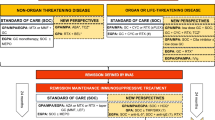
Abstract
The rapid expansion in the therapeutic modalities available for the treatment of anti-neutrophil cytoplasmic antibody-associated vasculitides (AAV), with clear limitations in existing strategies, prompted us to undertake a review of novel therapies reported in MEDLINE and EMBASE.
Tumour necrosis factor (TNF)-α antagonism with infliximab is described favourably in retrospective series and open-label trials. However, evidence from the WGET (Wegener’s Granulomatosis Etanercept Trial) does not support the clinical use of etanercept, and a significantly higher malignancy rate following TNFα inhibition questions the role of this approach. Uncontrolled evidence alone supports remission induction with rituximab-mediated B-lymphocyte depletion and may be less effective in predominantly granulomatous AAV. Remission following T-lymphocyte depletion can be achieved with alemtuzumab and anti-thymocyte globulin, but it is not yet clear what the clinical role will be for these agents in AAV. In addition, these agents are associated with prolonged lymphopenia and pulmonary complications, respectively. Stem cell transplantation to support immune reconstitution following the use of such agents has been trialled in AAV, but studies included very few patients.
Purine and pyrimidine antimetabolites mycophenolate mofetil and leflunomide are likely to play an important role in the treatment of AAV, but results supporting remission maintenance and induction in the former are limited to uncontrolled trials, such that their use remains experimental at this time. Similarly, 15-deoxyspergualin may provide an alternative to cyclophosphamide but awaits randomized controlled trial evidence. The MEPEX (MEthylprednisolone versus Plasma EXchange) trial supports plasma exchange in renal disease but this may be limited by pulmonary complications. Randomized controlled evidence also exists for intravenous immunoglobulin, although improvement may not be sustained. Antimicrobial therapy may be of use in Wegener’s granulomatosis patients with predominantly upper respiratory tract involvement.
Safety concerns, notably of infection and malignancy, were common and need to be explored in subsequent trials. In addition, concomitant immunosuppressants and non-standardized definitions were major limitations, and future studies of these and newer agents must follow agreed standards of study design and reporting to facilitate clearer interpretation of the circumstances (e.g. disease stage, severity or organ involvement) under which these agents perform optimally. Consequently, use is still limited to centres experienced in such agents and mostly in the context of clinical trials.
Similar content being viewed by others
References
Jennette JC, Falk RJ, Andrassy K, et al. Nomenclature of systemic vasculitides: proposal of an international consensus conference. Arthritis Rheum 1994; 37(2): 187–92
Leavitt RY, Fauci AS, Bloch DA, et al. The American College of Rheumatology 1990 criteria for the classification of Wegener’s granulomatosis. Arthritis Rheum 1990; 33(8): 1101–7
Masi AT, Hunder GG, Lie JT, et al. The American College of Rheumatology 1990 criteria for the classification of Churg-Strauss syndrome (allergic granulomatosis and angiitis). Arthritis Rheum 1990; 33(8): 1094–100
Lüdemann J, Utecht B, Gross WL. Anti-neutrophil cytoplasm antibodies in Wegener’s granulomatosis recognize an elastinolytic enzyme. J Exp Med 1990; 171(1): 357–62
Schreiber A, Luft FC, Kettritz R. Membrane proteinase 3 expression and ANCA-induced neutrophil activation. Kidney Int 2004; 65(6): 2172–83
Falk RJ, Jennette JC. Anti-neutrophil cytoplasmic autoanti-bodies with specificity for myeloperoxidase in patients with systemic vasculitis and idiopathic necrotizing and crescentic glomerulonephritis. N Engl J Med 1988; 318(25): 1651–7
Falk RJ, Terrell RS, Charles LA, et al. Anti-neutrophil cytoplasmic autoantibodies induce neutrophils to degranulate and produce oxygen radicals in vitro. Proc Natl Acad Sci U S A 1990; 87(11): 4115–9
Grimminger F, Hattar K, Papavassilis C, et al. Neutrophil activation by anti-proteinase 3 antibodies in Wegener’s granulomatosis: role of exogenous arachidonic acid and leukotriene B4 generation. J Exp Med 1996; 184(4): 1567–72
Reumaux D, Vossebeld PJ, Roos D, et al. Effect of tumor necrosis factor-induced integrin activation on Fc gamma receptor II-mediated signal transduction: relevance for activation of neutrophils by anti-proteinase 3 or anti-myeloperoxidase antibodies. Blood 1995; 86(8): 3189–95
