Abstract
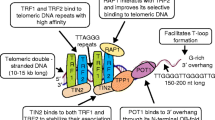
In the increasing search for cancer-specific vulnerabilities, considerable attention has been focused in the past few years on telomeres, the natural termini of eukaryotic chromosomes which are maintained by the enzymatic complex telomerase. The limited capacity to divide is a long-recognized characteristic of normal cells in culture and one that distinguishes them from transformed cells. This finite replicative potential is not linked to the chronological age of the culture, but to the number of cell divisions and to telomere length. Studies in yeast, mice, and humans have shown that telomerase-positive cells can grow indefinitely. However, when telomerase is absent the resulting loss of telomeric DNA from the ends of chromosomes results in the eventual cessation of cell division.
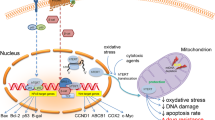
Although most normal human tissues lack telomerase, which limits their proliferative potential, telomerase is expressed in most human cancers. This has raised the intriguing possibility that telomere maintenance might be a block on the path to immortalization and thus may provide a cancer-specific target. Moreover, the recent finding that telomerase is expressed by endothelial cells of brain tumor vasculature strongly suggests that telomerase is involved in brain tumor angiogenesis, and confers a further value to telomerase-inhibiting strategies as a potential target both in neoplastic cells as well as in endothelial cells.

Similar content being viewed by others
References
Zakian VA. Structure and function of telomeres. Annu Rev Genet 1989; 23: 579–604
Blackburn EH, Gall JG. A tandemly repeated sequence at the termini of the extrachromosomal ribosomal RNA genes in Tetrahymena. J Mol Biol 1978; 120(1): 33–53
Klobutcher LA, Swanton MT, Donini P, et al. All gene-sized DNA molecules in four species of hypotrichs have the same terminal sequence and an unusual 3′ terminus. Proc Natl Acad Sci U S A1981; 78(5): 3015–9
Garvik B, Carson M, Hartwell L. S ingle-stranded DNA arising at telomeres in cdc13 mutants may constitute a specific signal for the RAD9 checkpoint. Mol Cell Biol 1995; 11(15): 6128–38
Gravel S, Larrivee M, Labrecque P, et al. Yeast Ku as a regulator of chromosomal DNA end structure. Science 1998; 280(5364): 741–4
vanSteensel B, Smogorzewska A, deLange T. TRF2 protects human telomeres from end-to-end fusions. Cell 1998; 92(3): 401–13
Stewart SA, BenPorath I, Carey VJ, et al. Erosion of the telomeric single-strand overhang at replicative senescence. Nat Genet 2003; 33(4): 492–6
Fang G, Cech TR. Telomerase RNA localized in the replication band and spherical subnuclear organelles inhypotrichousciliates. JCell Biol 1995; 130(2): 243–53
Horvath MP, Schweiker VL, Bevilacqua JM, et al. Crystal structure of the Oxytricha nova telomere end binding protein complexed with single strand DNA. Cell 1998; 95(7): 963–74
Loayza D, de Lange T. POT1 as a terminal transducer of TRF1 telomere length control. Nature 2003; 423(6943): 1013–8
Colgin LM, Baran K, Baumann P, et al. Human POT1 facilitates telomere elongation by telomerase. Curr Biol 2003; 13(11): 942–6
Shore D. Telomerase and telomere-binding proteins: controlling the endgame. Trends Biochem Sci 1997; 22(7): 233–5
Karl Seder J, Broccoli D, Dai Y, et al. p53- and ATM-dependent apoptosis induced by telomeres lacking TRF2. Science 1999; 283(5406): 1321–5
