Abstract
Introduction: The LIFE (Losartan Intervention For Endpoint reduction in hypertension) study demonstrated a 13% relative risk reduction in the primary composite endpoint (myocardial infarction, stroke or death) for patients with hypertension and electrocardiographically diagnosed left ventricular hypertrophy (LVH) treated with losartan compared with atenolol. Losartan recipients also had a 25% relative risk reduction for stroke compared with atenolol recipients. Incorporating the results found in the LIFE study into an economic model, an incremental cost-effectiveness analysis was performed comparing losartan with atenolol in the treatment of 67-year old patients with hypertension and LVH.
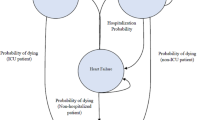
Methods: A Markov state transition model, based on published results of the LIFE trial (mean follow-up of 4.8 years), was utilised to extrapolate the outcomes observed in this trial to the patients’ lifetime. Utility estimates for the associated health states were obtained from various published sources. Lifetime treatment costs were calculated adopting a societal perspective. Both costs and benefits were discounted and incremental cost-effectiveness ratios (ICERs) were estimated. One-way and probabilistic sensitivity analyses were performed.
Results: The estimated ICER for losartan versus atenolol was $Can1337 per QALY gained ($Can1 = $US0.75, 2002 values). This ICER was robust to extensive sensitivity analysis, demonstrating a 95% probability that the ICER would be <$Can20 000 per QALY gained.
Conclusion: From a Canadian societal perspective, losartan appears to be a cost-effective alternative to atenolol in patients with hypertension and LVH. The estimated ICERs, including the sensitivity analyses, were within the range of cost-effectiveness ratios for various currently funded interventions and drugs in developed countries.









Similar content being viewed by others
References
Murray CJLM, Lopez AD. Evidence-based health policy-lessons from the global burden of disease study. Science 1996; 274: 740–743
Joffres MR, Ghadirian P, Fodor JG, et al. Awareness, treatment and control of hypertension in Canada. Am J Hypertens 1997; 10 (10 Pt 1): 1097–1102
Hansson L, Lindholm LH, Ekbom T, et al. Randomised trial of old and new anti-hypertensive drugs in elderly patients: cardiovascular mortality and morbidity, the Swedish trial in old patients with Hypertension-2 study. Lancet 1999; 354: 1751–1756
Neal B, MacMahon S, Chapman N. Fifects of ACE inhibitors, calcium antagonist, and other blood-pressure-Iowering drugs: results of prospectively designed overviews of randomized trials. Blood Pressure Lowering Treatment Trialists’ Collaboration. Lancet 2000; 356: 1955–1964
Yusuf S, Sleight P, Pogue J, et al. Fifects of an angiotensin-converting enzyme inhibitor, ramipril, on cardiovascular events in high-risk patients: the Heart Outcomes Prevention Evaluation Study investigators. N Engl J Med 2000; 342: 145–153
Davis BR, Cutler JA, Gordon DJ, et al. Rationale and design for the Antihypertensive and Lipid Lowering Treatment to Prevent Heart Attack Trial (ALLHAT). ALLHAT Research Group. Am J Hypertens 1996; 9: 342–360
Dahlof B, Devereux RB, Kjeldsen SE, et al. Cardiovascular morbidity and mortality in the Losartan Intervention for End point reduction in hypertension study (UFE): a randomised trial against atenolol. Lancet 2002; 359: 995–1003
Rundek T, Mast H, Hartmann A, et al. Predictors of resource use after acute hospitalization the northern Manhattan stroke study. Neurology 2000; 55: 1180–1187
Tengs JO, Lin TH. A meta-analysis of quality-of-life estimates for stroke. Pharmacoeconomics 2003; 21 (3): 191–200
Sinclair SE, Frighetto L, Loewen PS, et al. Cost utility analysis of tissue plasminogen activator therapy for acute ischaemic stroke: a Canadian healthcare perspective. Pharmacoeconomics 2001; 19 (9): 927–936
Stein JD, Brown GC, Brown MM, et al. The quality of life of patients with hypertension. J Clin Hypertens 2002 May–Jun; 4 (3), 181–188
Gore JM, Granger CB, Maarten SL, et al. Stroke afterthrorrbolysis: mortality and functional outcomes in the GUSTO-I trial. Circulation 1995; 92: 2811–2818
