Vigevano F, et al. Efficacy and tolerability of vigabatrin in children with refractory partial seizures: a single-blind dose-increasing study. Epilepsia 1995; 36(7): 687–91
Sheth RD, Buckley D, Penney S, et al. Vigabatrin in childhood epilepsy: comparable efficacy for generalized and partial seizures. Clin Neuropharmacol 1996; 19(4): 297–304
Aicardi J, Sabril IS investigator and peer review groups, Mumford JP, et al. Vigabatrin as initial therapy for infantile spasms: a European retrospective survey. Epilepsia 1996; 37(7): 638–42
Appleton RE. The role of vigabatrin in the management of infantile epileptic syndromes. Neurology 1993; 43 Suppl. 5: 21–3
White HL. 4-Aminobutyrate: 2-oxoglutarate aminotransferase in blood platelets. Science 1979; 205: 696–8
Eslami M, Stuart JD, Dean RW. Analysis of taurine in blood plasma of epileptic patients using an improved isocratic HPLC method for amino acids. J Liq Chromatogr 1987; 10: 977–95
Arteaga R, Herranz JL, Valdizán EM, et al. Gamma-vinyl-GABA (vigabatrin): relationship between dosage, plasma concentrations, platelet GABA-transaminase inhibition, and seizure reduction in epileptic children. Epilepsia 1992; 33: 923–31
Lowry OH, Rosebrough NJ, Farr AL, et al. Protein measurement with the folin-phenol reagent. J Biol Chem 1951; 193: 265–75
Michelucci R, Tassinari CA. Response to vigabatrin in relation to seizure type. Br J Clin Pharmacol 1989; 27 Suppl. 1: 119–24
Berman RG, Vaughan VC, editors. Nelson textbook of pediatrics. Philadelphia: Saunders Co., 1991
Valdizán E, Armijo JA. Relationship between platelet and brain GABA transaminase inhibition by single and multiple doses of vigabatrin in rats. Epilepsia 1991; 32: 735–42
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Arteaga, R., Herranz, J.L. & Armijo, J.A. Add-On Vigabatrin in Children with Refractory Epilepsy. Clin. Drug Invest. 12, 287–297 (1996). https://doi.org/10.2165/00044011-199612060-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00044011-199612060-00002