Savage CO, Pottinger BE, Gaskin G, et al. Autoantibodies developing to myeloperoxidase and proteinase 3 in systemic vasculitis stimulate neutrophil cytotoxicity toward cultured endothelial cells. Am J Pathol 1992; 141(2): 335–42
Radford DJ, Luu NT, Hewins P, et al. Antineutrophil cytoplasmic antibodies stabilize adhesion and promote migration of flowing neutrophils on endothelial cells. Arthritis Rheum 2001; 44(12): 2851–61
Heeringa P, Brouwer E, Tervaert JW, et al. Animal models of anti-neutrophil cytoplasmic antibody associated vasculitis. Kidney Int 1998; 53(2): 253–63
Brouwer E, Huitema MG, Klok PA, et al. Antimyeloperoxidase-associated proliferative glomerulonephritis: an animal model. J Exp Med 1993; 177(4): 905–14
Xiao H, Heeringa P, Hu P, et al. Antineutrophil cytoplasmic autoantibodies specific for myeloperoxidase cause glomerulo-nephritis and vasculitis in mice. J Clin Invest 2002; 110(7): 955–63
Little MA, Smyth CL, Yadav R, et al. Antineutrophil cytoplasm antibodies directed against myeloperoxidase augment leuko-cyte-microvascular interactions in vivo. Blood 2005; 106(6): 2050–8
Pfister H, Ollert M, Fröhlich LF, et al. Antineutrophil cytoplasmic autoantibodies against the murine homolog of proteinase 3 (Wegener autoantigen) are pathogenic in vivo. Blood 2004; 104(5): 1411–8
De’Oliviera J, Gaskin G, Dash A, et al. Relationship between disease activity and anti-neutrophil cytoplasmic antibody concentration in long-term management of systemic vasculitis. Am J Kidney Dis 1995; 25(3): 380–9
Schlieben DJ, Korbet SM, Kimura RE, et al. Pulmonary-renal syndrome in a newborn with placental transmission of AN-CAs. Am J Kidney Dis 2005; 45(4): 758–61
Savige J, Gillis D, Benson E, et al. International consensus statement on testing and reporting of antineutrophil cytoplasmic antibodies (ANCA). Am J Clin Pathol 1999; 111(4): 507–13
Walton EW. Giant-cell granuloma of the respiratory tract (Wegener’s granulomatosis). BMJ 1958; 2(5091): 265–70
Hoffman GS, Kerr GS, Leavitt RY, et al. Wegener granulomatosis: an analysis of 158 patients. Ann Intern Med 1992; 116(6): 488–98
Fauci AS, Wolff SM. Wegener’s granulomatosis: studies in eighteen patients and a review of the literature. 1973. Medicine (Baltimore) 1994; 73(6): 315–24
Fauci AS, Haynes BF, Katz P. Wegener’s granulomatosis: prospective clinical and therapeutic experience with 85 patients for 21 years. Ann Intern Med 1983; 98(1): 76–85
De Groot K, Jayne D, Tesar V, et al. Randomised controlled trial of daily oral versus pulse cyclophosphamide for induction of remission in ANCA-associated systemic vasculitis [abstract]. Kidney Blood Press Res 2005; 28: 195
De Groot K, Adu D, Savage CO, EUVAS (European Vasculitis Study Group). The value of pulse cyclophosphamide in ANCA-associated vasculitis: meta-analysis and critical review. Nephrol Dial Transplant 2001; 16(10): 2018–27
Jayne D, Rasmussen N, Andrassy K, et al. A randomized trial of maintenance therapy for vasculitis associated with antineutrophil cytoplasmic autoantibodies. N Engl J Med 2003; 349(1): 36–44
Langford CA, Talar-Williams C, Barron KS, et al. A staged approach to the treatment of Wegener’s granulomatosis: induction of remission with glucocorticoids and daily cyclophosphamide switching to methotrexate for remission maintenance. Arthritis Rheum 1999; 42: 2666–73
Groot KD, Rasmussen N, Bacon PA, et al. Randomized trial of cyclophosphamide versus methotrexate for induction of remission in early systemic antineutrophil cytoplasmic antibody-associated vasculitis. Arthritis Rheum 2005; 52(8): 2461–9
Hollander D, Manning RT. The use of alkylating agents in the treatment of Wegener’s granulomatosis. Ann Intern Med 1967; 67(2): 393–8
Beutler B, Cerami A. Cachectin: more than a tumor necrosis factor. N Engl J Med 1987; 316(7): 379–85
Noronha IL, Krüger C, Andrassy K, et al. In situ production of TNF-alpha, IL-1 beta and IL-2R in ANCA-positive glomerulo-nephritis. Kidney Int 1993; 43(3): 682–92
Lúdvíksson BR, Sneller MC, Chua KS, et al. Active Wegener’s granulomatosis is associated with HLA-DR+ CD4+ T cells exhibiting an unbalanced Th1-type T cell cytokine pattern: reversal with IL-10. J Immunol 1998; 160(7): 3602–9
Little MA, Bhangal G, Smyth CL, et al. Therapeutic effect of anti-TNF-alpha antibodies in an experimental model of antineutrophil cytoplasm antibody-associated systemic vasculitis. J Am Soc Nephrol 2006; 17(1): 160–9
