Abstract
Diabetes mellitus affects about 8% of the adult population. The estimated number of patients with diabetes, presently about 170 million people, is expected to increase by 50–70% within the next 25 years.
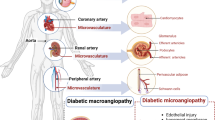
Diabetes is an important component of the complex of ‘common’ cardiovascular risk factors, and is responsible for acceleration and worsening of atherothrombosis. Major cardiovascular events cause about 80% of the total mortality in diabetic patients. Diabetes also induces peculiar microangiopathic changes leading to diabetic nephropathy conducive to end-stage renal failure, and to diabetic retinopathy that may progress to vision loss and blindness.
In terms of major cardiovascular events, coronary heart disease and ischaemic stroke are the main causes of morbidity and mortality in diabetic patients. Peripheral arterial disease frequently occurs, and is more likely to be conducive to critical limb ischaemia and amputation than in the absence of diabetes.
Although there are a number of differences in the pathogenesis and clinical features of diabetic macroangiopathy and microangiopathy, these two entities often coexist and induce mutually worsening effects. Endothelial injury, dysfunction and damage are common starting points for both conditions. Causes of endothelial injury can be distinguished into those ‘common’ to nondiabetic atherothrombosis, such as hypertension, dyslipidaemia, smoking, hypercoagulability and platelet activation; and those more specific and in some cases ‘unique’ to diabetes and directly related to the metabolic derangement of the disease, such as (i) desulfation of glycosaminoglycans (GAGs) of the vascular matrix; (ii) formation of advanced glycation end-products (AGE) and their endothelial receptors (RAGE); (iii) oxidative and reductive stress; (iv) decline in nitric oxide production; (v) activation of the renin-angiotensin aldosterone system (RAAS); and (vi) endothelial inflammation caused by glucose, insulin, insulin precursors and AGE/RAGE.
Prevention of major cardiovascular events with the antithrombotic agent aspirin (acetylsalicylic acid) is widely recommended, but reportedly underutilised in patients with diabetes. However, some data suggest that aspirin may be less effective than expected in preventing cardiovascular events and especially mortality in patients with diabetes, as well as in slowing progression of retinopathy.
In contrast, a recent study found picotamide, a direct thromboxane inhibitor, to be superior to aspirin in diabetic patients. Clopidogrel was either equivalent or less active in diabetic versus nondiabetic patients, depending upon different clinical settings.
Recent studies have shown that some GAG compounds are able to reduce micro-and macroalbuminuria in diabetic nephropathy, and hard exudates in diabetic retinopathy, but it is as yet unknown whether these agents also influence the natural history of microvascular complications of diabetes. Lifestyle changes and physical exercise are also essential in preventing cardiovascular events in diabetic patients.
Available data on the control of the metabolic state and the main risk factors show that careful adjustment of blood sugar and glycated haemoglobin is more effective in counteracting microvascular damage than in preventing major cardiovascular events. The latter objective requires a more comprehensive approach to the whole constellation of risk factors both specific for diabetes and common to atherothrombosis. This approach includes lifestyle modifications, such as dietary changes and smoking cessation and the use of HMG-CoA reductase inhibitors (statins), which are able to correct the lipid status and to prevent major cardiovascular events independently of the baseline lipidaemic or cardiovascular status.
Tight control of hypertension is essential to reduce not only major cardiovascular events but also microvascular complications. Among antihypertensive measures, blockade of the RAAS by means of ACE inhibitors or angiotensin II receptor antagonists recently emerged as a potentially polyvalent approach, not only for treating hypertension and reducing cardiovascular events, but also to prevent or reduce albuminuria, counteract diabetic nephropathy and lower the occurrence of new type 2 diabetes in individuals at risk.




