Summary
Recent epidemiological studies indicate that individuals who maintain a physically active lifestyle are much less likely to develop impaired glucose tolerance and non-insulin-dependent diabetes mellitus (NIDDM). Moreover, it was found that the protective effect of physical activity was strongest for individuals at highest risk of developing NIDDM. Reducing the risk of insulin resistance and NIDDM by regularly performed exercise is also supported by several aging studies. It has been found that older individuals who vigorously train on a regular basis exhibit a greater glucose tolerance and a lower insulin response to a glucose challenge than sedentary individuals of similar age and weight.
While the evidence is substantial that aerobic exercise training can reduce the risk of impaired glucose tolerance and NIDDM, the evidence that exercise training is beneficial in the treatment of NIDDM is not particularly strong. Many of the early studies investigating the effects of exercise training on NIDDM could not demonstrate improvements in fasting plasma glucose and insulin levels, or glucose tolerance. The adequacy of the training programmes in many of these studies, however, is questionable. More recent studies using prolonged, vigorous exercise-training protocols have produced more favourable results.
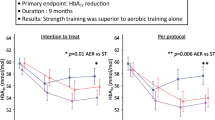
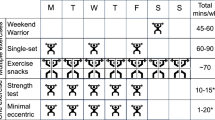
There are several important adaptations to exercise training that may be beneficial in the prevention and treatment of insulin resistance, impaired glucose tolerance and NIDDM. An increase in abdominal fat accumulation and loss of muscle mass are highly associated with the development of insulin resistance. Exercise training results in preferential loss of fat from the central regions of the body and should therefore contribute significantly in preventing or alleviating insulin resistance due to its development. Likewise, exercise training can prevent muscle atrophy and stimulate muscle development. Several months of weight training has been found to significantly lower the insulin response to a glucose challenge without affecting glucose tolerance, and to increase the rate of glucose clearance during a euglycaemic clamp.
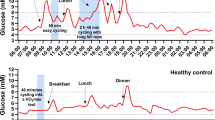
Muscle glucose uptake is equal to the product of the arteriovenous glucose difference and the rate of glucose delivery or muscle blood flow. While it has been known for many years that insulin will accelerate blood glucose extraction by insulin-sensitive peripheral tissues, recent evidence suggests that it can also acutely vasodilate skeletal muscle and increase muscle blood flow in a dose-dependent manner. A reduced ability of insulin to stimulate muscle blood flow is a characteristic of insulin-resistant obese individuals and individuals with NIDDM. Exercise training, however, has been found to help alleviate this problem, and substantially improve the control of insulin over blood glucose.
Improvements in insulin resistance and glucose tolerance with exercise training are highly related to an increased skeletal muscle insulin action. This increased insulin action is associated with an increase in the insulin-regulatable glucose transporters, GLUT4, and enzymes responsible for the phosphorylation, storage and oxidation of glucose. Changes in muscle morphology may also be important following training. With exercise training there is an increase in the conversion of fast twitch glycolytic IIb fibres to fast twitch oxidative IIa fibres, as well as an increase in capillary density. IIa fibres have a greater capillary density and are more insulin-sensitive and -responsive than IIb fibres. Evidence has been provided that morphological changes in muscle, particularly the capillary density of the muscle, are associated with changes in fasting insulin levels and glucose tolerance. Furthermore, significant correlations between glucose clearance, muscle capillary density and fibre type have been found in humans during a euglycaemic clamp.
Exercise training may also improve control over hepatic glucose production by increasing the control of insulin over blood free fatty acid (FFA) levels. An elevation in FFA levels, which is associated with obesity and NIDDM, stimulates hepatic gluconeogenesis which in turn stimulates hepatic glucose production. An increase in blood FFA levels also inhibits skeletal muscle uptake and storage. Therefore, an increase in control over blood FFA levels would serve to increase peripheral glucose clearance as well as reduce hepatic glucose production.