Jónasdóttir O, Petersen J, Bendtzen K. Tumour necrosis factor-alpha (TNF), lymphotoxin and TNF receptor levels in serum from patients with Wegener’s granulomatosis. APMIS 2001; 109(11): 781–6
Booth AD, Jayne DRW, Kharbanda RK, et al. Infliximab improves endothelial dysfunction in systemic vasculitis: a model of vascular inflammation. Circulation 2004; 109(14): 1718–23
Moreland LW, Schiff MH, Baumgartner SW, et al. Etanercept therapy in rheumatoid arthritis: a randomized, controlled trial. Ann Intern Med 1999; 130(6): 478–86
Weinblatt ME, Kremer JM, Bankhurst AD, et al. A trial of etanercept, a recombinant tumor necrosis factor receptor:Fc fusion protein, in patients with rheumatoid arthritis receiving methotrexate. N Engl J Med 1999; 340(4): 253–9
Stone JH, Uhlfelder ML, Hellmann DB, et al. Etanercept combined with conventional treatment in Wegener’s granulomatosis: a six-month open-label trial to evaluate safety. Arthritis Rheum 2001; 44(5): 1149–54
The WGET Group. Etanercept plus standard therapy for Wegener’s granulomatosis. N Engl J Med 2005; 352(4): 351–61
Stone JH, Holbrook JT, Marriott MA, et al. Solid malignancies among patients in the Wegener’s Granulomatosis Etanercept Trial. Arthritis Rheum 2006; 54(5): 1608–18
Seo P, Min Y, Holbrook JT, et al., W.G.E.T. Research Group. Damage caused by Wegener’s granulomatosis and its treatment: prospective data from the Wegener’s Granulomatosis Etanercept Trial (WGET). Arthritis Rheum 2005; 52(7): 2168–78
Feldmann M. Development of anti-TNF therapy for rheumatoid arthritis. Nat Rev Immunol 2002; 2(5): 364–71
Lamprecht P, Voswinkel J, Lilienthal T, et al. Effectiveness of TNF-alpha blockade with infliximab in refractory Wegener’s granulomatosis. Rheumatology (Oxford) 2002; 41(11): 1303–7
Booth AD, Jefferson HJ, Ayliffe W, et al. Safety and efficacy of TNFalpha blockade in relapsing vasculitis [letter]. Ann Rheum Dis 2002; 61(6): 559
Bartolucci P, Ramanoelina J, Cohen P, et al. Efficacy of the anti-TNF-alpha antibody infliximab against refractory systemic vasculitides: an open pilot study on 10 patients. Rheumatology (Oxford) 2002; 41(10): 1126–32
Booth A, Harper L, Hammad T, et al. Prospective study of TNF-alpha blockade with infliximab in anti-neutrophil cytoplasmic antibody-associated systemic vasculitis. J Am Soc Nephrol 2004; 15(3): 717–21
Aeberli D, Oertle S, Mauron H, et al. Inhibition of the TNF-pathway: use of infliximab and etanercept as remission-inducing agents in cases of therapy-resistant chronic inflammatory disorders. Swiss Med Wkly 2002; 132(29–30): 414–22
Arbach O, Gross WL, Gause A. Treatment of refractory Churg-Strauss-Syndrome (CSS) by TNF-alpha blockade. Immunobiology 2002; 206(5): 496–501
Sangle SR, Hughes GRV, D’Cruz DP. Infliximab in patients with systemic vasculitis that is difficult to treat: poor outcome and significant adverse effects. Ann Rheum Dis 2007; 66(4): 564–5
Sandborn WJ, Hanauer SB, Katz S, et al. Etanercept for active Crohn’s disease: a randomized, double-blind, placebo-controlled trial. Gastroenterology 2001; 121(5): 1088–94
Hanauer SB, Feagan BG, Lichtenstein GR, et al.,ACCENT I Study Group. Maintenance infliximab for Crohn’s disease: the ACCENT I randomised trial. Lancet 2002; 359(9317): 1541–9
Scallon B, Cai A, Solowski N, et al. Binding and functional comparisons of two types of tumor necrosis factor antagonists. J Pharmacol Exp Ther 2002; 301(2): 418–26
Askling J, Fored CM, Baecklund E, et al. Haematopoietic malignancies in rheumatoid arthritis: lymphoma risk and characteristics after exposure to tumour necrosis factor antagonists. Ann Rheum Dis 2005; 64(10): 1414–1420
Wallis RS, Broder MS, Wong JY, et al. Granulomatous infectious diseases associated with tumor necrosis factor antagonists. Clin Infect Dis 2004; 38(9): 1261–5
Keane J. TNF-blocking agents and tuberculosis: new drugs illuminate an old topic. Rheumatology (Oxford) 2005; 44(6): 714–20
Pisetsky DS. Tumor necrosis factor alpha blockers and the induction of anti-DNA autoantibodies. Arthritis Rheum 2004; 3(11): 2381–2
Eriksson C, Engstrand S, Sundqvist K, et al. Autoantibody formation in patients with rheumatoid arthritis treated with anti-TNF alpha. Ann Rheum Dis 2005; 64(3): 403–7
Csernok E, Trabandt A, Müller A, et al. Cytokine profiles in Wegener’s granulomatosis: predominance of type 1 (Th1) in the granulomatous inflammation. Arthritis Rheum 1999; 42(4): 742–50
Müller A, Trabandt A, Gloeckner-Hofmann K, et al. Localized Wegener’s granulomatosis: predominance of CD26 and IFN-gamma expression. J Pathol 2000; 192(1): 113–20
