Abstract
Objective
To determine the dose-limiting toxicity, maximum tolerated dose, and recommended phase II dosage of oral topotecan when administered in an intermittent schedule.
Study design
Phase I study.
Patients and methods
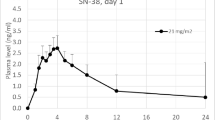
Patients received a fixed dose of oral topotecan daily for 5 of 7 days for 2 or 3 consecutive weeks followed by 1 week of rest (21- or 28-day cycles, respectively). Patients were accrued at seven different dosage levels of topotecan ranging from 1.25 to 3.0 mg/day.
Results
Fifty-five patients received a total of 153 cycles of therapy. Dose-limiting toxicities consisted of neutropenia, thrombocytopenia, and diarrhea and appeared to correlate with patient characteristics associated with low topotecan clearance, such as impaired renal function, low bodyweight, and decreased performance status. Three partial responses were reported in patients with lung cancer (non-small cell in two patients and small cell in one patient).
Conclusions
The recommended dosage for further phase II testing is 2.0 mg/day for 5 of 7 days for 2 weeks in patients with impaired renal function (calculated creatinine clearance ≤70 mL/min) and 2.5 mg/day for 5 of 7 days for 2 weeks in patients with normal renal function (calculated creatinine clearance >70 mL/min). Treatment cycles should be repeated every 21 days.






Similar content being viewed by others
References
Burris III HA, Hanauske AR, Johnson RK, et al. Activity of topotecan, a new topoisomerase I inhibitor against human tumor colony forming units in vitro. J Natl Cancer Inst 1992; 23: 1816–20
Creemers GJ, Lund B, Verweij J. Topoisomerase I inhibitors: topotecan and irinotecan. Cancer Treat Rev 1994; 20: 73–96
Burris III HA, Fields SM. Topoisomerase I inhibitors: an overview of the camptothecin analogs. Hematol Oncol Clin North Am 1994; 8: 333–55
Houghton PJ, Chesire PJ, Myer L, et al. Evaluation of 9-dimethylami-nomethyl-10-hydroxy-camptothecin (topotecan) against xenografts derived from adult and childhood tumors. Cancer Chemother Pharmacol 1991; 31: 229–39
Houghton PJ, Chesire PJ, Hallman JD, et al. Efficacy of topoisomerase I inhibitor topotecan and irinotecan administered at low dose levels in protracted schedules to mice bearing xenografts of human tumors. Cancer Chemother Pharmacol 1995; 36: 393–403
Guichard S, Montazeri A, Chatelut E, et al. Schedule-dependent activity of topotecan in OVCAR-3 ovarian carcinoma xenograft: pharmacokinetic and pharmacodynamic evaluation. Clin Cancer Res 2001; 7: 3222–8
Nakashio A, Fujita N, Rokudai S, et al. Prevention of phosphatidylinositol 3′-kinase-Akt survival signaling pathway during topotecan-induced apoptosis. Cancer Res 2000; 60: 5303–9
Creemers GJ, Gerrits CJH, Eckardt JR, et al. Phase I and pharmacologic study of oral topotecan administered twice daily for 21 days to adult patients with solid tumors. J Clin Oncol 1997; 15: 1087–93
Gerrits CJH, Burris III HA, Schellens JHM, et al. Oral topotecan given once or twice daily for ten days: a phase I and pharmacology study in adult patients with solid tumors. Clin Cancer Res 1998; 4: 1153–8
Bowman LC, Stewart CF, Zamboni WC, et al. Toxicity and pharmacodynamics of oral topotecan in pediatric patients with relapsed solid tumors [abstract]. Proc Am Soc Clin Oncol 1996; 15: 462
Hochster H, Liebes L, Speyer J, et al. Phase I trial of low-dose continuous topotecan infusion in patients with cancer: an active and well-tolerated regimen. J Clin Oncol 1994; 12: 553–9
Creemers GJ, Gerrits CJH, Schellens JHM, et al. Phase II and pharmacologic study of topotecan administered as a 21-day continuous infusion to patients with colorectal cancer. J Clin Oncol 1996; 14: 2540–5
Gerrits CJH, Burris H, Schellens JHM, et al. Five days of oral topotecan (Hycamtin®), a phase I and pharmacological study in adult patients with solid tumors. Eur J Cancer 1998; 34: 1030–5
Danks MK, Pawlik CA, Whipple DO, et al. Intermittent exposure of medulloblastoma cells to topotecan produces growth inhibition equivalent to continuous exposure. Clin Cancer Res 1997; 3: 1731–8
Green S, Weiss GR. Southwest Oncology Group standard response criteria, end- point definitions, and toxicity criteria. Invest New Drags 1992; 10: 239–53
Minami H, Beijnen JH, Verweij J, et al. Limited sampling model for area under the concentration time curve of topotecan. Clin Cancer Res 1996; 2: 43–6
Fleming GF, Kugler JW, Hoffman PC, et al. Phase II trial of paclitaxel and topotecan with granulocyte colony-stimulating factor support in stage IV breast cancer. J Clin Oncol 1998; 16: 2032–7
Takimoto CH, Arbuck SG. Topoisomerase I targeting agents: the campothecins. In: Chabner BA, Longo DL, editors. Cancer chemotherapy and biotherapy: principles and practice. 3rd ed. Philadelphia (PA): Lippincott Williams & Wilkins, 2001: 579–646
Mould DR, Holford NHG, Schellen JHM, et al. Population pharmacokinetic and adverse event analysis of topotecan in patients with solid tumors. Clin Pharmacol Ther 2002; 71: 334–48
Acknowledgment
This research was supported by a grant from GlaxoSmithKline and was presented in part at the American Society of Clinical Oncology 36th Annual Meeting, New Orleans, Louisiana, USA, May 20–23, 2000 (abstract #776). The authors have no conflicts of interest that are directly relevant to the content of this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jones, S.F., Kuhn, J.G., Greco, F.A. et al. Prolonged intermittent oral topotecan in patients with refractory and/or advanced malignancies. Am J Cancer 3, 317–324 (2004). https://doi.org/10.2165/00024669-200403050-00005
Published:
Issue Date:
DOI: https://doi.org/10.2165/00024669-200403050-00005