Similar content being viewed by others
References
Baron AD, Brechtel G, Wallace P, et al. Rates and tissue sites of non-insulin- and insulin-mediated glucose uptake in humans. Am J Physiol 1988; 255 (6 Pt 1): E769–E774
DeFronzo RA, Jacot E, Jequir E, et al. The effect of insulin on the disposal of intravenous glucose. Diabetes 1981; 30: 1000–7
Katz LD, Glickman MG, Rapoport S, et al. Splanchnic and peripheral disposal of oral glucose in man. Diabetes 1983; 32: 675–9
Kahn CR. Insulin resistance, insulin insensitivity, and insulin unresponsiveness: a necessary distinction. Metabolism 1978; 27Suppl. 2: 1893–902
National Diabetes Data Group. Classification and diagnosis of diabetes and other categories of glucose intolerance. Diabetes 1979; 28: 1039–57
World Health Organization Expert Committee. Second report on diabetes mellitus. Geneva: WHO, 1980. Technical report series no. 646
Everthart J, Knowler WC, Bennett PH. Incidence and risk factors for non-insulin-dependent diabetes. In: Harris MI, Hamman RF, editors. Diabetes in America. Washington, D.C.: National Diabetes Data Group, US Govt Printing Office, 1985. Report no. USDHHS-NIH-85-1468: 1–35
Palumbo PJ, Elveback LR, Chu CP, et al. Diabetes mellitus: incidence, prevalence, survivorship and causes of death in Rochester, Minnesota, 1945–1970. Diabetes 1976; 25: 566–73
Lipman RL, Raskin P, Love T, et al. Glucose intolerance during decreased physical activity in man. Diabetes 1972; 21: 101–7
Myllynen P, Koivisto VA, Nikkila EA. Glucose intolerance and insulin resistance accompany immobilization. Acta Med Scand 1987; 222: 75–81
Misbin RI, Moffa AM, Kappy MS. Insulin binding to monocytes in obese patients treated with carbohydrate restriction and changes in physical activity. J Clin Endocrinol Metab 1983; 56: 273–8
Lipman RL, Schnure JJ, Bradley EM, et al. Impairment of peripheral glucose utilization in normal subjects by prolonged bed rest. J Lab Clin Med 1970; 76: 221–30
Mikines KJ, Richter EA, Dela F, et al. Seven days of bed rest decrease insulin action on glucose uptake in leg and whole body. J Appl Physiol 1991; 70: 1245–54
Dolkas CB, Greenleaf JE. Insulin and glucose responses during bed rest with isotonic and isometric exercise. J Appl Physiol 1977; 43: 1033–8
King H, Zimmet P, Raper L, et al. Risk factors for diabetes in three Pacific populations. Am J Epidemiol 1984; 119: 396–409
Dowse GK, Zimmet PZ, Gareeboo H, et al. Abdominal obesity and physical inactivity are risk factors for NIDDM and impaired glucose tolerance in Indian, Creole, and Chinese Mauritians. Diabetes Care 1991; 14: 271–82
Helmrich SP, Ragland DR, Leung RW, et al. Physical activity and reduced occurrence of non-insulin-dependent diabetes mellitus. N Eng J Med 1991; 325: 147–52
Manson JE, Rimm EB, Stampfer MJ, et al. Physical activity and incidence of non-insulin-dependent diabetes mellitus in women. Lancet 1991; 338: 774–8
Manson JE, Nathan DM, Krolewski AS, et al. A prospective study of exercise and incidence of diabetes among US male physicians. J Am Med Assoc 1992; 268: 63–7
Fink RI, Kolterman OG, Griffin J, et al. Mechanisms of insulin resistance in aging. J Clin Invest 1983; 71: 1523–35
Shimokata H, Muller DC, et al. Age as independent determinant of glucose tolerance. Diabetes 1991; 40: 44–51
Zavaroni I, Dall’Aglio E, Braschi F, et al. Effect of age and environmental factors on glucose tolerance and insulin secretion in a worker population. J Am Geriat Soc 1986; 34: 271–5