Voswinkel J, Mueller A, Kraemer JA, et al. B lymphocyte maturation in Wegener’s granulomatosis: a comparative analysis of VH genes from endonasal lesions. Ann Rheum Dis 2006; 65(7): 859–64
Cupps TR, Edgar LC, Fauci AS. Suppression of human B lymphocyte function by cyclophosphamide. J Immunol 1982; 128(6): 2453–7
Silverman GJ, Weisman S. Rituximab therapy and autoimmune disorders: prospects for anti-B cell therapy. Arthritis Rheum 2003; 48(6): 1484–92
Edwards JCW, Szczepanski L, Szechinski J, et al. Efficacy of B-cell-targeted therapy with rituximab in patients with rheumatoid arthritis. N Engl J Med 2004; 350(25): 2572–81
Looney RJ, Anolik JH, Campbell D, et al. B cell depletion as a novel treatment for systemic lupus erythematosus: a phase I/II dose-escalation trial of rituximab. Arthritis Rheum 2004; 50(8): 2580–9
Specks U, Fervenza FC, McDonald TJ, et al. Response of Wegener’s granulomatosis to anti-CD20 chimeric monoclonal antibody therapy. Arthritis Rheum 2001; 44(12): 2836–40
Keogh KA, Wylam ME, Stone JH, et al. Induction of remission by B lymphocyte depletion in eleven patients with refractory antineutrophil cytoplasmic antibody-associated vasculitis. Arthritis Rheum 2005; 52(1): 262–8
Eriksson P. Nine patients with anti-neutrophil cytoplasmic antibody-positive vasculitis successfully treated with rituximab. J Intern Med 2005; 257(6): 540–8
Keogh KA, Ytterberg SR, Fervenza FC, et al. Rituximab for refractory Wegener’s granulomatosis: report of a prospective, open-label pilot trial. Am J Respir Crit Care Med 2006; 173(2): 180–7
Henes JC, Fritz J, Koch S, et al. Rituximab for treatment-resistant extensive Wegener’s granulomatosis-additive effects of a maintenance treatment with leflunomide. Clin Rheumatol 2007; 26(10): 1711–5
Aries PM, Hellmich B, Voswinkel J, et al. Lack of efficacy of rituximab in Wegener’s granulomatosis with refractory granulomatous manifestations. Ann Rheum Dis 2006; 65(7): 853–8
Aries PM, Lamprecht P, Gross WL. Rituximab in refractory Wegener’s granulomatosis: favorable or not? Am J Respir Crit Care Med 2006; 173(7): 815–6
Omdal R, Wildhagen K, Hansen T, et al. Anti-CD20 therapy of treatment-resistant Wegener’s granulomatosis: favourable but temporary response. Scand J Rheumatol 2005; 34(3): 229–32
Brihaye B, Aouba A, Pagnoux C, et al. Adjunction of rituximab to steroids and immunosuppressants for refractory/relapsing Wegener’s granulomatosis: a study on 8 patients. Clin Exp Rheumatol 2007, S27
Bacon PA. The spectrum of Wegener’s granulomatosis and disease relapse. N Engl J Med 2005; 352(4): 330–2
Smith KGC, Jones RB, Burns SM, et al. Long-term comparison of rituximab treatment for refractory systemic lupus erythematosus and vasculitis: remission, relapse, and re-treatment. Arthritis Rheum 2006; 54(9): 2970–82
Stasi R, Stipa E, Poeta GD, et al. Long-term observation of patients with anti-neutrophil cytoplasmic antibody-associated vasculitis treated with rituximab. Rheumatology (Oxford) 2006; 45(11): 1432–6
Golbin J, Keogh K, Fervenza F, et al. Repeated use of rituximab in refractory Wegener’ s granulomatosis: efficacy for glucocor-ticoid-free remission maintenance [abstract]. Arthritis Rheum 2006; 54 Suppl. 9: S527
Kallenbach M, Duan H, Ring T. Rituximab induced remission in a patient with Wegener’s granulomatosis. Nephron Clin Pract 2005; 99(3): c92–6
Cheung CMG, Murray PI, Savage COS. Successful treatment of Wegener’s granulomatosis associated scleritis with rituximab. Br J Ophthalmol 2005; 89(11): 1542
Ferraro AJ, Day CJ, Drayson MT, et al. Effective therapeutic use of rituximab in refractory Wegener’s granulomatosis. Ne-phrol Dial Transplant 2005; 20(3): 622–5
Bachmeyer C, Cadranel J, Demontis R. Rituximab is an alternative in a case of contra-indication of cyclophosphamide in Wegener’s granulomatosis. Nephrol Dial Transplant 2005; 20(6): 1274
Kaushik VV, Reddy HV, Bucknall RC. Successful use of rituximab in a patient with recalcitrant Churg-Strauss Syndrome. Ann Rheum Dis 2006; 65(8): 1116–7
Koukoulaki M, Smith KGC, Jayne DRW. Rituximab in Churg-Strauss Syndrome. Ann Rheum Dis 2006; 65(4): 557–9
Manz RA, Radbruch A. Plasma cells for a lifetime? Eur J Immunol 2002; 32(4): 923–7
Reff ME, Carner K, Chambers KS, et al. Depletion of B cells in vivo by a chimeric mouse human monoclonal antibody to CD20. Blood 1994; 83(2): 435–45
Kuus-Reichel K, Grauer LS, Karavodin LM, et al. Will immunogenicity limit the use, efficacy, and future development of therapeutic monoclonal antibodies? Clin Diagn Lab Immunol 1994; 1(4): 365–72
