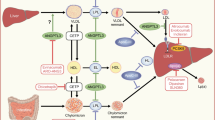
Abstract
The metabolic syndrome consists of a combination of cardiovascular risk factors that include hyperglycemia with or without type 2 diabetes mellitus, visceral obesity, elevated blood pressure, and atherogenic dyslipidemia. These interrelated disorders and their associated lipotoxicity, oxidative stress, and inflammatory state predispose to a constellation of cardiovascular conditions leading to high risk of heart attack, stroke, renal failure, blindness, and lower extremity amputation. Visceral obesity, a prime risk factor for type 2 diabetes and a major component of the metabolic syndrome, potentiates atherogenesis, atherosclerosis, organ lipotoxicity, and oxidative tissue damage.
Peroxisome proliferator-activated receptors (PPARs) are relatively recently discovered nuclear transcription factors that are modulated by dietary fatty acids, including the essential polyunsaturated fatty acids, arachidonic acid and its metabolites, and are essential to the control of energy metabolism. Of the three PPAR isoforms (α, γ, and δ), synthetic pharmaceutical ligands that activate PPARα (the antidyslipidemic fibric acid derivatives [‘fibrates’]) and PPARγ (the antidiabetic thiazolidinediones) have been studied extensively. Recently developed dual PPARα/γ agonists may combine the therapeutic effects of these drugs, creating the expectation of greater efficacy, and perhaps other advantages in the treatment of type 2 diabetes and the metabolic syndrome. However, thiazolidinediones are hampered by adverse effects related to increased weight gain and fluid overload. It remains to be seen whether the dual PPARα/γ agonists currently under development have similar limitations. Nevertheless, existing clinical data imply that the combined effects of thiazolidinediones and fibrates are likely to be emulated by dual PPARα/γ agonists, providing superior efficacy to these classes for the treatment of type 2 diabetes, the metabolic syndrome, and their cardiovascular and other end-organ complications.




Similar content being viewed by others
References
Zimmet P, Alberti KG, Shaw J. Global and societal implications of the diabetes epidemic. Nature 2001; 414: 782–7
Eckel RH, Grundy SM, Zimmet PZ. The metabolic syndrome. Lancet 2005; 365: 1415–28
Amos AF, McCarty DJ, Zimmet P. The rising global burden of diabetes and its complications: estimates and projections to the year 2010. Diabet Med 1997; 14Suppl. 5: S1–85
Haffner SM, Lehto S, Ronnemaa T, et al. Mortality from coronary heart disease in subjects with type 2 diabetes and in nondiabetic subjects with or without prior myocardial infarction. N Engl J Med 1998; 339: 229–34
Grundy SM, Brewer JR HB, Cleeman JI, et al. Definition of metabolic syndrome: report of the National Heart, Lung, and Blood Institute/American Heart Association conference on scientific issues related to definition. Circulation 2004; 109: 433–8
Ford ES. Risks for all-cause mortality, cardiovascular disease, and diabetes associated with the metabolic syndrome: a summary of the evidence. Diabetes Care 2005; 28: 1769–78
Cameron AJ, Shaw JE, Zimmet PZ. The metabolic syndrome: prevalence in worldwide populations. Endocrinol Metab Clin North Am 2004; 33: 351–75
Park YW, Zhu S, Palaniappan L, et al. The metabolic syndrome: prevalence and associated risk factor findings in the US population from the Third National Health and Nutrition Examination Survey, 1988–1994. Arch Intern Med 2003; 163: 427–36
Unger RH. Lipid overload and overflow: metabolic trauma and the metabolic syndrome. Trends Endocrinol Metab 2003; 14: 398–403
