Abstract
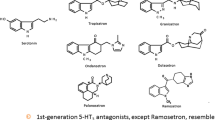
Selective serotonin 5-HT3 receptor antagonists have proven safe and effective for the management of postoperative nausea and vomiting. Dolasetron, granisetron, ondansetron and tropisetron selectively and competitively bind to 5-HT3 receptors, blocking serotonin binding at vagal afferents in the gut and in the regions of the CNS involved in emesis, including the chemoreceptor trigger zone and the nucleus tractus solitarii. Despite their shared mechanism of action, 5-HT3 receptor antagonists have different chemical structures and exhibit differences in receptor binding affinity, dose response and duration of effect. Furthermore, although dolasetron, granisetron, ondansetron and tropisetron are all extensively metabolised by the cytochrome P450 (CYP) system, different components of this system predominate in the metabolism of each of these agents. Hence, although these agents are considered equally effective in the overall population, their pharmacokinetic and pharmacodynamic differences may explain the variability in individual responses to these drugs. This review discusses the pharmacological profiles of dolasetron, granisetron, ondansetron and tropisetron, and the clinical implications of differences in their profiles.







Similar content being viewed by others
References
Herrstedt J. Development of antiemetic therapy in cancer patients. Acta Oncologica 1995; 34: 637–40
Gregory RE, Ettinger DS. 5-HT3 receptor antagonists for the prevention of chemotherapy-induced nausea and vomiting: a comparison of their pharmacology and clinical efficacy. Drugs 1998; 55: 173–89
Kovac AL. Prevention and treatment of postoperative nausea and vomiting. Drugs 2000; 59: 213–43
Sung YF. Risks and benefits of drugs used in the management of postoperative nausea and vomiting. Drug Saf 1996; 14:181–97
Naylor RJ, Inall FC. The physiology and pharmacology of postoperative nausea and vomiting. Anaesthesia 1994; 49 Suppl.: 2–5
Andrews PL, Bhandari P, Davey PT, et al. Are all 5-HT3 receptor antagonists the same? Eur J Cancer 1992; 28ASuppl.1: S2–6
Kazemi-Kjellberg F, Henzi I, Tramer MR. Treatment of established postoperative nausea and vomiting: a quantitative systematic review. BMC Anesthesiol 2001; 1(1): 2
Loewen PS, Marra CA, Zed PJ. 5-HT3 receptor antagonists vs traditional agents for the prophylaxis of postoperative nausea and vomiting. Can J Anaesth 2000; 47: 1008–18
Gregory RE, Ettinger DS. 5-HT3 receptor antagonists for the prevention of chemotherapy-induced nausea and vomiting: a comparison of their pharmacology and clinical efficacy. Drugs 1998; 55: 173–89
Sanwald P, David M, Dow J. Characterization of the cytochrome P450 enzymes involved in the in vitro metabolism of dolasetron: comparison with other indole-containing 5-HT3 antagonists. Drug Metab Dispos 1996; 24: 602–9
Higashi H, Nishi S. 5-Hydroxytryptamine receptors of visceral primary afferent neurones on rabbit nodose ganglia. J Physiol 1982; 323: 543–67
Turvill JL, Connor P, Farthing MJ. The inhibition of cholera toxin-induced 5-HT release by the 5-HT(3) receptor antagonist, granisetron, in the rat. Br J Pharmacol 2000; 130: 1031–6
Broomhead CJ. Physiology of postoperative nausea and vomiting. Br J Hosp Med 1995; 53: 327–30
ANZEMET® injection (dolasetron mesylate injection) [package insert]. Kansas City (MO): Aventis Pharmaceuticals Inc., 2002
KYTRIL® (granisetron hydrochloride) injection [package insert]. Nutley (NJ): Roche Pharmaceuticals, 2002
Pritchard JF, Bryson JC, Kernodle AE, et al. Age and gender effects on ondansetron pharmacokinetics: evaluation of healthy aged volunteers. Clin Pharmacol Ther 1992; 51: 51–5
Blower PR. 5-HT3-receptor antagonists and the cytochrome P450 system: clinical implications. Cancer J 2002; 8: 405–14
Kees F, Farber L, Bucher M, et al. Pharmacokinetics of therapeutic doses of tropisetron in healthy volunteers. Br J Clin Pharmacol 2001; 52: 705–7