Similar content being viewed by others
References
American Diabetes Association. ADA clinical practice recommendations. Diabetes Care 2004; 27: S1–143
Cheta D, Panaite C, Balas B, et al. Cardiovascular complications of diabetes mellitus: magnitude of the problem. In: Cheta D, editor. Vascular involvement in diabetes. Basel: Karger, 2005: 3–11
Beckman JA, Creager MA, Libby P. Diabetes and atherosclerosis: epidemiology, pathophysiology and management. JAMA 2002; 287: 2570–81
Braunwald E. Preface. In: Cheta D, editor. Vascular involvement in diabetes. Basel: Karger, 2005: XI
McAlpine RR, Morris AD, Emslie-Smith A, et al. The annual incidence of diabetic complications in a population with type 1 and 2 diabetes. Diabet Med 2005; 22: 348–52
Guja C, Ionescu-Tirgoviste C. Endothelial dysfunction and type 2 diabetes mellitus. In: Cheta D, editor. Vascular involvement in diabetes. Basel: Karger, 2005: 205–20
Rossi R, Nuzzo A, Grimaldi T, et al. Diabetes and cardiovascular disease: a close and dangerous connection. Heart Int 2005; 1: 18–23
Booth GL, Kapral MK, Fung K, et al. Relation between age and cardiovascular disease in men and women with diabetes compared with non-diabetic people: a population-based retrospective cohort study. Lancet 2006; 368: 29–36
Turner RC, Millns H, Neil HA, et al. Risk factors for coronary artery disease in non-insulin dependent diabetes mellitus: United Kingdom Prospective Diabetes Study (UKPDS: 23). BMJ 1998; 316: 823–8
Fuller JH, Stevens LK, Wang SL. Risk factors for cardiovascular mortality and morbidity: the WHO Multinational Study of Vascular Disease in Diabetes. Diabetologia 2001; 44 Suppl. 2: S54-64
Bartnik M, Ryden L, Ferrari R, et al. The prevalence of abnormal glucose regulation in patients with coronary artery disease across Europe: the Euro Heart Survey on diabetes and the heart. Eur Heart J 2004; 25: 1880–90
Chyun D, Vaccarino V, Murillo J, et al. Acute myocardial infarction in the elderly with diabetes. Heart Lung 2002; 31: 327–39
Mamcarz A. Chmielewsky M. Braksator W, et al. Factors influencing cardiac complications in patients with type-2 diabetes mellitus and silent myocardial ischeamia: five-year follow-up. Pol Arch Med Wewn 2004; 112: 1433–43
Chyun D, Obata J, Kling J, et al. In-hospital mortality after acute myocardial infarction in patients with diabetes mellitus. Am J Crit Care 2000; 9: 168–79
Lenzen M, Ryden L, Oehrvik J, et al. Diabetes known or newly detected, but not impaired glucose regulation, has a negative influence on 1-year outcome in patients with coronary artery disease: a report from the Euro Heart Survey on diabetes and the heart. Eur Heart J 2006; 27: 2969–74.
Corbett CF, Cook D, Setter SM. OASIS and beyond: improving outcomes for home health patients. Diabetes Educ 2003; 29: 83–9
Huxley R, Barzi F, Woodward M. Excess risk of fatal coronary heart disease associated with diabetes in men and women: analysis of 37 prospective cohort studies. BMJ 2006; 332: 73–8
Juutilainen A, Kortelainen S, Lehto S, et al. Gender difference in the impact of type 2 diabetes on coronary heart disease risk. Diabetes Care 2004; 27: 2898–904
Bartnick M, Malmberg K, Norhammar A, et al. Newly detected abnormal glucose tolerance: an important predictor of long-term outcome after myocardial infarction. Eur Heart J 2004; 25: 1990–7
Norhammar A, Tenerz A, Nilsson G, et al. Glucose metabolism in patient with acute myocardial infarction and no previous diagnosis of diabetes mellitus: a prospective study. Lancet 2002; 359: 2140–4
Wallander M, Bartnick M, Efendic S, et al. Beta cell dysfunction in patients with acute myocardial infarction but without previously known type 2 diabetes: a report from the GAMI study. Diabetologia 2005; 48: 2229–35