Kahn R, Buse J, Ferrannini E, et al. The metabolic syndrome: time for a critical appraisal: joint statement from the American Diabetes Association and the European Association for the study of diabetes. Diabetes Care 2005; 28: 2289–304
Kliewer SA, Umesono K, Noonan DJ, et al. Convergence of 9-cis retinoic acid and peroxisome proliferator signalling pathways through heterodimer formation of their receptors. Nature 1992; 358: 771–4
Mangelsdorf DJ, Evans RM. The RXR heterodimers and orphan receptors. Cell 1995; 83: 841–50
Schoonjans K, Martin G, Staels B, et al. Peroxisome proliferator-activated receptors, orphans with ligands and functions. Curr Opin Lipidol 1997; 8: 159–66
Lehmann JM, Moore LB, Smith-Oliver TA, et al. An antidiabetic thiazolidinedione is a high affinity ligand for peroxisome proliferator-activated receptor gamma (PPAR gamma). J Biol Chem 1995; 270: 12953–6
Kersten S, Desvergne B, Wahli W. Roles of PPARs in health and disease. Nature 2000; 405: 421–4
Willson TM, Brown PJ, Sternbach DD, et al. The PPARs: from orphan receptor to drug discovery. J Med Chem 2000; 43: 527–50
Miller AR, Etgen GJ. Novel peroxisome proliferator-activated receptor ligands for type 2 diabetes and the metabolic syndrome. Expert Opin Investig Drugs 2003; 12: 1489–500
Henke BR. Peroxisome proliferator-activated receptor alpha/gamma dual agonists for the treatment of type 2 diabetes. J Med Chem 2004 Aug 12; 47(17): 4118–27
Staels B, Fruchart JC. Therapeutic roles of peroxisome proliferator-activated receptor agonists. Diabetes 2005; 54: 2460–70
Shulman AI, Mangelsdorf DJ. Retinoid x receptor heterodimers in the metabolic syndrome. N Engl J Med 2005; 353: 604–15
Berger JP, Akiyama TE, Meinke PT. PPARs: therapeutic targets for metabolic disease. Trends Pharmacol Sci 2005; 26: 244–51
Kliewer SA, Xu HE, Lambert MH, et al. Peroxisome proliferator-activated receptors: from genes to physiology. Recent Prog Horm Res 2001; 56: 239–63
Lakka HM, Laaksonen DE, Lakka TA, et al. The metabolic syndrome and total and cardiovascular disease mortality in middle-aged men. JAMA 2002; 288: 2709–16
O’Brien PE, Dixon JB. The extent of the problem of obesity. Am J Surg 2002; 184: 4S–8S
Unger RH. Lipotoxic diseases. Annu Rev Med 2002; 53: 319–36
Laaksonen DE, Lakka HM, Niskanen LK, et al. Metabolic syndrome and development of diabetes mellitus: application and validation of recently suggested definitions of the metabolic syndrome in a prospective cohort study. Am J Epidemiol 2002; 156: 1070–7
NCEP/ATP III. Executive summary of the third report of the national cholesterol education program expert panel on detection, evaluation and treatment of high blood cholesterol in adults (Adult Treatment Panel III). JAMA 2001; 285: 2486–97
World Health Organization. Report of a WHO consultation. In: Alwan A, King H, editors. Definition, diagnosis and classification of diabetes mellitus and its complications: part 1. Diagnosis and classification of diabetes mellitus. Geneva: World Health Organization, Department of Noncommunicable Disease Surveillance, 1999: 1–59
Moller DE, Kaufman KD. Metabolic syndrome: a clinical and molecular perspective. Annu Rev Med 2005; 56: 45–62
International Diabetes Federation. International Diabetes Federation worldwide definition of the metabolic syndrome [online]. Available from URL: http://www.idf.org/webdata/docs/IDF_Metasyndrome_definition.pdf [Accessed 2005 Nov 20]
Hotamisligil GS, Shargill NS, Spiegelman BM. Adipose expression of tumor necrosis factor-alpha: direct role in obesity-linked insulin resistance. Science 1993; 259: 87–91
Kern PA, Saghizadeh M, Ong JM, et al. The expression of tumor necrosis factor in human adipose tissue: regulation by obesity, weight loss, and relationship to lipoprotein lipase. J Clin Invest 1995; 95: 2111–9