ZOFRAN® (ondansetron hydrochloride) injection; ZOFRAN® (ondansetron hydrochloride) injection premixed [package insert]. Research Triangle Park (NC): GlaxoSmithKline, 2001
Novaban (tropisetron) [prescribing information, in French]; Novartis Pharmaceuticals 2001 [online]. Available from URL: http://www.novartis.nl/pdf/ib/Novaban_capsule_injectie.pdf [Accessed 2005 Jan 9]
ANZEMET® tablets (dolasetron mesylate injection) [package insert]. Kansas City (MO): Avenus Pharmaceuticals Inc., 2000
ZOFRAN® (ondansetron hydrochloride) tablets; ZOFRAN ODT® (ondansetron) orally disintegrating tablets; ZOFRAN® (ondansetron hydrochloride) oral solution [package insert]. Research Triangle Park (NC): GlaxoSmithKline, 2002
Kaiser R, Sezer O, Papies A, et al. Patient-tailored antiemetic treatment with 5-hydroxytryptamine type 3 receptor antagonists according to cytochrome P-450 2D6 genotypes. J Clin Oncol 2002; 20: 2805–11
Bloomer JC, Baldwin SJ, Smith GJ, et al. Characterisation of the cytochrome P450 enzymes involved in the in vitro metabolism of granisetron. Br J Clin Pharmacol 1994; 38: 557–66
Dixon CM, Colthup PV, Serabjit-Singh CJ, et al. Multiple forms of cytochrome P450 are involved in the metabolism of ondansetron in humans. Drug Metab Dispos 1995; 23: 1225–30
Fischer V, Baldeck JP, Tse FL. Pharmacokinetics and metabolism of the 5-hydroxytryptamine antagonist tropisetron after single oral doses in humans. Drug Metab Dispos 1992; 20: 603–7
Vickers AE, Fischer V, Connors MS, et al. Biotransformation of the antiemetic 5-HT3 antagonist tropisetron in liver and kidney slices of human, rat and dog with a comparison to in vivo. Eur J Drug Metab Pharmacokinet 1996; 21: 43–50
Fischer V, Vickers AE, Heitz F, et al. The polymorphic cytochrome P-4502D6 is involved in the metabolism of both 5-hydroxytryptamine antagonists, tropisetron and ondansetron. Drug Metab Dispos 1994; 22: 269–74
Shu Y, Cheng ZN, Liu ZQ, et al. Interindividual variations in levels and activities of cytochrome P-450 in liver microsomes of Chinese subjects. Acta Pharmacol Sin 2001; 22: 283–8
Tamminga WJ, Werner J, Oosterhuis B, et al. CYP2D6 and CYP2C19 activity in a large population of Dutch healthy volunteers: indications for oral contraceptive-related gender differences. Eur J Clin Pharmacol 1999; 55: 177–84
Transon C, Lecoeur S, Leemann T, et al. Interindividual variability in catalytic activity and immunoreactivity of three major human liver cytochrome P450 isozymes. Eur J Clin Pharmacol 1996; 51: 79–85
Sweeney BP. Why does smoking protect against PONV? Br J Anaesth 2002; 89: 810–3
van Wijngaarden I, Tulp MT, Soudijn W. The concept of selectivity in 5-HT receptor research. Eur J Pharmacol 1990; 188: 301–12
Newberry NR, Watkins CJ, Sprosen TS, et al. BRL 46470 potently antagonizes neural responses activated by 5-HT3 receptors. Neuropharmacology 1993; 32: 729–35
Cooper SM, Arnold BDC, Rapeport WG. Inhibition of 5-HT induced axon-reflex flares by BRL 43694, a novel 5-HT3 receptor antagonist. Br J Clin Pharmacol 1988; 25: 106P–7P
Upward JW, Arnold BD, Link C, et al. The clinical pharmacology of granisetron (BRL 43694), a novel specific 5-HT3 antagonist. Eur J Cancer 1990; 26Suppl. 1: S12–5
Kovac AL, O’Connor TA, Pearman MH, et al. Efficacy of repeat intravenous dosing of ondansetron in controlling postoperative nausea and vomiting: a randomized, double-blind, placebo-controlled multicenter trial. J Clin Anesth 1999; 11: 453–9
Carmichael J, Keizer HJ, Cupissol D, et al. Use of granisetron in patients refractory to previous treatment with antiemetics. Anticancer Drugs 1998; 9: 381–5
de Wit R, de Boer AC, vd Linden GH, et al. Effective cross-over to granisetron after failure to ondansetron, a randomized double blind study in patients failing ondansetron plus dexamethasone during the first 24 hours following highly emetogenic chemotherapy. Br J Cancer 2001; 85(8): 1099–101
Blower P, Aapro M. Granisetron vs ondansetron: is it a question of duration of 5-HT3 receptor blockade? Br J Cancer 2002; 86: 1662–3