Griffith JD, Comeau L, Rosenfield S, et al. Mammalian telomeres end in a large duplex loop. Cell 1999; 97(4): 503–14
Smith S, Giriat I, Schmitt A, et al. Tankyrase, a poly(ADP-ribose) polymerase at human telomeres. Science 1998; 282(5393): 1484–7
Smith S, deLange T. Tankyrase promotes telomere elongation in human cells. Curr Biol 2000; 10(20): 1299–302
Cook BD, Dynek JN, Chang W, et al. Role for the related poly(ADP-Ribose) polymerases tankyrase 1 and 2 at human telomeres. Mol Cell Biol 2002; 22(1): 332–42
Kim SH, Kaminker P, Campisi J. TIN2, a new regulator of telomere length in human cells. Nat Genet 1999; 23(4): 405–12
Levy MZ, Allsopp RC, Futcher AB, et al. Telomere end-replication problem and cell aging. J Mol Biol 1992; 225(4): 951–60
Olovnikov AM. Principle of marginotomy in template synthesis of polynucleotides [in Russian]. Dokl Akad Nauk SSSR 1971; 201(6): 1496–9
Watson JD. Origin of concatemeric T7 DNA. Nat New Biol 1972; 239(94): 197–201
Lydall D. Hiding at the ends of yeast chromosomes: telomeres, nucleases and checkpoint pathways. J Cell Sci 2003; 116 (Pt 20): 4057–65
Hayflick L. How and why we age. Exp Gerontol 1998; 33(7-8): 639–53
Dimri GP, Lee X, Basile G, et al. A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc Natl Acad Sci U S A 1995; 92(20): 9363–7
Greider CW, Blackburn EH. Identification of a specific telomere terminal transferase activity in Tetrahymena extracts. Cell 1985; 43 (2 Pt 1): 405–13
Morin GB. The human telomere terminal transferase enzyme is a ribonucleoprotein that synthesizes TTAGGG repeats. Cell 1989; 59(3): 521–9
Kim NW, Piatyszek MA, Prowse KR, et al. Specific association of human telomerase activity with immortal cells and cancer. Science 1994; 266(5193): 2011–5
Feng J, Funk WD, Wang SS, et al. The RNA component of human telomerase. Science 1995; 269(5228): 1236–41
Meyerson M, Counter CM, Eaton EN, et al. hEST2, the putative human telomerase catalytic subunit gene, is up-regulated in tumor cells and during immortalization. Cell 1997; 90(4): 785–95
Nakamura TM, Morin GB, Chapman KB, et al. Telomerase catalytic subunit homologs from fission yeast and human. Science 1997; 277(5328): 955–9
Harle-Bachor C, Boukamp P. Telomerase activity in the regenerative basal layer of the epidermis inhuman skin and in immortal and carcinoma-derived skin keratinocytes. Proc Natl Acad Sci U S A 1996; 93(13): 6476–81
Hiyama K, Hirai Y, Kyoizumi S, et al. Activation of telomerase in human lymphocytes and hematopoietic progenitor cells. J Immunol 1995; 155(8): 3711–5
Masutomi K, Yu EY, Khurts S, et al. Telomerase maintains telomere structure in normal human cells. Cell 2003; 114(2): 241–53
Shay JW, Bacchetti S. A survey of telomerase activity in human cancer. Eur J Cancer 1997; 33(5): 787–91
Masutomi K, Kaneko S, Hayashi N, et al. Telomerase activity reconstituted in vitro with purified human telomerase reverse transcriptase and human telomerase RNA component. J Biol Chem 2000; 275(29): 22568–73
Bodnar AG, Ouellette M, Frolkis M, et al. Extension of life-span by introduction of telomerase into normal human cells. Science 1998; 279(5349): 349–52
Nakayama J, Tahara H, Tahara E, et al. Telomerase activation by hTRT in human normal fibroblasts and hepatocellular carcinomas. Nat Genet 1998; 18(1): 65–8