Matsuzawa Y, Funahashi T, Nakamura T. Molecular mechanism of metabolic syndrome X: contribution of adipocytokines adipocyte-derived bioactive substances. Ann N Y Acad Sci 1999; 892: 146–54
Hotta K, Funahashi T, Arita Y, et al. Plasma concentrations of a novel, adiposespecific protein, adiponectin, in type 2 diabetic patients. Arterioscler Thromb Vasc Biol 2000; 20: 1595–9
Kumada M, Kihara S, Sumitsuji S, et al. Osaka CAD Study Group: coronary artery disease: association of hypoadiponectinemia with coronary artery disease in men. Arterioscler Thromb Vasc Biol 2003; 23: 85–9
Funahashi T, Nakamura T, Shimomura I, et al. Role of adipocytokines on the pathogenesis of atherosclerosis in visceral obesity. Intern Med 1999; 38: 202–6
Adamczak M, Wiecek A, Funahashi T, et al. Decreased plasma adiponectin concentration in patients with essential hypertension. Am J Hypertens 2003; 16: 72–5
Yamamoto Y, Hirose H, Saito I, et al. Correlation of the adipocyte-derived protein adiponectin with insulin resistance index and serum high-density lipoprotein-cholesterol, independent of body mass index, in the Japanese population. Clin Sci (Lond) 2002; 103: 137–42
Kazumi T, Kawaguchi A, Sakai K, et al. Young men with high-normal blood pressure have lower serum adiponectin, smaller LDL size, and higher elevated heart rate than those with optimal blood pressure. Diabetes Care 2002; 25: 971–6
Pershadsingh HA. Treating the metabolic syndrome using angiotensin receptor antagonists that selectively modulate peroxisome proliferator-activated receptor-gamma. Int J Biochem Cell Biol. Epub 2005 Sep 9
Staels B. PPARγ and atherosclerosis. Curr Med Res Opin 2005; 21Suppl. 1: S13–20
Israelian-Konaraki Z, Reaven PD. Peroxisome proliferator-activated receptor-alpha and atherosclerosis: from basic mechanisms to clinical implications. Cardiology 2005; 103: 1–9
Kliewer SA, Sundseth SS, Jones SA, et al. Fatty acids and eicosanoids regulate gene expression through direct interactions with peroxisome proliferator-activated receptors alpha and gamma. Proc Natl Acad Sci U S A 1997; 94: 4318–23
Xu HE, Lambert MH, Montana VG, et al. Molecular recognition of fatty acids by peroxisome proliferator-activated receptors. Mol Cell 1999; 3: 397–403
Delerive P, Fruchart JC, Staels B. Peroxisome proliferator-activated receptors in inflammation control. J Endocrinol 2001; 169: 453–9
Marx N, Duez H, Fruchart JC, et al. Peroxisome proliferator-activated receptors and atherogenesis: regulators of gene expression in vascular cells. Circ Res 2004; 94: 1168–78
Peters JM, Hennuyer N, Staels B, et al. Alterations in lipoprotein metabolism in peroxisome proliferator-activated receptor alpha-deficient mice. J Biol Chem 1997; 272: 27307–12
Hunninghake DB, Peters JR. Effect of fibric acid derivatives on blood lipid and lipoprotein levels. Am J Med 1987; 83: 44–9
Staels B, Dallongeville J, Auwerx J, et al. Mechanism of action of fibrates on lipid and lipoprotein metabolism. Circulation 1998; 98: 2088–93
Hertz R, Bishara-Shieban J, Bar-Tana J. Mode of action of peroxisome proliferators as hypolipidemic drugs: suppression of apolipoprotein C-III. J Biol Chem 1995; 270: 13470–5
Ziouzenkova O, Perrey S, Asatryan L, et al. Lipolysis of triglyceride-rich lipoproteins generates PPAR ligands: evidence for an antiinflammatory role for lipoprotein lipase. Proc Natl Acad Sci U S A 2003; 100: 2730–5
Lee CH, Olson P, Evans RM. Minireview: lipid metabolism, metabolic diseases, and peroxisome proliferator-activated receptors. Endocrinology 2003; 144: 2201–7
Gotto Jr AM. Risk factor modification: rationale for management of dyslipidemia. Am J Med 1998; 104: 6S–8S
Gotto Jr AM, Grundy SM. Lowering LDL cholesterol: questions from recent meta-analyses and subset analyses of clinical trial data issues from the Interdisciplinary Council on Reducing the Risk for Coronary Heart Disease, ninth council meeting. Circulation 1999; 99: E1–7