Edwards JCW, Szczepanski L, Szechinski J, et al. Efficacy of B-cell-targeted therapy with rituximab in patients with rheumatoid arthritis. N Engl J Med 2004; 350(25): 2572–81
Eisenberg R. Update on rituximab. Ann Rheum Dis 2005; 64 Suppl. 4: iv55-7
Anolik JH, Campbell D, Felgar RE, et al. The relationship of FcgammaRIIIa genotype to degree of B cell depletion by rituximab in the treatment of systemic lupus erythematosus. Arthritis Rheum 2003; 48(2): 455–9
FDA. Information for healthcare professionals: rituximab [online]. Available from URL: http://www.fda.gov/CDER/Drug/InfoSheets/HCP/rituximab.pdf [Accessed 2008 Feb 14]
Calabrese LH, Molloy ES, Huang D, et al. Progressive multifocal leukoencephalopathy in rheumatic diseases: evolving clinical and pathologic patterns of disease. Arthritis Rheum 2007; 56(7): 2116–28
Schlesier M, Kaspar T, Gutfleisch J, et al. Activated CD4+ and CD8+ T-cell subsets in Wegener’s granulomatosis. Rheumatol Int 1995; 14(5): 213–9
Moosig F, Csernok E, Wang G, et al. Costimulatory molecules in Wegener’s granulomatosis (WG): lack of expression of CD28 and preferential up-regulation of its ligands B7-1 (CD80) and B7-2 (CD86) on T cells. Clin Exp Immunol 1998; 114(1): 113–8
Komocsi A, Lamprecht P, Csernok E, et al. Peripheral blood and granuloma CD4(+)CD28(−) T cells are a major source of interferon-gamma and tumor necrosis factor-alpha in Wegener’s granulomatosis. Am J Pathol 2002; 160(5): 1717–24
Abdulahad WH, Stegeman CA, Limburg PC, et al. CD4-posi-tive effector memory T cells participate in disease expression in ANCA-associated vasculitis. Ann N Y Acad Sci 2007; 1107: 22–31
Brouwer E, Tervaert JW, Horst G, et al. Predominance of IgGl and IgG4 subclasses of anti-neutrophil cytoplasmic autoanti-bodies (ANCA) in patients with Wegener’s granulomatosis and clinically related disorders. Clin Exp Immunol 1991; 83(3): 379–86
Domagata A, Kurpisz M. CD52 antigen-a review. Med Sci Monit 2001;7(2): 325–31
Lockwood CM, Thiru S, Stewart S, et al. Treatment of refractory Wegener’s granulomatosis with humanized monoclonal antibodies. QJM 1996; 89(12): 903–12
Walsh M, Chaudhry A, Jayne DR. Long-term follow-up of relapsing/refractory ANCA associated vasculitis treated with the lymphocyte depleting antibody alemtuzumab (CAMPATH-1H). Ann Rheum Dis. Epub 2007 Nov 29
Hagen EC, de Keizer RJ, Andrassy K, et al. Compassionate treatment of Wegener’s granulomatosis with rabbit anti-thymocyte globulin. Clin Nephrol 1995; 43(6): 351–9
Lukas R, Keller F. Anti-thymocyte globulin therapy in a patient with pANCA vasculitis and crescentic glomerulonephritis [letter]. Nephron 1998; 78(2): 231
Schmitt WH, Hagen EC, Neumann I, et al. Treatment of refractory Wegener’s granulomatosis with antithymocyte globulin (ATG): an open study in 15 patients. Kidney Int 2004; 65(4): 1440–8
Tyndall A, Saccardi R. Haematopoietic stem cell transplantation in the treatment of severe autoimmune disease: results from phase I/II studies, prospective randomized trials and future directions. Clin Exp Immunol 2005; 141(1): 1–9
Kötter I, Daikeler T, Amberger C, et al. Autologous stem cell transplantation of treatment-resistant systemic vascularis: a single centre experience and review of the literature. Clin Nephrol 2005; 64(6): 485–9
Kötter I, Daikeler T, Amberger C, et al. Autologous stem cell transplantation of treatment-resistant systemic vasculitis-a single center experience and review of the literature. Clin Nephrol 2005; 4(6): 485–9
Daikeler T, Kötter I, Tyndall CB, et al. Haematopoietic stem cell transplantation for vasculitis including Behcet’s disease and polychondritis: a retrospective analysis of patients recorded in the European Bone Marrow Transplantation and European League Against Rheumatism databases and a review of the literature. Ann Rheum Dis 2007; 66(2): 202–7
Allison AC, Eugui EM. Purine metabolism and immunosup-pressive effects of mycophenolate mofetil (MMF). Clin Transplant 1996; 10 (1 Pt 2): 77–84
Nowack R, Göbel U, Klooker P, et al. Mycophenolate mofetil for maintenance therapy of Wegener’s granulomatosis and microscopic polyangiitis: a pilot study in 11 patients with renal involvement. J Am Soc Nephrol 1999; 10(9): 1965–71
Langford CA, Talar-Williams C, Sneller MC. Mycophenolate mofetil for remission maintenance in the treatment of Wegener’s granulomatosis. Arthritis Rheum 2004; 51(2): 278–83