Weinrich SL, Pruzan R, Ma L, et al. Reconstitution of human telomerase with the template RNA component hTR and the catalytic protein subunit hTRT. Nat Genet 1997; 17(4): 498–502
Takakura M, Kyo S, Kanaya T, et al. Cloning of human telomerase catalytic subunit (hTERT) gene promoter and identification of proximal core promoter sequences essential for transcriptional activation in immortalized and cancer cells. Cancer Res 1999; 59(3): 551–7
Hahn WC, Counter CM, Lundberg AS, et al. Creation of human tumour cells with defined genetic elements. Nature 1999; 400(6743): 464–8
Wang J, Xie LY, Allan S, et al. Myc activates telomerase. Genes Dev 1998; 12(12): 1769–74
Wu KJ, Grandori C, Amacker M, et al. Direct activation of TERT transcription by c-MYC. Nat Genet 1999; 21(2): 220–4
Greenberg RA, O’Hagan RC, Deng H, et al. Telomerase reverse transcriptase gene is a direct target of c-Myc but is not functionally equivalent in cellular transformation. Oncogene 1999; 18(5): 1219–26
Falchetti ML, Falcone G, D’Ambrosio E, et al. Induction of telomerase activity in v-myc-transformed avian cells. Oncogene 1999; 18(7): 1515–9
Veldman T, Liu X, Yuan H, et al. Human papillomavirus E6 and Myc proteins associate in vivo and bind to and cooperatively activate the telomerase reverse transcriptase promoter. Proc Natl Acad Sci U S A 2003; 100(14): 8211–6
Ulaner GA, Hu JF, Vu TH, et al. Telomerase activity in human development is regulated by human telomerase reverse transcriptase (hTERT) transcription and by alternate splicing of hTERT transcripts. Cancer Res 1998; 58(18): 4168–72
Colgin LM, Wilkinson C, Englezou A, et al. The hTERT alpha splice variant is a dominant negative inhibitor of telomerase activity. Neoplasia 2000; 2(5): 426–32
Yi X, White DM, Aisner DL, et al. An alternate splicing variant of the human telomerase catalytic subunit inhibits telomerase activity. Neoplasia 2000; 2(5): 433–40
Kang SS, Kwon T, Kwon DY, et al. Akt protein kinase enhances human telomerase activity through phosphorylation of telomerase reverse transcriptase subunit. J Biol Chem 1999; 274(19): 13085–90
Li H, Zhao L, Yang Z, et al. Telomerase is controlled by protein kinase C alpha in human breast cancer cells. J Biol Chem 1998; 273(50): 33436–42
Won J, Yim J, Kim TK. Sp1 and Sp3 recruit histone deacetylase to repress transcription of human telomerase reverse transcriptase (hTERT) promoter in normal human somatic cells. J Biol Chem 2002; 277(41): 38230–8
Akiyama M, Hideshima T, Hayashi T, et al. Nuclear factor-kappaB p65 mediates tumor necrosis factor alpha-induced nuclear translocation of telomerase reverse transcriptase protein. Cancer Res 2003; 63(1): 18–21
Blasco MA, Rizen M, Greider CW, et al. Differential regulation of telomerase activity and telomerase RNA during multi-stage tumorigenesis. Nat Genet 1996; 12(2): 200–4
Lustig AJ. Crisis intervention: the role of telomerase. Proc Natl Acad Sci U S A 1999; 96(7): 3339–41
Kyo S, Kanaya T, Ishikawa H, et al. Telomerase activity in gynecological tumors. Clin Cancer Res 1996; 2(12): 2023–8
Snijders PJ, vanDuin M, Walboomers JM, et al. Telomerase activity exclusively in cervical carcinomas and a subset of cervical intraepithelial neoplasia grade III lesions: strong association with elevated messenger RNA levels of its catalytic subunit and high-risk human papillomavirus DNA. Cancer Res 1998; 58(17): 3812–8