Watts GF, Dimmitt SB. Fibrates, dyslipoproteinaemia and cardiovascular disease. Curr Opin Lipidol 1999; 10: 561–74
Despres JP, Lemieux I, Robins SJ. Role of fibric acid derivatives in the management of risk factors for coronary heart disease. Drugs 2004; 64: 2177–98
Rubins HB, Robins SJ, Collins D, et al. Gemfibrozil for the secondary prevention of coronary heart disease in men with low levels of high-density lipoprotein cholesterol: Veterans Affairs High-Density Lipoprotein Cholesterol Intervention Trial Study Group. N Engl J Med 1999; 341: 410–8
Diabetes Atherosclerosis Intervention Study. Effect of fenofibrate on progression of coronary-artery disease in type 2 diabetes: DAIS a randomised study. Lancet 2001; 357: 905–10
BIP Study Group. Secondary prevention by raising HDL cholesterol and reducing triglycerides in patients with coronary artery disease: the Bezafibrate Infarction Prevention (BIP) study. Circulation 2000; 102: 21–7
Tenenbaum A, Motro M, Fisman EZ, et al. Bezafibrate for the secondary prevention of myocardial infarction in patients with metabolic syndrome. Arch Intern Med 2005; 165: 1154–60
Tenenbaum A, Motro M, Fisman EZ, et al. Effect of bezafibrate on incidence of type 2 diabetes mellitus in obese patients. Eur Heart J. Epub 2005 May 4
Xue JC, Schwarz EJ, Chawla A, et al. Distinct stages in adipogenesis revealed by retinoid inhibition of differentiation after induction of PPARgamma. Mol Cell Biol 1996; 16: 1567–75
Fajas L, Auboeuf D, Raspe E, et al. The organization, promoter analysis, and expression of the human PPARgamma gene. J Biol Chem 1997; 272: 18779–89
Elbrecht A, Chen Y, Cullinan CA, et al. Molecular cloning, expression and characterization of human peroxisome proliferator activated receptors gamma 1 and gamma 2. Biochem Biophys Res Commun 1996; 224: 431–7
Vidal-Puig AJ, Considine RV, Jimenez-Linan M, et al. Peroxisome proliferator-activated receptor gene expression in human tissues: effects of obesity, weight loss, and regulation by insulin and glucocorticoids. J Clin Invest 1997; 99: 2416–22
Pershadsingh HA. Pharmacological peroxisome proliferator-activated receptor gamma ligands: emerging clinical indications beyond diabetes. Expert Opin Investig Drugs 1999; 8: 1859–72
Hevener AL, He W, Barak Y, et al. Muscle-specific Pparg deletion causes insulin resistance. Nat Med 2003; 9: 1491–7
Morikang E, Benson SC, Kurtz TW, et al. Effects of thiazolidinediones on growth and differentiation of human aorta and coronary myocytes. Am J Hypertens 1997; 10: 440–6
Roberts AW, Thomas A, Rees A, et al. Peroxisome proliferator-activated receptor-gamma agonists in atherosclerosis: current evidence and future directions. Curr Opin Lipidol 2003; 14: 567–73
Pershadsingh HA. Peroxisome proliferator-activated receptor-gamma: therapeutic target for diseases beyond diabetes: quo vadis? Expert Opin Investig Drugs 2004; 13: 215–28
Way JM, Harrington WW, Brown KK, et al. Comprehensive messenger ribonucleic acid profiling reveals that peroxisome proliferator-activated receptor gamma activation has coordinate effects on gene expression in multiple insulin-sensitive tissues. Endocrinology 2001; 142: 1269–77
Young PW, Cawthorne MA, Coyle PJ, et al. Repeat treatment of obese mice with BRL 49653, a new potent insulin sensitizer, enhances insulin action in white adipocytes: association with increased insulin binding and cell-surface GLUT4 as measured by photoaffinity labeling. Diabetes 1995; 44: 1087–92
Aitman TJ, Glazier AM, Wallace CA, et al. Identification of Cd36 (Fat) as an insulin-resistance gene causing defective fatty acid and glucose metabolism in hypertensive rats. Nat Genet 1999 Jan; 21(1): 76–83
