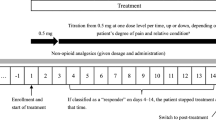
Abstract
Fentanyl is a synthetic opioid agonist which interacts primarily with the µ-opioid receptor. The low molecular weight, high potency and lipid solubility of fentanyl make it suitable for delivery by the transdermal therapeutic system. These patches are designed to deliver fentanyl at a constant rate (25, 50, 75 and 100 µg/h), and require replacement every 3 days.
Data from randomized, nonblind trials suggest that transdermal fentanyl is as effective as sustained-release oral morphine in the treatment of chronic cancer pain, as reported by patients using visual and numerical analogue scales as well as verbal description scales. No obvious differences in health-related quality of life were found in patients with chronic cancer pain when comparing transdermal fentanyl with sustained-release oral morphine. Nevertheless, significantly more patients expressed a preference for transdermal fentanyl than for sustained-release oral morphine after a randomized, nonblind, crossover trial.
Because of the formation of a fentanyl depot in the skin tissue, serum fentanyl concentrations increase gradually following initial application, generally levelling off between 12 and 24 hours. Thereafter, they remain relatively constant, with some fluctuation, for the remainder of the 72-hour application period. Once achieved, steady-state plasma fentanyl concentrations can be maintained for as long as the patches are renewed.
The most frequently observed adverse events during transdermal fentanyl administration (as with other opioid agonists) included vomiting, nausea and constipation. Data from nonblind, randomized trials suggest that constipation occurs less frequently in patients receiving transdermal fentanyl than in those given sustained-release oral morphine. The most serious adverse event reported in US premarketing trials was hypoventilation, which occurred with an incidence of approximately 2%. Adverse reactions related to skin and appendages (i.e. rash and application site reactions - erythema, papules, itching and oedema) were reported in 153 patients with cancer at a frequency between 1 and 3%.
Conclusion: Transdermal fentanyl is a useful opioid-agonist for the treatment of moderate to severe chronic cancer pain. The advantages of transdermal fentanyl include ease of administration and the 3-day application interval. These factors coupled with a lower incidence of constipation are likely to contribute to the reported patient preference of transdermal fentanyl over sustained-release oral morphine.
Similar content being viewed by others
References
Villiger JW, Ray LJ, Taylor KM. Characteristics of [3H]fentanyl binding to the opiate receptor. Neuropharmacology 1983; 22(4): 447–52
Donner B, Zenz M. Transdermal fentanyl: a new step on the therapeutic ladder. Anticancer Drugs 1995 Apr; 6: 39–43
VonCube B, Teschemacher-Herz HJ, Hess R, et al. Permeation morphinartig wirksamer Substanzen an den Ort der antinociceptiven Wirkung im Gehirn in Abhangigkeit van ihrer Lipoidloslichkeit nach intravenoser und nach intraventrikularer Applikation. Naunyn Schmiedebergs Arch Pharmacol 1970; 265: 455–73
Herz A, Albus K, Metys J, et al. On the central sites for the antinociceptive action of morphine and fentanyl. Neuropharmacology 1970; 98: 539–51
Janssen Pharmaceutica. Durogesic (Fentanyl transdermal system) full prescribing information (US). 2001
Tabatabai M, Kitahata LM, Collins JG, et al. Disruption of the rhythmic activity of the medullary inspiratory neurons and phrenic nerve by fentanyl and reversal with nalbuphine. Anesthesiology 1989; 70: 489–95
Chen S-W, Maguire PA, Davies MF, et al. Evidence for µ l-opioid receptor involvement in fentanyl-mediated respiratory depression. Eur J Pharmacol 1996; 312(2): 241–4
Basta S, Anderson DL. Mechanisms and management of constipation in the cancer patient. J Pharm Care Pain Symptom Control 1998; 6(3): 21–40
Collett B-J. Opioid tolerance: the clinical perspective. Br J Anaesth 1998 Jul; 81: 58–68
Hedner T, Cassuto J. Opioids and opioid receptors in peripheral tissues. Scand J Gastroenterol 1987; 22 Suppl. 130: 27–46
Megens AHP, Artois K, Vermeire J, et al. Comparison of the analgesic and intestinal effects of fentanyl and morphine in rats. J Pain Symptom Manage 1998; 15(4): 253–7
Ahmedzai S, Brooks D. Transdermal fentanyl versus sustained-release oral morphine in cancer pain: preference, efficacy, and quality of life. TTS-Fentanyl Comparative Trial Group. J Pain Symptom Manage 1997 May; 13(5): 254–61