Zhang X, Ying K, Cai X. Detection of telomerase activity in bronchoscopic brush-off cells in lung cancer patients [in Chinese]. Zhonghua Zhong Liu Za Zhi 1998; 20(6): 431–3
Kolquist KA, Ellisen LW, Counter CM, et al. Expression of TERT in early premalignant lesions and a subset of cells in normal tissues. Nat Genet 1998; 19(2): 182–6
Greaves M. Is telomerase activity in cancer due to selection of stem cells and differentiation arrest? Trends Genet 1996; 12(4): 127–8
Blasco MA, Lee HW, Hande MP, et al. Telomere shortening and tumor formation by mouse cells lacking telomerase RNA. Cell 1997; 91(1): 25–34
Rudolph KL, Chang S, Lee HW, et al. Longevity, stress response, and cancer in aging telomerase-deficient mice. Cell 1999; 96(5): 701–12
Greenberg RA, Chin L, Femino A, et al. Short dysfunctional telomeres impair tumorigenesis in the INK4a(delta2/3) cancer-prone mouse. Cell 1999; 97(4): 515–25
Chin L, Artandi SE, Shen Q, et al. p53 deficiency rescues the adverse effects of telomere loss and cooperates with telomere dysfunction to accelerate carcino-genesis. Cell 1999; 97(4): 527–38
Shay JW.Telomerase in human development and cancer. J Cell Physiol 1997; 173(2): 266–70
Bryan TM, Englezou A, Gupta J, et al. Telomere elongation in immortal human cells without detectable telomerase activity. EMBO J 1995; 14(17): 4240–8
Bryan TM, Marusic L, Bacchetti S,et al. The telomere lengthening mechanism in telomerase-negative immortal human cells does not involve the telomerase RNA subunit. HumMol Genet 1997; 6(6): 921–6
Henson JD, Neumann AA, Yeager TR, et al. Alternative lengthening of telomeres in mammalian cells. Oncogene 2002; 21(4): 598–610
Stewart SA, Hahn WC, O’Conner BF, et al. Telomerase contributes to tumorigenesis by a telomere length-independent mechanism. Proc Natl Acad Sci U S A 2002; 26(5): 725–7
Elenbaas B, Spirio L, Koerner F, et al. Human breast cancer cells generated by oncogenic transformation of primary mammary epithelial cells. Genes Dev 2001; 15(1): 50–65
Hahn WC, Dessain SK, Brooks MW, et al. Enumeration of the simian virus 40 early region elements necessary for human cell transformation [published erratum appears in Mol Cell Biol 2002; 22 (10): 3562]. Mol Cell Biol 2002; 22(7): 2111–23
Rich JN, Guo C, McLendon E, et al. A genetically tractable model of human glioma formation. Cancer Res 2001; 61(9): 3556–60
Smith LL, Coller HA, Roberts JM. Telomerase modulates expression of growth-controlling genes and enhances cell proliferation. Nat Cell Biol 2003; 5(5): 474–9
Pallini R, Pierconti F, Falchetti ML, et al. Evidence for telomerase involvement in the angiogenesis of astrocytic tumors: expression of human telomerase reverse transcriptase messenger RNA by vascular endothelial cells. J Neurosurg 2001; 94(6): 961–71
Falchetti ML, Pierconti F, Casalbore P, et al. Glioblastoma induces vascular endothelial cells to express telomerase in vitro. Cancer Res 2003; 63(13): 3750–4
Franco S, Segura I, Riese HH, et al. Decreased B16F10 melanoma growth and impaired vascularization in telomerase-deficient mice with critically short telomeres. Cancer Res 2002; 62(2): 552–9
Venetsanakos E, Mirza A, Fanton C, et al. Induction of tubulogenesis in telomer-ase-immortalized human microvascular endothelial cells by glioblastoma cells. Exp Cell Res 2002; 273(1): 21–33
Holt SE, Shay JW. Role of telomerase in cellular proliferation and cancer. J Cell Physiol 1999; 180(1): 10–8