Tontonoz P, Hu E, Spiegelman BM. Stimulation of adipogenesis in fibroblasts by PPAR gamma 2, a lipid-activated transcription factor. Cell 1994; 79: 1147–56
Schoonjans K, Peinado-Onsurbe J, Lefebvre AM, et al. PPARalpha and PPAR-gamma activators direct a distinct tissue-specific transcriptional response via a PPRE in the lipoprotein lipase gene. EMBO J 1996; 15: 5336–48
Martin G, Schoonjans K, Lefebvre AM, et al. Coordinate regulation of the expression of the fatty acid transport protein and acyl-CoA synthetase genes by PPARalpha and PPARgamma activators. J Biol Chem 1997; 272: 28210–7
Yang WS, Jeng CY, Wu TJ, et al. Synthetic peroxisome proliferator-activated receptor-gamma agonist, rosiglitazone, increases plasma levels of adiponectin in type 2 diabetic patients. Diabetes Care 2002; 25: 376–80
Miyazaki Y, Mahankali A, Wajcberg E, et al. Effect of pioglitazone on circulating adipocytokine levels and insulin sensitivity in type 2 diabetic patients. J Clin Endocrinol Metab 2004; 89: 4312–9
Kadowaki T, Hara K, Yamauchi T, et al. Molecular mechanism of insulin resistance and obesity. Exp Biol Med (Maywood) 2003; 228: 1111–7
Goldberg RB, Kendall DM, Deeg MA, et al. A comparison of lipid and glycemic effects of pioglitazone and rosiglitazone in patients with type 2 diabetes and dyslipidemia. Diabetes Care 2005; 28: 1547–54
Delea TE, Edelsberg JS, Hagiwara M, et al. Use of thiazolidinediones and risk of heart failure in people with type 2 diabetes: a retrospective cohort study. Diabetes Care 2003; 26: 2983–9
Scheen AJ. Combined thiazolidinedione-insulin therapy: should we be concerned about safety? Drug Saf 2004; 27: 841–56
Mudaliar S, Chang AR, Henry RR. Thiazolidinediones, peripheral edema, and type 2 diabetes: incidence, pathophysiology, and clinical implications. Endocr Pract 2003; 9: 406–16
Shearer BG, Hoekstra WJ. Recent advances in peroxisome proliferator-activated receptor science. Curr Med Chem 2003; 10: 267–80
Skrumsager BK, Nielsen KK, Muller M, et al. Ragaglitazar: the pharmacokinetics, pharmacodynamics, and tolerability of a novel dual PPAR alpha and gamma agonist in healthy subjects and patients with type 2 diabetes. J Clin Pharmacol 2003; 43: 1244–56
Zhang H, Zhang A, Kohan DE, et al. Collecting duct-specific deletion of peroxisome proliferator-activated receptor gamma blocks thiazolidinedione-induced fluid retention. Proc Natl Acad Sci U S A 2005; 102: 9406–11
Guan Y, Hao C, Cha DR, et al. Thiazolidinediones expand body fluid volume through PPARgamma stimulation of ENaC-mediated renal salt absorption. Nat Med 2005; 11: 861–6
Chaput E, Saladin R, Silvestre M, et al. Fenofibrate and rosiglitazone lower serum triglycerides with opposing effects on body weight. Biochem Biophys Res Commun 2000; 271: 445–50
Guerre-Millo M, Gervois P, Raspe E, et al. Peroxisome proliferator-activated receptor alpha activators improve insulin sensitivity and reduce adiposity. J Biol Chem 2000; 275: 16638–42
Jeong S, Kim M, Han M, et al. Fenofibrate prevents obesity and hypertriglyceridemia in low-density lipoprotein receptor-null mice. Metabolism 2004; 53: 607–13
Citeline Intelligence Solutions (PO Box 869, Petaluma, CA 94953, USA). Metabolic syndrome: dual PPAR agonists and anti-obesity drugs dominate drug development efforts. A Citeline intelligence report, February 2005. Available to purchase from: http:ўw.citeline.com [Accessed 2005 Sep 22]
Rubin CJ, Mohideen P, Ledeine J-M, et al. Improvement of glycemic control with muraglitazar, a novel dual PPAR-α/γ agonist, in combination with metformin in patients with type 2 diabetes: a double-blind, randomized, pioglitazone-controlled study: 65th Scientific Sessions [abstract no. 14-OR]. San Diego (CA): American Diabetes Association, 2005