Sun R, Klein KW, White PF. The effect of timing of ondansetron administration in outpatients undergoing otolaryngologic surgery. Anesth Analg 1997; 84: 331–6
Cieslak GD, Watcha MF, Phillips MB, et al. The dose-response relation and cost-effectiveness of granisetron for the prophylaxis of pediatric postoperative emesis. Anesthesiology 1996; 85: 1076–85
Hamid SK, Selby IR, Sikich N, et al. Vomiting after adenotonsillectomy in children: a comparison of ondansetron, dimenhydrinate, and placebo. Anesth Analg 1998; 86: 496–500
Wilson AJ, Diemunsch P, Lindeque BG, et al. Single-dose i.v. granisetron in the prevention of postoperative nausea and vomiting. Br J Anaesth 1996; 76: 515–8
Mikawa K, Takao Y, Nishina K, et al. The antiemetic efficacy of prophylactic granisetron in gynecologic surgery. Anesth Analg 1995; 80: 970–4
Taylor AM, Rosen M, Diemunsch PA, et al. A double-blind, parallel-group, placebo-controlled, dose-ranging, multicenter study of intravenous granisetron in the treatment of postoperative nausea and vomiting in patients undergoing surgery with general anesthesia. J Clin Anesth 1997; 9: 658–63
Gan TJ, Coop A, Chen YM, et al. Randomized, double-blind, non-inferiority study of granisetron plus dexamethasone versus ondansetron plus dexamethasone in the prevention of postoperative nausea and vomiting in patients undergoing abdominal hysterectomy [abstract]. Anesthesiology 2004, A52 [online]. Available from URL: http://www.asaabstracts.com/strands/asaabstracts/home.htm;jsessionid=2513A11691ED091791E2B749F81EBD93 [Accessed 2005 Jan 9]
Graczyk SG, McKenzie R, Kallar S, et al. Intravenous dolasetron for the prevention of postoperative nausea and vomiting after outpatient laparoscopic gynecologic surgery. Anesth Analg 1997; 84: 325–30
Gan TJ, Meyer T, Apfel CC, et al. Consensus guidelines for managing postoperative nausea and vomiting. Anesth Analg 2003; 97: 62–71
Kranke P, Eberhart LH, Apfel CC, et al. Tropisetron for prevention of postoperative nausea and vomiting: a quantitative systematic review [in German]. Anaesthesist 2002; 51: 805–14
Alon E, Buchser E, Herrera E, et al. Tropisetron for treating established postoperative nausea and vomiting: a randomized, double-blind, placebo-controlled study. Anesth Analg 1998; 86: 617–23
Tramer MR. A rational approach to the control of postoperative nausea and vomiting: evidence from systematic reviews: part I. Efficacy and harm of antiemetic interventions, and methodological issues. Acta Anaesthesiol Scand 2001; 45: 4–13
Tramer MR, Reynolds DJ, Moore RA, et al. Efficacy, dose-response, and safety of ondansetron in prevention of postoperative nausea and vomiting: a quantitative systematic review of randomized placebo-controlled trials. Anesthesiology 1997; 87: 1277–89
Henzi I, Walder B, Tramer MR. Metoclopramide in the prevention of postoperative nausea and vomiting: a quantitative systematic review of randomized, placebo-controlled studies. Br J Anaesth 1999; 83: 761–71
Korttila K, Clergue F, Leeser J, et al. Intravenous dolasetron and ondansetron in prevention of postoperative nausea and vomiting: a multicenter, double-blind, placebo-controlled study. Acta Anaesthesiol Scand 1997; 41: 914–22
Naguib M, el Bakry AK, Khoshim MH, et al. Prophylactic antiemetic therapy with ondansetron, tropisetron, granisetron and metoclopramide in patients undergoing laparoscopic cholecystectomy: a randomized, double-blind comparison with placebo. Can J Anaesth 1996; 43: 226–31
Walker JB. Efficacy of single-dose intravenous dolasetron versus ondansetron in the prevention of postoperative nausea and vomiting. Clin Ther 2001; 23: 932–8
Zarate E, Watcha MF, White PF, et al. A comparison of the costs and efficacy of ondansetron versus dolasetron for antiemetic prophylaxis. Anesth Analg 2000; 90: 1352–8
Roberson CR, McLeskey CH, Meyer T, et al. IV dolasetron vs ondansetron for the treatment of PONV in ambulatory surgery patients [abstract no. A-4]. American Society of Anesthesiologists Annual Meeting; 1999 Oct 11; Dallas (TX). Anesthesiology 1999; 91: 4