Joy MS, Hogan SL, Jennette JC, et al. A pilot study using mycophenolate mofetil in relapsing or resistant ANCA small vessel vasculitis. Nephrol Dial Transplant 2005; 20(12): 2725–32
Koukoulaki M, Jayne DRW. Mycophenolate mofetil in anti-neutrophil cytoplasm antibodies-associated systemic vasculitis. Nephron Clin Pract 2006; 102(3-4): c100–7
Haubitz M, de Groot K. Tolerance of mycophenolate mofetil in end-stage renal disease patients with ANCA-associated vasculitis. Clin Nephrol 2002; 57(6): 421–4
Maes B, Oellerich M, Ceuppens JL, et al. A new acute inflammatory syndrome related to the introduction of mycophenolate mofetil in patients with Wegener’s granulomatosis. Nephrol Dial Transplant 2002; 17(5): 923–6
Stassen PM, Tervaert JWC, Stegeman CA. Induction of remission in active anti-neutrophil cytoplasmic antibody-associated vasculitis with mycophenolate mofetil in patients who cannot be treated with cyclophosphamide. Ann Rheum Dis 2007; 66(6): 798–802
Fox RI, Herrmann ML, Frangou CG, et al. Mechanism of action for leflunomide in rheumatoid arthritis. Clin Immunol 1999; 93(3): 198–208
Manna SK, Aggarwal BB. Immunosuppressive leflunomide metabolite (A77 1726) blocks TNF-dependent nuclear factor-kappa B activation and gene expression. J Immunol 1999; 162(4): 2095–102
Smolen JS, Kalden JR, Scott DL, et al. Efficacy and safety of leflunomide compared with placebo and sulphasalazine in active rheumatoid arthritis: a double-blind, randomised, multi-centre trial. European Leflunomide Study Group. Lancet 1999; 353(9149): 259–66
Poór G, Strand V, Group LMS. Efficacy and safety of leflunomide 10mg versus 20mg once daily in patients with active rheumatoid arthritis: multinational double-blind, randomized trial. Rheumatology (Oxford) 2004; 43(6): 744–9
Kremer JM, Genovese MC, Cannon GW, et al. Concomitant leflunomide therapy in patients with active rheumatoid arthritis despite stable doses of methotrexate: a randomized, double-blind, placebo-controlled trial. Ann Intern Med 2002; 137(9): 726–33
Bruyn GA, Griep EN, Korff KJ. Leflunomide for active rheumatoid arthritis [letter]. Lancet 1999; 353(9167): 1883
Chan ATY, Bradlow A, McNally J. Leflunomide induced vasculitis-a dose-response relationship. Rheumatology (Oxford) 2003; 42(3): 492–3
Metzler C, Fink C, Lamprecht P, et al. Maintenance of remission with leflunomide in Wegener’s granulomatosis. Rheumatology (Oxford) 2004; 43(3): 315–20
Metzler C, Miehle N, Manger K, et al. Elevated relapse rate under oral methotrexate versus leflunomide for maintenance of remission in Wegener’s granulomatosis. Rheumatology (Oxford) 2007; 46(7): 1087–91
Umezawa H, Kondo S, Iinuma H, et al. Structure of an an-titumor antibiotic, spergualin. J Antibiot (Tokyo) 1981; 34(12): 1622–4
Nadler SG, Tepper MA, Schacter B, et al. Interaction of the immunosuppressant deoxyspergualin with a member of the Hsp70 family of heat shock proteins. Science 1992; 258(5081): 484–6
Tepper MA, Nadler SG, Esselstyn JM, et al. Deoxyspergualin inhibits kappa light chain expression in 70Z/3 pre-B cells by blocking lipopolysaccharide-induced NF-kappa B activation. J Immunol 1995; 155(5): 2427–36
Takeuchi T, Iinuma H, Kunimoto S, et al. A new antitumor antibiotic, spergualin: isolation and antitumor activity. J Antibiot (Tokyo) 1981; 34(12): 1619–21
Amemiya H, Suzuki S, Ota K, et al. A novel rescue drug, 15-deoxyspergualin: first clinical trials for recurrent graft rejection in renal recipients. Transplantation 1990; 49(2): 337–43
Birck R, Newman M, Braun C, et al. 15-Deoxyspergualin and cyclophosphamide, but not mycophenolate mofetil, prolong survival and attenuate renal disease in a murine model of ANCA-associated crescentic nephritis. Nephrol Dial Transplant 2006; 21(1): 58–63
Birck R, Warnatz K, Lorenz HM, et al. 15-Deoxyspergualin in patients with refractory ANCA-associated systemic vasculitis: a six-month open-label trial to evaluate safety and efficacy. J Am Soc Nephrol 2003; 14(2): 440–7
Schmitt WH, Birck R, Heinzel PA, et al. Prolonged treatment of refractory Wegener’s granulomatosis with 15-deoxyspergualin: an open study in seven patients. Nephrol Dial Transplant 2005; 20(6): 1083–92
Kälsch A, Schmitt WH, Breedijk A, et al. In vivo effects of cyclic administration of 15-deoxyspergualin on leucocyte function in patients with Wegener’s granulomatosis. Clin Exp Immunol 2006; 146(3): 455–62
Flossmann O, Cohen Tervaert JW, Hellmich B, et al. Deoxys-pergaulin for refractory Wegener’s granulomatosis [abstract]. 13th International ANCA/vasculitis workshop; 2007 Apr 28; Cancun