Kyo S, Takakura M, Tanaka M, et al. Expression of telomerase activity in human chorion. Biochem Biophys Res Commun 1997; 241(2): 498–503
Zhang W, Kapusta LR, Slingerland JM, et al. Telomerase activity in prostate cancer, prostatic intraepithelial neoplasia, and benign prostatic epithelium. Cancer Res 1998; 58(4): 619–21
Soria JC, Gauthier LR, Raymond E, et al. Molecular detection of telomerase-positive circulating epithelial cells in metastatic breast cancer patients. Clin Cancer Res 1999; 5(5): 971–5
Chen XQ, Bonnefoi H, Pelte MF, et al. Telomerase RNA as a detection marker in the serum of breast cancer patients. Clin Cancer Res 2000; 6(10): 3823–6
Heine B, Hummel M, Muller M, et al. Non-radioactive measurement of telomerase activity in human bladder cancer, bladder washings, and in urine. J Pathol 1998; 184(1): 71–6
Tahara H, Yasui W, Tahara E, et al. Immuno-histochemical detection of human telomerase catalytic component, hTERT, in human colorectal tumor and nontumor tissue sections. Oncogene 1999; 18(8): 1561–7
Hiyama E, Hiyama K, Yokoyama T, et al. Correlating telomerase activity levels with human neuroblastoma outcomes. Nat Med 1995; 1(3): 249–55
Hiyama E, Hiyama K, Yokoyama T, et al. Length of telomeric repeats in neuroblastoma: correlation with prognosis and other biological characteristics. Jpn J Cancer Res 1992; 83(2): 159–64
Langford LA, Piatyszek MA, Xu R, et al. Telomerase activity in ordinary meningiomas predicts poor outcome. Hum Pathol 1997; 28(4): 416–20
Falchetti ML, Pallini R, Larocca LM, et al. Telomerase expression in intracranial tumours: prognostic potential for malignant gliomas and meningiomas. J Clin Pathol 1999; 52(3): 234–6
Simon M, Park TW, Leuenroth S, et al. Telomerase activity and expression of the telomerase catalytic subunit, hTERT, in meningioma progression. J Neurosurg 2000; 92(5): 832–40
Al-Mefty O, Borba LAB. Skull base chordomas: a management challenge. J Neurosurg 1997; 86(2): 182–9
Pallini R, Maira G, Pierconti F, et al. Chordoma of the skull base: predictors of tumor recurrence. J Neurosurg 2003; 98(4): 812–22
Collins K, Mitchell JR. Telomerase in the human organism. Oncogene 2002; 21(4): 564–79
Dokal I. Dyskeratosis congenita in all its forms. Br J Haematol 2000; 110(4): 768–79
White LK, Wright WE, Shay JW. Telomerase inhibitors. Trends Biotechnol 2001; 19(3): 114–20
Herbert B, Pitts AE, Baker SI, et al. Inhibition of human telomerase in immortal human cells leads to progressive telomere shortening and cell death. Proc Natl Acad Sci U S A 1999; 96(25): 14276–81
Zhang X, Mar V, Zhou W, et al. Telomere shortening and apoptosis in telomerase-inhibited human tumor cells. Genes Dev 1999; 13(18): 2388–99
Kim MM, Rivera MA, Botchkina IL, et al. A low threshold level of expression of mutant-template telomerase RNA inhibits human tumor cell proliferation. Proc Natl Acad Sci U S A 2001; 98(14): 7982–7
Boklan J, Nanjangud G, Mac Kenzie KL, et al. Limited proliferation and telomere dysfunction following telomerase inhibition in immortal murine fibroblasts. Cancer Res 2002; 62(7): 2104–14
Gonzalez-Suarez E, Samper E, Flores JM, et al. Telomerase-deficient mice with short telomeres are resistant to skin tumorigenesis. Nat Genet 2000; 26(1): 114–7
de Lange T, Jacks T. For better or worse? Telomerase inhibition and cancer. Cell 1999; 98(3): 273–5