Seals DR, Hagberg JM, Allen WK, et al. Glucose tolerance in young and older athletes and sedentary men. J Appl Physiol 1984; 56: 1521–5
Yamanouchi K, Nakajima H, Shinozaki T, et al. Effects of daily physical activity on insulin action in the elderly. J Appl Physiol 1992; 73: 2241–5
Rogers MA, King DS, Hagberg JM, et al. Effect of 10 days of inactivity on glucose tolerance in master athletes. J Appl Physiol 1990; 68: 1833–7
Davidson PC, Shane SR, Albrink JM. Decreased glucose tolerance following a physical conditioning program. Circulation 1966; 33: 7–11
Hartley LH, Mason JW, Hogan RP, et al. Multiple hormone responses to prolonged exercise in relation to physical training. J Appl Physiol 1972; 33: 607–10
Heath GW, Gavin JR III, Hinderliter JM, et al. Effects of exercise and lack of exercise on glucose tolerance and insulin sensitivity. J Appl Physiol 1983; 55: 512–7
LeBlanc J, Nadeau A, Richard R, et al. Studies on the sparing effect of exercise on insulin requirements in human subjects. Metabolism 1981; 30: 1119–24
Lohman D, Liebold F, Heilman W, et al. Diminished insulin response in highly trained athletes. Metabolism 1978; 27: 521–4
Rodnick KJ, Haskell WL, Swislocki ALM, et al. Improved insulin action in muscle, liver, and adipose tissue in physically trained human subjects. Am J Physiol 1987; 253: E489–E495
Cüppers HJ, Erdmann D, Schubert H, et al. Glucose tolerance, serum insulin, serum lipids in athletes. In: Berger M, Christacopoulus P, Wahren J, editors. Diabetes and exercise. Bern: Han Huber, 1982: 115–65
Miller WJ, Sherman WM, Ivy JL. Effect of strength training on glucose tolerance and post-glucose insulin response. Med Sci Sports Exerc 1984; 16: 539–43
Dela F, Mikines KJ, Von Linstow M, et al. Effect of training on insulin-mediated glucose uptake in human muscle. Am J Physiol 1992; 263 (6 Pt 1): E1134–E1143
King DS, Dalsky GP, Staten MA, et al. Insulin action and secretion in endurance-trained and untrained humans. J Appl Physiol 1987; 63: 2247–52
King DS, Dalsky GP, Clutter WE, et al. Effects of exercise and lack of exercise on insulin sensitivity and responsiveness. J Appl Physiol 1988; 64: 1942–6
Mikines KJ, Sonne B, Farrell PA, et al. Effect of training on the dose-response relationship for insulin action in men. J Appl Physiol 1989; 66: 695–703
Mikines KJ, Sonne B, Tronier B, et al. Effects of acute exercise and detraining on insulin action in trained men. J Appl Physiol 1989; 66: 704–11
Kahn SE, Larson VG, Beard JC, et al. Effect of exercise on insulin action, glucose tolerance, and insulin secretion in aging. Am J Physiol 1990; 258 (6 Pt 1): E397–E943
King DS, Staten MA, Kohrt WM, et al. Insulin secretory capacity in endurance- trained and untrained young men. Am J Physiol 1990; 259 (2 Pt 1): E155–E161
Mikines KJ, Sonne B, Tronier B, et al. Effects of training and detraining on dose-response relationship between glucose and insulin secretion. Am J Physiol 1989; 256 (5 Pt 1): E588–E596
Holloszy JO, Schultz J, Kusnierkiewicz J, et al. Effects of exercise on glucose tolerance and insulin resistance. Acta Med Scand Suppl 1986; 711: 55–65
Hughes VA, Fiatarone MA, Fielding RA, et al. Exercise increases muscle GLUT-4 levels and insulin action in subjects with impaired glucose tolerance. Am J Physiol 1993; 264 (6 Pt 1): E855–E862