Nissen SE, Wolski K, Topol EJ. Effect of muraglitazar on death and major adverse cardiovasular events in patients with type 2 diabetes mellitus. JAMA 2005; 294: 2581–6
Goldstein BJ, Rosentock J, Anzalone D, et al. Tesaglitazar improves glucose and lipid abnormalities in patients with type 2 diabetes: 65th Scientific Sessions [abstract no. 83-OR]. San Diego (CA): American Diabetes Association, 2005
Fagerberg B, Edwards S, Halmos T, et al. Tesaglitazar, a novel dual peroxisome proliferator-activated receptor alpha/gamma agonist, dose-dependently improves the metabolic abnormalities associated with insulin resistance in a non-diabetic population. Diabetologia 2005; 48: 1716–25
Langenfeld MR, Forst T, Hohberg C, et al. Pioglitazone decreases carotid intima-media thickness independently of glycemic control in patients with type 2 diabetes mellitus: results from a controlled randomized study. Circulation 2005; 111: 2525–31
Nakamura T, Matsuda T, Kawagoe Y, et al. Effect of pioglitazone on carotid intima-media thickness and arterial stiffness in type 2 diabetic nephropathy patients. Metabolism 2004; 53: 1382–6
Sidhu JS, Kaposzta Z, Markus HS, et al. Effect of rosiglitazone on common carotid intima-media thickness progression in coronary artery disease patients without diabetes mellitus. Arterioscler Thromb Vasc Biol 2004; 24: 930–4
Takagi T, Yamamuro A, Tamita K, et al. Pioglitazone reduces neointimal tissue proliferation after coronary stent implantation in patients with type 2 diabetes mellitus: an intravascular ultrasound scanning study. Am Heart J 2003; 146: E5
Choi D, Kim SK, Choi SH, et al. Preventative effects of rosiglitazone on restenosis after coronary stent implantation in patients with type 2 diabetes. Diabetes Care 2004; 27: 2654–60
Osman A, Otero J, Brizolara A, et al. Effect of rosiglitazone on restenosis after coronary stenting in patients with type 2 diabetes. Am Heart J 2004; 147: e23
Dormandy JA, Charbonnel B, Eckland DJ, et al. Secondary prevention of macrovascular events in patients with type 2 diabetes in the PROactive Study: a randomized controlled trial. Lancet 2005; 366: 1279–89
Keech A, Simes RJ, Barter P, et al. Effects of long-term fenofibrate therapy on cardiovascular events in 9795 people with type 2 diabetes mellitus (the FIELD study): randomised controlled trial. Lancet 2005; 366: 1849–61
Spoelstra-de Man AM, Brouwer CB, Stehouwer CD, et al. Rapid progression of albumin excretion is an independent predictor of cardiovascular mortality in patients with type 2 diabetes and microalbuminuria. Diabetes Care 2001; 24: 2097–101
Guan Y. Peroxisome proliferator-activated receptor family and its relationship to renal complications of the metabolic syndrome. J Am Soc Nephrol 2004; 15: 2801–15
Nakamura T, Ushiyama C, Osada S, et al. Pioglitazone reduces urinary podocyte excretion in type 2 diabetes patients with microalbuminuria. Metabolism 2001; 50: 1193–6
Bakris G, Viberti G, Weston WM, et al. Rosiglitazone reduces urinary albumin excretion in type II diabetes. J Hum Hypertens 2003; 17: 7–12
Pistrosch F, Herbrig K, Kindel B, et al. Rosiglitazone improves glomerular hyperfiltration, renal endothelial dysfunction, and microalbuminuria of incipient diabetic nephropathy in patients. Diabetes 2005; 54: 2206–11
Hetzel J, Balletshofer B, Rittig K, et al. Rapid effects of rosiglitazone treatment on endothelial function and inflammatory biomarkers. Arterioscler Thromb Vasc Biol 2005; 25: 1804–9
Petrofsky J, Lee S, Cuneo M. Effects of aging and type 2 diabetes on resting and post occlusive hyperemia of the forearm; the impact of rosiglitazone. BMC Endocr Disord 2005; 5: 4