Rossi F, Jayne DR, Lockwood CM, et al. Anti-idiotypes against anti-neutrophil cytoplasmic antigen autoantibodies in normal human polyspecific IgG for therapeutic use and in the remission sera of patients with systemic vasculitis. Clin Exp Immunol 1991; 83(2): 298–303
Brooks CJ, King WJ, Radford DJ, et al. IL-1 beta production by human polymorphonuclear leucocytes stimulated by anti-neutrophil cytoplasmic autoantibodies: relevance to systemic vasculitis. Clin Exp Immunol 1996; 106(2): 273–9
Jayne DR, Esnault VL, Lockwood CM. Anti-idiotype antibodies to anti-myeloperoxidase autoantibodies in patients with systemic vasculitis. J Autoimmun 1993; 6(2): 221–6
Jayne DR, Chapel H, Adu D, et al. Intravenous immunoglobulin for ANCA-associated systemic vasculitis with persistent disease activity. QJM 2000; 93(7): 433–9
Jayne DR, Davies MJ, Fox CJ, et al. Treatment of systemic vasculitis with pooled intravenous immunoglobulin. Lancet 1991; 337(8750): 1137–9
Jayne DR, Esnault VL, Lockwood CM. ANCA anti-idiotype antibodies and the treatment of systemic vasculitis with intravenous immunoglobulin. J Autoimmun 1993; 6(2): 207–19
Jayne DR, Lockwood CM. Intravenous immunoglobulin as sole therapy for systemic vasculitis. Br J Rheumatol 1996; 35(11): 1150–3
Richter C, Schnabel A, Csernok E, et al. Treatment of anti-neutrophil cytoplasmic antibody (ANCA)-associated systemic vasculitis with high-dose intravenous immunoglobulin. Clin Exp Immunol 1995; 101(1): 2–7
Ito-Ihara T, Ono T, Nogaki F, et al. Clinical efficacy of intravenous immunoglobulin for patients with MPO-ANCA-associated rapidly progressive glomerulonephritis. Nephron Clin Pract 2006; 102(1): c35–42
Muso E, Ito-Ihara T, Ono T, et al. Intravenous immunoglobulin (IVIg) therapy in MPO-ANCA related polyangiitis with rapidly progressive glomerulonephritis in Japan. Jpn J Infect Dis 2004; 57(5): S17–8
Tesar V, Jelínková E, Masek Z, et al. Influence of plasma exchange on serum levels of cytokines and adhesion molecules in ANCA-positive renal vasculitis. Blood Purif 1998; 16(2): 72–80
Pusey CD, Rees AJ, Evans DJ, et al. Plasma exchange in focal necrotizing glomerulonephritis without anti-GBM antibodies. Kidney Int 1991; 40(4): 757–63
Frascà GM, Soverini ML, Falaschini A, et al. Plasma exchange treatment improves prognosis of antineutrophil cytoplasmic antibody-associated crescentic glomerulonephritis: a case-control study in 26 patients from a single center. Ther Apher Dial 2003; 7(6): 540–6
Nakamura T, Matsuda T, Kawagoe Y, et al. Plasmapheresis with immunosuppressive therapy vs immunosuppressive therapy alone for rapidly progressive anti-neutrophil cytoplasmic autoantibody-associated glomerulonephritis. Nephrol Dial Transplant 2004; 19(7): 1935–7
Aasarød K, Iversen BM, Hammerström J, et al. Clinical outcome of patients with Wegener’s granulomatosis treated with plasma exchange. Blood Purif 2002; 20(2): 167–73
Klemmer PJ, Chalermskulrat W, Reif MS, et al. Plasmapheresis therapy for diffuse alveolar hemorrhage in patients with small-vessel vasculitis. Am J Kidney Dis 2003; 42(6): 1149–53
Jayne DRW, Gaskin G, Rasmussen N, et al. Randomized trial of plasma exchange or high-dosage methylprednisolone as adjunctive therapy for severe renal vasculitis. J Am Soc Nephrol 2007; 18(7): 2180–8
Stegeman CA, Tervaert JW, Sluiter WJ, et al. Association of chronic nasal carriage of Staphylococcus aureus and higher relapse rates in Wegener granulomatosis. Ann Intern Med 1994; 120(1): 12–7
Reinhold-Keller E, Groot KD, Rudert H, et al. Response to trimethoprim/sulfamethoxazole in Wegener’s granulomatosis depends on the phase of disease. QJM 1996; 89(1): 15–23
Stegeman CA, Tervaert JW, de Jong PE, et al. Trimethoprim-sulfamethoxazole (co-trimoxazole) for the prevention of relapses of Wegener’s granulomatosis. Dutch Co-Trimoxazole Wegener Study Group. N Engl J Med 1996; 335(1): 16–20
de Groot K, Reinhold-Keller E, Tatsis E, et al. Therapy for the maintenance of remission in sixty-five patients with generalized Wegener’s granulomatosis: methotrexate versus trimethoprim/sulfamethoxazole. Arthritis Rheum 1996; 39(12): 2052–61
Chung JB, Armstrong K, Schwartz JS, et al. Cost-effectiveness of prophylaxis against Pneumocystis carinii pneumonia in patients with Wenger’s granulomatosis undergoing immunosuppresive therapy. Arthritis Rheum 2000; 43(8): 1841–8
The European Vasculitis Study Group. RITUXVAS clinical trial protocol [online]. Available from URL: http://www.vasculitis.org/protocols/RITUXVAS.pdf [Accessed 2008 Feb 14]