Dela F, Larsen JJ, Mikines KJ, et al. Insulin-stimulated muscle glucose clearance in patients with NIDDM. Diabetes 1995; 44: 1010–20
Reitman JS, Vasquez B, Klimes I, et al. Improvement of glucose homeostasis after exercise-training in non-insulin-dependent diabetes. Diabetes Care 1984; 7: 434–41
DeFronzo RA. Glucose intolerance and aging: evidence for tissue insensitivity to insulin. Diabetes 1979; 28: 1095–101
Seals DR, Hagberg JM, Hurley BF, et al. Endurance training in older men and women 1. Cardiovascular responses to exercise. J Appl Physiol 1984; 57: 1024–9
Kirwan JP, Kohrt WM, Wojta DM, et al. Endurance exercise training reduces glucose-stimulated insulin levels in 60- to 70-yr-old men and women. J Gerontol 1993; 48(3): M84–M90
Ruderman NB, Granda OP, Johansen K. The effect of physical training on glucose tolerance and plasma lipids in maturity onset diabetes. Diabetes 1979; 28: 89–92
Saltin B, Lindgarde F, Houston M, et al. Physical training and glucose tolerance in middle-aged men with clinical diabetes. Diabetes 1979; 28: 30–2
Holloszy JO, Narahara HT. Studies of tissue permeability, X: changes in permeability to 3-methylglucose associated with contraction of isolated frog muscles. J Biol Chem 1965; 240: 3492–500
Wallberg-Henriksson H, Holloszy JO. Contractile activity increases glucose uptake by muscle in severely diabetic rats. J Appl Physiol 1984; 57: 1045–9
Cartee GD, Young DA, Sleeper MD, et al. Prolonged increase in insulin-stimulated glucose transport in muscle after exercise. Am J Physiol 1989; 256 (4 Pt 1): E494–9
Garetto LP, Richter EA, Goodman MN, et al. Enhanced muscle glucose metabolism after exercise in the rat: the two phases. Am J Physiol 1984; 246 (6 Pt 1): E471–5
Bjömtorp P. Metabolic implications of body fat distribution. Diabetes Care 1991; 14: 1132–43
Kohrt WM, Kirwan JP, Staten MA, et al. Insulin resistance in aging is related to abdominal obesity. Diabetes 1993; 42: 273–81
Després J-P, Nadeau A, Bouchard C. Physical training and changes in regional adipose tissue distribution. Acta Med Scand Suppl 1988; 723: 205–12
Kohrt WM, Malley MT, Dalsky GP, et al. Body composition of healthy sedentary and trained, young and older men and women. Med Sci Sports Exerc 1992; 24: 832–7
Dengel DR, Pratley RE, Hagberg JM, et al. Distinct effects of aerobic exercise training and weight loss on glucose homeostasis in obese sedentary men. J Appl Physiol 1996; 81: 318–25
Craig BW, Everhart J, Brown R. The influence of high-resistance training on glucose tolerance in young and elderly subjects. Mech Ageing Dev 1989; 49: 147–57
Stephens JM, Pekala PH. Transcriptional repression of the GLUT4 and C/EBP genes in 3T3-L1 adipocytes by tumor necrosis factor-alpha. J Biol Chem 1991; 266: 21839–45
Hotamisligil GS, Spiegelman BM. Tumor necrosis factor a: a key component of the obesity-diabetes link. Diabetes 1994; 43: 1271–8
Fluckey JD, Hickey MS, Brambrink JK, et al. Effects of resistance exercise on glucose tolerance in normal and glucose-intolerant subjects. J Appl Physiol 1994; 77: 1087–92
Torgan CE, Jr Brozinick JT, Bank EA, et al. Exercise training and clenbuterol reduce insulin resistance of obese Zucker rats. Am J Physiol 1993; 264 (3 Pt 1): E373–E379
Edelman SV, Laakso M, Wallace P, et al. Kinetics of insulin-mediated and non-insulin-mediated glucose uptake in humans. Diabetes 1990; 39: 955–64
Laakso M, Edelman SV, Brechtel G, et al. Impaired insulin-mediated skeletal muscle blood flow in patients with NIDDM. Diabetes 1992; 41: 1076–83