Hackett JA, Greider CW. Balancing instability: dual roles for telomerase and telomere dysfunction in tumorigenesis. Oncogene 2002; 21(4): 619–26
Harrington L, Robinson MO. Telomere dysfunction: multiple paths to the same end. Oncogene 2002; 21(4): 592–7
Gonzalez-Suarez E, Samper E, Ramirez A, et al. Increased epidermal tumors and increased skin wound healing in transgenic mice overexpressing the catalytic subunit of telomerase, mTERT, in basal keratinocytes. EMBO J 2001; 20(11): 2619–30
Bryan TM, Englezou A, Dalla-Pozza L, et al. Evidence for an alternative mechanism for maintaining telomere length in human tumors and tumorderived cell lines. Nat Med 1997; 3(11): 1271–4
Lundblad V. Telomere maintenance without telomerase. Oncogene 2002; 21(4): 522–31
Hahn WC, Stewart SA, Brooks MW, et al. Inhibition of telomerase limits the growth of human cancer cells. Nat Med 1999; 5(10): 1164–70
Teng L, Specht MC, Barden CB. et al. Antisense hTERT inhibits thyroid cancer cell growth. J Clin Endocrinol Metab 2003; 88(3): 1362–66
Zhang Y, He DM. Effect of antisense hTERT mRNA oligodoexynucleotide on telomerase activity of leukemic cells Cell Biol Int 2002; 26(5): 427–31
Kondo S, Kondo Y, Li G, et al. Targeted therapy of human malignant glioma in a mouse model by 2-5A antisense directed against telomerase RNA. Oncogene 1998; 16(25): 3323–30
Mukai S, Kondo Y, Koga S, et al. 2-5A antisense telomerase RNA therapy for intracranial malignant gliomas. Cancer Res 2000; 60(16): 4461–7
Kondo Y, Koga S, Komata T, et al. Treatment of prostate cancer in vitro and in vivo with 2-5A-anti-telomerase RNA component. Oncogene 2000; 19(18): 2205–11
Kushner DM, Paranjape JM, Bandyopadhyay B, et al. 2-5A antisense directed against telomerase RNA produces apoptosis in ovarian cancer cells. Gynecol Oncol 2000; 76(2): 183–92
Koga S, Kondo Y, Komata T, et al. Treatment of bladder cancer cells in vitro and in vivo with 2-5A antisense telomerase RNA. Gene Ther 2001; 8(8): 654–8
Vonderheide RH, Schultze JL, Anderson KS, et al. Equivalent induction of telomerase-specific cytotoxic T lymphocytes from tumor-bearing patients and healthy inderviduals. Cancer Res 2001; 23(61): 8366–70
Vonderheide RH, Anderson KS, Hahn WC, et al. Characterization of HLA- A3-restricted cytotoxic T lymphocytes reactive against the widely expressed tumor antigen telomerase. Clin Cancer Res 2001; 7(11): 3343–8
Nair SK, Heiser A, Boczkowski D, et al. Induction of cytotoxic T cell responses and tumor immunity against unrelated tumors using telomerase reverse transcriptase RNA transfected dendritic cells. Nat Med 2000; 6(9): 1011–7
Minev B, Hipp J, Firat H, et al. Cytotoxic T cell immunity against telomerase reverse transcriptase in humans. Proc Natl Acad Sci U S A 2000; 97(9): 4796–801
Yokoyama Y, Wan X, Shinohara A, et al. Hammerhead ribozymes to modulate telomerase activity of endometrial carcinoma cells. Hum Cell 2001; 14(3): 223–31
Strahl C, Blackburn EH. Effects of reverse transcriptase inhibitors on telomere length and telomerase activity in two immortalized human cell lines. Mol Cell Biol 1996; 16(1): 53–65
Sun D, Thompson B, Cathers BE, et al. Inhibition of human telomerase by a Gquadruplex-interactive compound. J Med Chem 1997; 40(14): 2113–6
Gu J, Kagawa S, Takakura M, et al. Tumor-specific transgene expression from the human telomerase reverse transcriptase promoter enables targeting of the therapeutic effects of the Bax gene to cancers. Cancer Res 2000; 60(19): 5359–64