Allan L, Hays H, Jensen NH, et al. Randomised crossover trial of transdermal fentanyl and sustained release oral morphine for treating chronic non-cancer pain. BMJ 2001; 322(7295): 1154–8
Rosow CE, Moss J, Philbin DM, et al. Histamine release during morphine and fentanyl anesthesia. Anesthesiology 1982; 56: 93–6
Bilfinger TV, Fimiani C, Stefano GB. Morphine’s immunoregulatory actions are not shared by fentanyl. Int J Cardiol 1998 Apr 30; 64 Suppl. 1: 61–6
Roy SD, Flynn GL. Transdermal delivery of narcotic analgesics: comparative permeabilities of narcotic analgesics through human cadaver skin. Pharm Res 1989; 6(10): 825–32
Hwang SS, Nichols KC, Southam M. Transdermal permeation: physiological and physicochemical aspects. In: Lehmann KA, Zech D, editors. Transdermal fentanyl: a new approach to prolonged pain control. 1st ed. Barlin: Springer-Verlag, 1991: 1–7
Southam MA. Transdermal fentanyl therapy: system design, pharmacokinetics and efficacy. Anticancer Drugs 1995 Apr; 6: 29–34
Varvel JR, Shafer SL, Hwang SS, et al. Absorption characteristics of transdermally administered fentanyl. Anesthesiology 1989; 70: 928–34
Mather LE. Clinical pharmacokinetics of fentanyl and its newer derivatives. Clin Pharmacokinet 1983; 8: 422–46
Gourlay GK, Kowalski SR, Plummer JL, et al. The transdermal administration of fentanyl in the treatment of postoperative pain: pharmacokinetics and pharmacodynamic effects. Pain 1989; 37: 193–202
Holley FO, van Steennis C. Postoperative analgesia with fentanyl: pharmacokinetics and pharmacodynamics of constant-rate i.v. and transdermal delivery. Br J Anaesth 1988; 60: 608–13
Labroo RB, Paine MF, Thummel KE, et al. Fentanyl metabolism by human hepatic and intestinal cytochrome P450 3A4: implications for interindividual variability in disposition, efficacy, and drug interactions. Drug Metab Dispos 1997 Sep; 25: 1072–80
Tateishi T, Krivoruk Y, Ueng YF, et al. Identification of human liver cytochrome P-450 3A4 as the enzyme responsible for fentanyl and sulfanyl N-dealkylation. Anesth Analg 1996; 82(1): 167–72
Feierman DE, Lasker JM. Metabolism of fentanyl, a synthetic opioid analgesic by human liver microsomes. Role of CYP3A4. Drug Metab Dispos 1996; 24(9): 932–9
Wong J-O, Chiu G-L, Tsao C-J, et al. Comparison of oral controlled-release morphine with transdermal fentanyl in terminal cancer pain. Acta Anaesthesiol Sin 1997 Mar; 35(1): 25–32
Kongsgaard UE, Poulain P. Transdermal fentanyl for pain control in adults with chronic cancer pain. European Journal of Pain 1998; 2: 53–62
Grond S, Radbruch L, Lehmann KA. Clinical pharmacokinetics of transdermal opioids: focus on transdermal fentanyl. Clin Pharmacokinet 2000 Jan; 38: 59–89
Hanks GW, de Conno F, Cherny N, et al. Morphine and alternative opioids in cancer pain: the EAPC recommendations. Br J Cancer 2001; 84(5): 587–93
Hui Ming Chang. Cancer pain management. Med Clin North Am 1999; 83(3): 711–36
Makin MK. Strong opioids for cancer pain. J R Soc Med 2001; 94: 17–21
Payne R, Mathias SD, Pasta DJ, et al. Quality of life and cancer pain: satisfaction and side effects with transdermal fentanyl versus oral morphine. J Clin Oncol 1998 Apr; 16: 1588–93
Olkkola KT, Palkama VJ, Neuvonen PJ. Effect of itraconazole on the pharmacokinetics and pharmacodynamics of intravenous fentanyl [abstract]. Anesthesiology 1997 Sep; 87 Suppl.: 333
Janssen-Cilag. Durogesic (Transdermal Fentanyl) Fachinformation (Prescribing information for Germany). 2000
Janssen-Cilag Ltd. UK. Summary of Product Characteristics UK. 2001
Acknowledgements
The full text article in Drugs 2001; 61(15): 2289-2307 was reviewed by: M.Barnett, Coventry and University of Warwick, Warwick, England; S. Mercadante, La Maddalena Cancer Center, Palermo, Italy; L. Radbruch, University of Cologne, Cologne, Germany; F. Takeda, Saitama Cancer Center, Saitama City, Japan;D. Thorpe, The Huntsman Cancer Institute, University of Utah, Salt Lake City, Utah, USA; J. Wong, St. Martin De Porres Hospital, Chia-Yi City, Taiwan.
Author information
Authors and Affiliations
Corresponding author
Additional information
The full text of this article was published in Drugs 2001; 61(15): 2289–2307.
Rights and permissions
About this article
Cite this article
Muijsers, R.B.R., Wagstaff, A.J. Spotlight on Transdermal Fentanyl in Chronic Cancer Pain. Am J Cancer 1, 149–152 (2002). https://doi.org/10.2165/00024669-200201020-00008
Published:
Issue Date:
DOI: https://doi.org/10.2165/00024669-200201020-00008