Laakso M, Edelman SV, Olefsky JM, et al. Kinetics of in vivo muscle insulin-mediated glucose uptake in human obesity. Diabetes 1990; 39: 965–74
Cleroux J, Koname N, Nadeau A, et al. After-effects of exercise on regional and systemic hemodynamics in hypertension. Hypertension 1992; 19: 183–91
Gilders RM, Dudley GA. Endurance exercise training and treatment of hypertension. Sports Med 1992; 13(2): 71–7
Utriainen T, Holmäng A, Björntorp P, et al. Physical fitness, muscle morphology, and insulin-stimulated limb blood flow in normal subjects. Am J Physiol 1996; 270: E905–E911
Kono T, Barham FW. The relationship between the insulin-binding capacity of fat cells and the cellular response to insulin studies with intact and trypsin-treated fat cells. J Biol Chem 1971; 246: 6210–16
LeMarchand-Brustel Y, Jeanrenaud B, Freychet P. Insulin binding and effects in isolated soleus muscles of lean and obese mice. Am J Physiol 1978 Apr; 234: E348–58
Olefsky JM. The insulin receptor: Its role in insulin resistance in obesity and diabetes. Diabetes 1976; 25: 1154–64
Olefsky JM, Reaven GM. Decreased insulin binding to lymphocytes from diabetic patients. J Clin Invest 1974; 54: 1323–8
Crettaz M, Prentki M, Zaninettie D, et al. Insulin resistance in soleus muscle from obese Zucker rats: involvement of several defective sites. Biochem J 1980; 186: 525–34
Caro JF, Sinha MK, Raju SM, et al. Insulin receptor kinase in human skeletal muscle from obese subjects with and without noninsulin dependent diabetes. J Clin Invest 1987; 79: 1330–7
LeBlanc J, Nadeau M, Bonlay M, et al. Effects of physical training and adiposity on glucose metabolism and 125I-insulin binding. J Appl Physiol 1979; 46: 235–9
Burstein R, Polyehronakos C, Toews CJ, et al. Acute reversal of the enhanced insulin action in trained athletes: association with insulin receptor changes. Diabetes 1985; 34: 756–60
Pederson O, Beck-Nielsen H, Heding L. Increased insulin receptors after exercise in patients with insulin-dependent diabetes mellitus. N Eng J Med 1980; 302: 886–92
Craig BW, Hammons GT, Garthwaite SM, et al. Adaptations of fat cells to exercise: response of glucose uptake and oxidation to insulin. J Appl Physiol 1981; 51: 1500–6
Koivisto VA, Yki-Järvinen H. Effect of exercise on insulin binding and glucose transport in adipocytes of normal humans. J Appl Physiol 1987; 63: 1319–23
Dohm GL, Sinha MK, Caro JF. Insulin receptor binding and protein kinase activity in muscles of trained rats. Am J Physiol 1987; 252 (2 Pt 1): E170–E175
Bonen A, Tan MH, Clune P, et al. Chronic exercise increases insulin binding in muscles but not liver. Am J Physiol 1986; 251 (2 Pt 1): E196–E202
Dela F, Handberg A, Mikines KJ, et al. GLUT4 and insulin receptor binding and kinase activity in trained human muscle. J Physiol 1993; 469: 615–24
Hirshman MF, Goodyear LJ, Wardzala LJ, et al. Identification of an intracellular pool of glucose transporters from basal and insulin-stimulated rat skeletal muscle. J Biol Chem 1990; 265: 987–91
Bell GI, Kayano T, Buse JB, et al. Molecular biology of mammalian glucose transporters. Diabetes Care 1990; 13: 198–208
Klip A, Paquet MR. Glucose transport and glucose transporters in muscle and their metabolic regulation. Diabetes Care 1990; 13: 228–43
Henriksen EJ, Bourney RE, Rodnick KJ, et al. Glucose transporter protein content and glucose transport capacity in rat skeletal muscles. Am J Physiol 1990; 259 (4 Pt 1): E593–8