Immune Tolerance Network. RAVE: rituximab for ANCA-associated vasculitis [online]. Available from URL: http://www.immunetolerance.org/RAVE/ [Accessed 2008 Feb 12]
National Institutes of Heath. RATTRAP: infliximab versus rituximab in systemic necrotizing vasculitides [online]. Available from URL: http://www.clinicaltrials.gov/show/NCT00307593 [Accessed 2008 Feb 12]
The European Vasculitis Study Group. International mycophenolate mofetil protocol to reduce outbreaks of vasculitides: IMPROVE [online]. Available from URL: http://www.vascu-litis.org/protocols/IMPROVE.pdf [Accessed 2008 Feb 14]
The European Vasculitis Study Group. MYCYC clinical trial protocol [online]. Available from URL: http://www.vasculitis.org/protocols/MYCYC.pdf [Accessed 2008 Feb 14]
Jayne DR, Rasmussen N. Treatment of antineutrophil cytoplasm autoantibody-associated systemic vasculitis: initiatives of the European Community Systemic Vasculitis Clinical Trials Study Group. Mayo Clin Proc 1997; 72(8): 737–47
The European Vasculitis Study Group. Clinical trial protocol: REMAIN [online]. Available from URL: http://www.vasculitis.org/protocols/REMAIN%20JAN%202006.pdf [Accessed 2008 Feb 14]
Clark M. Antibody humanization: a case of the ’Emperor’s new clothes’? Immunol Today 2000; 21(8): 397–402
The European Vasculitis Study Group. A pilot study examining the effect of abatacept in ANCA associated vasculitis: ABAVAS [online]. Available from URL: http://www.vasculitis.org/protocols/abavas.pdf [Accessed 2008 Feb 14]
Kumar S, Boehm J, Lee JC. p38 MAP kinases: key signalling molecules as therapeutic targets for inflammatory diseases. Nat Rev Drug Discov 2003; 2(9): 717–26
Karin M, Yamamoto Y, Wang QM. The IKK NF-kappa B system: a treasure trove for drug development. Nat Rev Drug Discov 2004; 3(1): 17–26
Boudes P. Purely granulomatous Wegener’s granulomatosis: a new concept for an old disease. Semin Arthritis Rheum 1990; 19(6): 365–70
Schmitt WH, Heesen C, Csernok E, et al. Elevated serum levels of soluble interleukin-2 receptor in patients with Wegener’s granulomatosis: association with disease activity. Arthritis Rheum 1992; 35(9): 1088–96
Wang G, Hansen H, Tatsis E, et al. High plasma levels of the soluble form of CD30 activation molecule reflect disease activity in patients with Wegener’s granulomatosis. Am J Med 1997; 102(6): 517–23
Jayne D. Update on the European Vasculitis Study Group trials. Curr Opin Rheumatol 2001; 13(1): 48–55
Lamprecht P, Gross WL. A brief history of Wegener’s granulomatosis: on limited, localized, and generalized forms of the disease: comment on the article by the Wegener’s Granulomatosis Etanercept Trial Research Group. Arthritis Rheum 2004; 50(1): 334–5
Luqmani RA, Bacon PA, Moots RJ, et al. Birmingham Vasculitis Activity Score (BVAS) in systemic necrotizing vasculitis. QJM 1994; 87(11): 671–8
Stone JH, Hoffman GS, Merkel PA, et al., for the Study of the Systemic Vasculitides (INSSYS), I.N. A disease-specific activity index for Wegener’s granulomatosis: modification of the Birmingham Vasculitis Activity Score. International Network for the Study of the Systemic Vasculitides (INSSYS). Arthritis Rheum 2001; 44(4): 912–920
Silman A, Klareskog L, Breedveld F, et al. Proposal to establish a register for the long term surveillance of adverse events in patients with rheumatic diseases exposed to biological agents: the EULAR Surveillance Register for Biological Compounds. Ann Rheum Dis 2000; 59(6): 419–20
Hellmich B, Flossmann O, Gross WL, et al. EULAR recommendations for conducting clinical studies and/or clinical trials in systemic vasculitis: focus on anti-neutrophil cytoplasm antibody-associated vasculitis. Ann Rheum Dis 2007; 66(5): 605–17
Merkel PA. Part 1: the need for novel treatment regimens for ANCA-associated vasculitis. Clin Exp Rheumatol 2007; 25 (1 Suppl. 44): S72-3
Acknowledgements
We acknowledge the comprehensive advice and assistance from evidence-based and knowledge management librarian Ben Skinner at Brighton and Sussex University Hospitals with regard to the literature search undertaken.
No sources of funding were used to assist in the preparation of this manuscript. The authors have no conflicts of interest that are directly relevant to the content of this review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, R.W., D’Cruz, D.P. Novel Therapies for Anti-Neutrophil Cytoplasmic Antibody-Associated Vasculitis. Drugs 68, 747–770 (2008). https://doi.org/10.2165/00003495-200868060-00003
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-200868060-00003