Komata T, Kondo Y, Kanzawa T, et al. Treatment of malignant glioma cells with the transfer of constitutively active caspase-6 using the human telomerase catalytic subunit (human telomerase reverse transcriptase) gene promoter. Cancer Res 2001; 61(15): 5796–802
Koga S, Hirohata S, Kondo Y, et al. A novel tel omerase-specific gene therapy: gene transfer of caspase-8 utilizing the human telomerase catalytic subunit gene promoter. Hum Gene Ther 2000; 11(10): 1397–406
Kosciolek BA, Kalantidis K, Tabler M, et al. Inhibition of telomerase activity in human cancer cells by RNA interference. Mol Cancer Ther 2003; 2(3): 209–16
Khatsenko O, Morgan R, Truong L, et al. Absorption of antisense oligonucleotides in rat intestine: effect of chemistry and length. Antisense Nucleic Acid Drug Dev 2000; 10(1): 35–44
Banerjee D.Genasense(GentaInc).Curr Opin Investig Drugs 2001; 2(4): 574–80
Mergny JL, Helene C.G-quadruplex DNA: a target for drug design. Nat Med 1998; 4(12): 1366–7
Vonderheide RH. Telomerase as a universal tumor-associated antigen for cancer immunotherapy. Oncogene 2002; 21(4): 674–9
Lee KH, Rudolph KL, Ju YJ, et al. Telomere dysfunction alters the chemotherapeutic profile of transformed cells. Proc Natl Acad Sci U S A 2001; 98(6): 3381–6
Wong KK, Chang S, Weiler SR, et al. Telomere dysfunction impairs DNA repair and enhances sensitivity to ionizing radiation. Nat Genet 2000; 26(1): 85–8
Latre L, Tusell L, Martin M, et al. Shortened telomeres join to DNA breaks interfering with their correct repair. Exp Cell Res 2003; 287(2): 282–8
Hannon GJ. RNA interference. Nature 2002; 418(6894): 244–51
Hutvagner G, Zamore PD. RNAi: nature abhors a double-strand. Curr Opin Genet Dev 2002; 12(2): 225–32
Sharp PA. RNA interference: 2001. Genes Dev 2001; 15(5): 485–90
Elbashir SM, Harborth J, Lendeckel W, et al. Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature 2001; 411(6836): 494–8
Caplen NJ, Parrish S, Imani F, et al. Specific inhibition of gene expression by small double-stranded RNAs in invertebrate and vertebrate systems. Proc Natl Acad Sci U S A 2001; 98(17): 9742–70
Billy E, Brondani V, Zhang H, et al. Specific interference with gene expression induced by long, double-stranded RNA in mouse embryonal teratocarcinoma cell lines. Proc Natl Acad Sci U S A 2001; 98(25): 14428–33
Yang S, Tutton S, Pierce E, et al. Specific double-stranded RNA interference in undifferentiated mouse embryonic stem cells. Mol Cell Biol 2001; 21(22): 7807–16
McManus MT, Sharp PA. Gene silencing in mammals by small interfering RNAs. Nat Rev Genet 2002; 3(10): 737–47
Brummelkamp TR, Bernards R, Agami R. A system for stable expression of short interfering RNAs in mammalian cells. Science 2002; 296(5567): 550–3
Brummelkamp TR, Bernards R, Agami R. Stable suppression of tumorigenicity by virus-mediated RNA interference. Cancer Cell 2002; 2(3): 243–7
Acknowledgements
Our work is supported by Compagnia di San Paolo and Fondi d’Ateneo, Università Cattolica. The authors declare that they have no competing financial interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Falchetti, M.L., Pallini, R. & Levi, A. Telomerase and Cancer. Am J Cancer 3, 1–11 (2004). https://doi.org/10.2165/00024669-200403010-00001
Published:
Issue Date:
DOI: https://doi.org/10.2165/00024669-200403010-00001