Kern M, Wells JA, Stephens JM, et al. Insulin responsiveness in skeletal muscle is determined by glucose transporter (GLUT4) protein level. Biochem J 1990; 270: 397–400
Ploug T, Stallknecht BM, Pedersen O, et al. Effect of endurance training on glucose transport capacity and glucose transporter expression in rat skeletal muscle. Am J Physiol 1990; 259 (6 Pt 1): E778–86
Banks EA, Jr Brozinick JT, Yaspelkis BB III, et al. Muscle glucose transport, GLUT-4 content, and degree of exercise training in obese Zucker rats. Am J Physiol 1992; 263 (5 Pt 1): E1010–E1015
Phillips SM, Han XX, Green HJ, et al. Increments in skeletal muscle GLUT-1 and GLUT-4 after endurance training in humans. Am J Physiol 1996; 270 (3 Pt 1): E451–E462
Houmard JA, Shinebarger MH, Dolan PL, et al. Exercise training increases GLUT-4 protein concentration in previously sedentary middle-aged men. Am J Physiol 1993; 264 (6 Pt 1): E896–E901
Dela F, Ploug T, Handberg A, et al. Physical training increases muscle GLUT4 protein and mRNA in patients with NIDDM. Diabetes 1994; 43: 862–5
Goodyear LJ, Hirshman MF, Valyou PM, et al. Glucose transporter number, function, and subcellular distribution in rat skeletal muscle after exercise training. Diabetes 1992; 41: 1091–9
Brozinick JTJr, Etgen GJ Jr, YaspelkisIII BB, et al. Effects of exercise training on muscle GLUT-4 protein content and translocation in obese Zucker rats. Am J Physiol 1993; 265 (3 Pt 1): E419–E427
Hardin DS, Azzarelli B, Edwards J, et al. Mechanisms of enhanced insulin sensitivity in endurance-trained athletes: effects on blood flow and differential expression of GLUT4 in skeletal muscles. J Clin Endocrinol Met 1995; 80: 2437–46
Ebeling P, Bourey R, Koranyi L, et al. Mechanism of enhanced insulin sensitivity in athletes. Increased blood flow, muscle glucose transport protein (GLUT4) concentration, and glycogen synthase activity. J Clin Invest 1993; 92: 1623–31
Garvey WT, Maianu L, Hancock JA, et al. Gene expression of GLUT4 in skeletal muscle from insulin-resistant patients with obesity, IGT, GDM, and NIDDM. Diabetes 1992; 41: 465–75
Lund S, Vestergaad H, Andersen PH, et al. GLUT-4 content in plasma membrane of muscle from patients with non-insulin-dependent diabetes mellitus. Am J Physiol 1993; 265 (6 Pt 1): E889–E897
Ivy JL, Holloszy JO. Persistent increase in glucose uptake by rat skeletal muscle following exercise. Am J Physiol 1981; 241(5): C200–C203
Lillioja S, Matt DM, Zawadzki JK, et al. Glucose storage is a major determinant of in vivo ‘insulin resistance’ in subjects with normal glucose tolerance. J Clin Endocrinol Met 1986; 62: 922–7
Kelley DE, Mokan M, Mandarino LJ. Intracellular defects in glucose metabolism in obese patients with NIDDM. Diabetes 1992; 41: 698–706
Schalin-Jäntti C, Härkönen M, Groop LC. Impaired activation of glycogen synthase in people at increased risk for developing NIDDM. Diabetes 1992; 41: 598–604
Holloszy JO. Adaptation of skeletal muscle to endurance exercise. Med Sci Sports 1975; 7: 155–64
Saltin B, Henriksson J, Nygaard E, et al. Fiber types and metabolic potentials of skeletal muscles in sedentary man and endurance runners. Ann NY Acad Sci 1977; 301: 3–29
Ivy JL, Jr Brozinick JT, Torgan CE, et al. Skeletal muscle glucose transport in obese Zucker rats after exercise training. J Appl Physiol 1989; 66: 2635–41