Kolominsky-Rabas PL, Sarti C, Heuschmann PU, et al. A prospective community-based study of stroke in Germany: the Eclangen Stroke Project. Stroke 1998; 29 (12): 2501–2506
Tu N, Austin PC, Walld R, et al. Development and validation of the Ontario acute myocardial infarction mortality prediction rules. J Am Coll Cardiol 2001; 37 (4): 992–997
Statistics Canada. Life table [online]. Available from URL: http://www.statcan.ca/english/freepub/84-537-XIE/free.htm [Accessed 2003 Nov]
Government of British Columbia. Pharmacare low cost alternative reference drug program booklet. Version 1.1. 2003 Jan 15 [online]. Available from URL: http://www.healthservices.gov.bc.ca/pharme/outgoing/lca-rdpbooklet2004-11-24.pdf [Accessed 2003 Nov]
British Columbia Ministry of Health Services. Pharmacare Newsletter. January 23, 2002 [online]. Available from URL: http://www.healthservices.gov.bc.ca/pharme/newsletter/02001news.pdf [Accessed 2003 Nov]
British Columbia Ministry of Health Service. Pharmacare maximum days’ supply policy [online]. Available from URL: http://www.hlth.gov.bc.ca/pharme/generalinfo/maxdays.html [Accessed 2003 Nov]
Management Information Systems Project Steering Committee. Guidelines for management information systems in Canadian healthcare facilities. Ottawa (ON): The MIS Group, 2002
British Columbia Medical Association. BCMA guide to fees. 2002 [online]. Available from URL: http://www.bcma.org/public/news_publications/publications/fee_guide.htm [Accessed 2003 Nov]
Canadian Institute for Health Information [online]. Available from URL: http://secure.cihi.ca/cihiweb/dispPagejsp?cw_page=statistics_topic_e [Accessed 2003 Nov]
Tung CY, Granger CB, Sloan MA, et al. Effects of stroke on medical resource and costs in acute myocardial infarction. Circulation 1999 Jan 26; 99 (3): 370–376
Canadian Institute for Health Information [online]. Available from http://secure.cihi.ca/cihiweb/dispPage.jsp?cw_page=statistics_topic_e [Accessed 2003 Nov]
Adams HP, Adams RJ, Brott T, et al. Guidelines for the early management of patients with ischemic stroke: a scientific statement from the Stroke Council of the American Stroke Association. Stroke 2003; 42: 1056–1083
Hollander MJ. Unfinished business: the case for chronic home care services, a policy paper. Victoria (BC): Hollander Analytical Services Ltd, 2003 Aug
Dickstein K, Kjekshus J. Comparison of baseline data, initial course and management: losartan vs captopril following acute myocardial infarction. Am J Cardiol 2001; 87: 766–771
Hart RG, Halpern JL. Atrial fibrillation and stroke: concepts and controversies. Stroke 2001; 32: 803–808
Drummond MF, O’Brien BJ, Stoddart GL, et al. Methods for the economic evaluation of health care programmes. 2nd ed. Toronto: University Oxford Press, 1997: 164
O’Brien B. Economic evaluation of pharmaceuticals: Frankenstein’s monster or vampire of trials? Med Care 1996; 34 (12 Suppl.): DS99–DS108
Product monograph. Cozaar® losartan potassium tablets. Whitehouse Station (NJ): Merck & Co Inc [online]. Available from URL: http://www.merckfrosst.com/e/cozaar/home.html [Accessed 2003 Nov]
Beck JR, Pauker SG. The Markov process in medical prognosis. Med Decis Making 1983; 3: 419–458
Sonnenberg FA, Beck JR. Markov models in medical decision making: a practical guide. Med Decis Making 1993; 13: 322–338
Naimark D, Krahn MD, Naglie G, et al. Primer on medical decision analysis: part 5. Working with Markov processes. Med Decis Making 1997; 17: 152–159
Kemeny JG, Snell JL. Finite Markov chains. New York: Springer-Verlag, 1976
Palepu A, Tyndall MW, Leon H, et al. Hospital utilization and costs in a cohort of injection drug users. CMAJ 2001; 165 (4): 415–420
Bjorholt I, Andersson FL, Kahan T, et al. The cost-effectiveness of ramipril in the treatment of patients at high risk of cardiovascular events. J Intern Med 2002; 251 (6): 508–517
Statistics Canada. Health and personal care consumer price index. Series V735518, Table no. 3260001 [online]. Available from URL: www.statcan.ca [Accessed 2003 Nov]
Goldstein LB, Adams R, Becker K, et al. Primary prevention of ischemic stroke: a statement for healthcare professionals from the Stroke Council of the American Heart Association. Circulation 2001 Jan 2; 103 (1): 163–182
Briggs AH, O’Brien BJ, Blackbouse G. Thinking outside of the box. Annu Rev Public Health 2002; 23: 377–401