Wang TD, Chen WJ, Lin JW. Effects of rosiglitazone on endothelial function, C-reactive protein, and components of the metabolic syndrome in nondiabetic patients with the metabolic syndrome. Am J Cardiol 2004; 93: 362–5
Murata T, Hata Y, Ishibashi T, et al. Response of experimental retinal neovascularization to thiazolidinediones. Arch Ophthalmol 2001; 119: 709–17
Murata T, He S, Hangai M, et al. Peroxisome proliferator-activated receptor-gamma ligands inhibit choroidal neovascularization. Invest Ophthalmol Vis Sci 2000; 41: 2309–17
Te Sligte K, Bourass I, Sels JP, et al. Non-alcoholic steatohepatitis: review of a growing medical problem. Eur J Intern Med 2004; 15: 10–21
Promrat K, Lutchman G, Uwaifo GI, et al. A pilot study of pioglitazone treatment for nonalcoholic steatohepatitis. Hepatology 2004; 39: 188–96
Neuschwander-Tetri BA, Brunt EM, Wehmeier KR, et al. Improved nonalcoholic steatohepatitis after 48 weeks of treatment with the PPAR-gamma ligand rosiglitazone. Hepatology 2003; 38: 1008–17
Ruttmann E, Brant LJ, Concin H, et al. Gamma-glutamyltransferase as a risk factor for cardiovascular disease mortality: an epidemiological investigation in a cohort of 163,944 Austrian adults. Circulation 2005; 112: 2130–7
Thamer C, Tschritter O, Haap M, et al. Elevated serum GGT concentrations predict reduced insulin sensitivity and increased intrahepatic lipids. Horm Metab Res 2005; 37: 246–51
Sepilian V, Nagamani M. Effects of rosiglitazone in obese women with polycystic ovary syndrome and severe insulin resistance. J Clin Endocrinol Metab 2005; 90: 60–5
Tarkun I, Cetinarslan B, Turemen E, et al. Effect of rosiglitazone on insulin resistance, C-reactive protein and endothelial function in non-obese young women with polycystic ovary syndrome. Eur J Endocrinol 2005; 153: 115–21
Ortega-Gonzalez C, Luna S, Hernandez L, et al. Responses of serum androgen and insulin resistance to metformin and pioglitazone in obese, insulin-resistant women with polycystic ovary syndrome. J Clin Endocrinol Metab 2005; 90: 1360–5
Brettenthaler N, De Geyter C, Huber PR, et al. Effect of the insulin sensitizer pioglitazone on insulin resistance, hyperandrogenism, and ovulatory dysfunction in women with polycystic ovary syndrome. J Clin Endocrinol Metab 2004; 89: 3835–40
Clark RB. The role of PPARs in inflammation and immunity. J Leukoc Biol 2002; 71: 388–400
Ellis CN, Varani J, Fisher GJ, et al. Troglitazone improves psoriasis and normalizes models of proliferative skin disease: ligands for peroxisome proliferator-activated receptor-gamma inhibit keratinocyte proliferation. Arch Dermatol 2000; 136: 609–16
Feinstein DL, Galea E, Gavrilyuk V, et al. Peroxisome proliferator-activated receptor-gamma agonists prevent experimental autoimmune encephalomyelitis. Ann Neurol 2002; 51: 694–702
Lovett-Racke AE, Hussain RZ, Northrop S, et al. Peroxisome proliferator-activated receptor alpha agonists as therapy for autoimmune disease. J Immunol 2004; 172: 5790–8
Lewis JD, Lichtenstein GR, Stein RB, et al. An open-label trial of the PPAR-gamma ligand rosiglitazone for active ulcerative colitis. Am J Gastroenterol 2001; 96: 3323–8
Schupp M, Janke J, Clasen R, et al. Angiotensin type 1 receptor blockers induce peroxisome proliferator-activated receptor-γ activity. Circulation 2004; 109: 2054–7
Benson SC, Pershadsingh HA, Ho CI, et al. Identification of telmisartan as a unique angiotensin II receptor antagonist with selective PPARγ-modulating activity. Hypertension 2004; 43: 993–1002
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pershadsingh, H.A. Dual Peroxisome Proliferator-Activated Receptor-α/γ Agonists. Mol Diag Ther 5, 89–99 (2006). https://doi.org/10.2165/00024677-200605020-00003
Published:
Issue Date:
DOI: https://doi.org/10.2165/00024677-200605020-00003