James DE, Kraegen EW, Chirholm DJ. Effects of exercise training on in vivo insulin action in individual tissues of the rat. J Clin Invest 19985; 76: 657–62
Richter EA, Garetto LP, Goodman MN, et al. Muscle glucose metabolism following exercise in the rat. Increased insulin sensitivity. J Clin Invest 1982; 69: 785–93
Holm G, Krotkiewski M. Potential importance of the muscles for the development of insulin resistance in obesity. Acta Med Scand Suppl 1988; 723: 95–101
Krotkiewski M, Bylund-Fallenius AC, Holm J, et al. Relationship between muscle morphology and metabolism in obese women: the effects of long-term physical training. Euro J Clin Invest 1983; 13: 5–12
Lillioja S, Young AA, Cutler CL, et al. Skeletal muscle capillary density and fiber type are possible determinants of in vivo insulin resistance in man. J Clin Invest 1987; 80: 415–24
Marin P, Andersson B, Krotkiewski M, et al. Muscle fiber composition and capillary density in women and men with NIDDM. Diabetes Care 1994; 17: 382–6
Hickey MS, Carey JO, Azevedo JL, et al. Skeletal muscle fiber composition is related to adiposity and in vitro glucose transport rate in humans. Am J Physiol 1995; 268 (3 Pt 1): E453–457
Krotkiewski M, Björntorp P. Muscle tissue in obesity with different distribution of adipose tissue. Effects of physical training. Int J Obesity 1986; 10: 331–41
Biermann EL, Dole VP, Roberts TN. N abnormality of nonesterified fatty acid metabolism in diabetes mellitus. Diabetes 1957; 6: 475–9
Munkner C. Fasting concentrations of non-esterified fatty acids in diabetic and non-diabetic plasma and diurnal variations in normal subjects. Scand J Clin Lab Invest 1959; 11: 388–93
Golay A, Swislocki AM, Chen Y-DI, et al. Relationships between plasma-free fatty acid concentration, endogenous glucose production, and fasting hyperglycemia in normal and non-insulin-dependent diabetic individuals. Metabolism 1987; 36: 692–6
Williamson JR, browning T, Scholz R. Control mechanisms of gluconeogenesis and ketogenesis, 1: effects of oleate on gluconeogenesis in perfused rat liver. J Biol Chem 1969; 244: 4607–16
Boden G, Jadali F, White J, et al. Effects of fat on insulin-stimulated carbohydrate metabolism in normal men. J Clin Invest 1991; 88: 960–6
Piatti PM, Monti LD, Pacchioni M, et al. Forearm insulin- and non-insulin-mediated glucose uptake and muscle metabolism in man: role of free fatty acids and blood glucose levels. Metabolism 1991; 40: 926–33
Nestel PJ, Ishikawa T, Goldrick RB. Diminished plasma free fatty acid clearance in obese subjects. Metabolism 1978; 27: 589–97
Craig BW, Thompson K, Holloszy JO. Effects of stopping training on size and response to insulin of fat cells in female rats. J Appl Physiol 1983; 54: 571–5
Schneider SH, Amorosa LF, Kachadorian AK, et al. Studies of the mechanisms of improved glucose control during regular exercise in type 2 (non-insulin dependent) diabetes. Diabetologia 1984; 26: 355–60
Cortez MY, Torgan CE, Jr Brozinick JT, et al. Insulin resistance of obese Zucker rats exercise trained at two different intensities. Am J Physiol 1991; 261 (5 Pt 1): E613–E619
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ivy, J.L. Role of Exercise Training in the Prevention and Treatment of Insulin Resistance and Non-Insulin-Dependent Diabetes Mellitus. Sports Med 24, 321–336 (1997). https://doi.org/10.2165/00007256-199724050-00004
Published:
Issue Date:
DOI: https://doi.org/10.2165/00007256-199724050-00004