Scholz J, Hennes HJ, Steinfath M, et al. Tropisetron or ondansetron compared with placebo for prevention of postoperative nausea and vomiting. Eur J Anaesthesiol 1998; 15: 676–85
Tsui SL, Ng KF, Wong LC, et al. Prevention of postoperative nausea and vomiting in gynaecological laparotomies: a comparison of tropisetron and ondansetron. Anaesth Intensive Care 1999; 27: 471–6
Jokela R, Koivuranta M, Kangas-Saarela T, et al. Oral ondansetron, tropisetron or metoclopramide to prevent postoperative nausea and vomiting: a comparison in high-risk patients undergoing thyroid or parathyroid surgery. Acta Anaesthesiol Scand 2002; 46: 519–24
Keefe DL. The cardiotoxic potential of the 5-HT(3) receptor antagonist antiemetics: is there cause for concern? Oncologist 2002; 7: 65–72
Hof RP, Hof A, Novosel D, et al. Antiarrhythmic and hemodynamic effects of tropisetron in anesthetized rabbits. J Cardiovasc Pharmacol 1993; 22: 499–505
Kuryshev YA, Brown AM, Wang L, et al. Interactions of the 5-hydroxytryptamine 3 antagonist class of antiemetic drugs with human cardiac ion channels. J Pharmacol Exp Ther 2000; 295: 614–20
Scholtysik G, Imoto Y, Yatani A, et al. 5-Hydroxytryptamine antagonist ICS 205-930 blocks cardiac potassium, sodium and calcium currents. J Pharmacol Exp Ther 1988; 245: 773–8
Lischke V, Behne M, Doelken P, et al. Droperidol causes a dose-dependent prolongation of the QT interval. Anesth Analg 1994; 79: 983–6
Hunt TL, Cramer M, Shah A, et al. A double-blind, placebo-controlled, dose-ranging safety evaluation of single-dose intravenous dolasetron in healthy male volunteers. J Clin Pharmacol 1995; 35: 705–12
Hesketh P, Navari R, Grote T, et al. Double-blind, randomized comparison of the antiemetic efficacy of intravenous dolasetron mesylate and intravenous ondansetron in the prevention of acute cisplatin-induced emesis in patients with cancer: Dolasetron Comparative Chemotherapy-induced Emesis Prevention Group. J Clin Oncol 1996; 14: 2242–9
Benedict CR, Arbogast R, Martin L, et al. Single-blind study of the effects of intravenous dolasetron mesylate versus ondansetron on electrocardiographic parameters in normal volunteers. J Cardiovasc Pharmacol 1996; 28: 53–9
Boike SC, Ilson B, Zariffa N, et al. Cardiovascular effects of i.v. granisetron at two administration rates and of ondansetron in healthy adults. Am J Health Syst Pharm 1997; 54: 1172–6
Audhuy B, Cappelaere P, Martin M, et al. A double-blind, randomised comparison of the anti-emetic efficacy of two intravenous doses of dolasetron mesilate and granisetron in patients receiving high dose cisplatin chemotherapy. Eur J Cancer 1996; 32A: 807–13
Koriech OM. Fluoxetine treatment comprises the antiemetic efficacy of ondansetron in cancer patients. Clin Oncol (R Coll Radiol) 1995; 7: 371–2
De Witte JL, Schoenmaekers B, Sessler DI, et al. The analgesic efficacy of tramadol is impaired by concurrent administration of ondansetron. Anesth Analg 2001; 92: 1319–21
Arcioni R, Delia RM, Romano S, et al. Ondansetron inhibits the analgesic effects of tramadol: a possible 5-HT(3) spinal receptor involvement in acute pain in humans. Anesth Analg 2002; 94: 1553–7
Stamer UM, Stuber F. Analgesic efficacy of tramadol if coadministered with ondansetron [letter]. Anesth Analg 2001; 93: 1626
Acknowledgements
This work was supported by Roche Laboratories, Inc., Nutley, New Jersey, with editorial support from Accel Healthcare Communications, Inc., New York, New York. The authors have previously received grant support from GlaxoSmithKline, Abbott, Roche Laboratories, Inc., Merck, Pfizer and Baxter, Inc.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gan, T.J. Selective Serotonin 5-HT3 Receptor Antagonists for Postoperative Nausea and Vomiting. CNS Drugs 19, 225–238 (2005). https://doi.org/10.2165/00023210-200519030-00004
Published:
Issue Date:
DOI: https://doi.org/10.2165/00023210-200519030-00004