Julius S, Kjeldsen SE, Weber M, et al. Outcomes in hypertensive patients at high risk treated with regimens based on valsartan or amlodipine: the VALUE randomized trial. Lancet 2004; 363 (9426): 2022–2031
Lewis EJ, Hunsicker LG, Clarke WR, et al. Renoprotective effect of the angiotensin-receptor antagonist irbesartan in patients with nephropathy due to type 2 diabetes. N Engl J Med 2001; 345 (12): 851–860
Parving HR, Lehnert H, Brochner-Mortensen J, et al. The effect of irbesartan on the development of diabetic nephropathy in patients with type 2 diabetes. N Engl J Med 2001; 345 (12): 870–878
Brenner BM, Cooper ME, de Zeeuw D, et al. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med 2001; 345 (12): 861–869
Kassler-Taub K, Littlejohn T, Elliott W, et al. Comparative efficacy of two angiotensin II receptor antagonists, irbesartan and losartan in mild-to-moderate hypertension: irbesartan/losartan study investigators. Am J Hypertens 1998; 11 (4): 445–453
Mallion J, Siche J, Lacourciere Y. AABPM comparison of the antihypertensive profiles of the selective angiotensin II anatagonists telmisartan and losartan in patients with mild to moderate hypertension. J Hum Hypertens 1999; 13 (10): 657–664
Anderson OK, Neldom S. The antihypertensive effect and tolerability of candesartan cilexetil, a new generation angiotensin II antagonist, in comparison with losartan. Blood Press 1998; 7 (1), 53–59
Hedner T, Oparil S, Rasmussen K, et al. A comparison of the angiotensin II antagonists valsartan and losartan in the treatment of essential hypertension. Am J Hypertens 1999; 12 (4): 414–417
Johannesson M. The cost-effectiveness of the switch towards more expensive antihypertensive drugs. Health Policy 1994; 28 (1), 1–13
Laupacis A, Feeny D, Detsky AS, et al. How attractive does a new technology have to be to warrant adoption and utilization: tentative guidelines for using clinical and economic evaluations. CMAJ 1992; 46: 473–481
Palmer AJ, Annemans L, Roze S, et al. Cost-effectiveness of early irbesartan treatment versus control (standard antihypertensive medications excluding ACE inhibitors, other angiotensin-2 receptor antagonists, and dihydropyridine calcium channel blockers) or late irbesartan treatment in patients with type 2 diabetes, hypertension, and renal disease. Diabetes Care 2004; 27 (8): 1897–1903
Nordmann AJ, Krahn M, Logan AG, et al. The cost effectiveness of ACE inhibitors as first-line antihypertensive therapy. Pharmacoeconomics 2003; 21 (8): 573–585
Kalra L, Evans A, Perez I, et al. A randomized controlled comparison of alternative strategies in stroke care. Health Technol Assess 2005; 9 (18): 1–94
van Exel J, Koopmanschap MA, van Wijngaarden IDH. Costs of stroke and stroke services: determinants of patient costs and a comparison of costs of regular care and care organized in stroke services. Cost Eff Resour Alloc 2003; 1: 2–11
Hamon M, Wiedermann CJ, Poole-Wilson PA, et al. Losartan for cardiovascular disease in patients with and without diabetes in the LIFE study [correspondence]. The Lancet 2002; 359 (9324): 2199–2204
Carlberg B, Samuelsson O, Lindholm LH. Atenolol in hypertension: is it a wise choice? Lancet 2004; 364: 1684–1689
Klingbeil AU, Schneider M, Martus P, et al. A meta-analysis of the effects of treatment on left ventricular mass in essential hypertension. Am J Med 2003; 115: 41–46
PROGRESS Collaborative Group. Randomised trial of a perindopril-based blood-pressure-lowering regimen among 6105 individuals with previous stroke or transient ischaemic attack. Lancet 2001; 358: 1033–1041
The Canadian hypertension education program recommendations [online]. Available from URL: http://www.cmaj.ca [Accessed 2003 Nov]
Acknowledgements
Marc Brisson is an employee of Merck Frosst, Canada. The authors have no other conflicts of interest to report. Unconditional funding for this research was provided by Merck Frosst, Canada.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Anis, A.H., Sun, H., Singh, S. et al. A Cost-Utility Analysis of Losartan versus Atenolol in the Treatment of Hypertension with Left Ventricular Hypertrophy. Pharmacoeconomics 24, 387–400 (2006). https://doi.org/10.2165/00019053-200624040-00008
Published:
Issue Date:
DOI: https://doi.org/10.2165/00019053-200624040-00008