Haffner SN, Lehto S, Ronnemaa T, et al. Mortality from coronary heart disease in subjects with type 2 diabetes and in nondiabetic subjects with and without prior myocardial infarction. N Engl J Med 1998; 339: 229–34
Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive summary of the third report of the National Cholesterol Education Program (NCEP). JAMA 2001; 285: 2486–97
Evans JM, Wang J, Morris AD. Comparison of cardiovascular risk between patients with type 2 diabetes and those who had had a myocardial infarction. BMJ 2002; 324: 939–42
Danaei G, Lawes CM, Vander Hoorn S, et al. Global and regional mortality from ischaemic heart disease and stroke attributable to higher-than-optimum blood glucose concentration: comparative risk assessment. Lancet 2006; 368: 1651–9
Jorgensen H, Nakayama H, Raaschou HO, et al. Stroke in patients with diabetes: the Copenhagen Stroke Study. Stroke 1994; 25: 1977–84
Laing SP, Swerdlow AJ, Carpenter LM, et al. Mortality from cerebrovascular disease in a cohort of 23000 patients with insulin-treated diabetes. Stroke 2003; 34: 418–21
Mulnier HE, Seaman HE, Raleigh VS, et al. Risk of stroke in people with type 2 diabetes in the UK: a study using the general practice research database. Diabetologia 2006; 49: 2859–65
Karapanayiotides T, Piechowsky-Jozwiak B, van Melle G, et al. Stroke patterns, ethiology and prognosis in patients with diabetes mellitus. Neurology 2004; 62: 1558–62
Hu G, Sarti C, Jousilahti P, et al. The impact of history of hypertension and type 2 diabetes at baseline on the incidence of stroke and stroke mortality. Stroke 2005; 36: 2538–43
Hillen T, Coshal C, Tilling K, et al., on behalf of the South London Stroke Register. Cause of stroke recurrence is multifactorial: patterns, risk factors, and outcome of stroke recurrence in the South London Stroke Register. Stroke 2003; 34: 1457–63
Kragsterman B, Bjorck M, Lindbaeck J, et al. Long-term survival after carotid endarterectomy for asymptomatic stenosis. Stroke 2006; 37: 2886–91
Rajala U, Laakso M, Paivansalo M, et al. Blood pressure and atherosclerotic plaques in carotid, aortic and femoral arteries in elderly Finns with diabetes mellitus or impaired glucose tolerance. J Hum Hypertens 2005; 19: 85–91
Mohaved MR, Hashemzadeh M, Jamal MM. Diabetes mellitus is a strong, independent risk factor for atrial fibrillation and flutter in addition to other cardiovascular disease. Int J Cardiol 2005; 105: 315–8
Lip GYH, Hart RG, Conway DSG. Antithrombotic therapy for atrial fibrillation. BMJ 2002; 325: 1022–5
Fonarow GC, Srikanthan P. Diabetic cardiomyopathy. Endocrinol Metab Clin North Am. 2006; 35: 575–99
Arauz A, Murillo L, Cantu C, et al. Prospective study of single and multiple lacunar infarct using magnetic resonance imaging: risk factors, recurrence and outcome in 175 consecutive cases. Stroke 2003; 34: 2453–8
Garg R, Chaudhuri A, Munschauer F, et al. Hyperglycemia, insulin and acute ischemic stroke: a mechanistic justification for a trial of insulin infusion therapy. Stroke 2006; 37: 267–73
American Diabetes Association. Peripheral arterial disease in people with diabetes. Diabetes Care 2003; 26: 3333–41
Jude EB, Oyibo SO, Chalmers N, et al. Peripheral arterial disease in diabetic and nondiabetic patients: a comparison of severity and outcome. Diabetes Care 2001; 24: 1433–7
Adler AI, Stevens RJ, Neil A, et al. UKPDS 59: hyperglycemia and other potentially modifiable risk factor for peripheral vascular disease in type 2 diabetes. Diabetes Care 2002; 25: 894–9
Marso SP, Hiatt WR. Peripheral arterial disease in patients with diabetes. J Am Coll Cardiol 2006; 7: 921–9
Vereanu I, Patrascu T. The diabetic foot: vasculopathy versus neuropathy. In: Cheta D, editor. Vascular involvement in diabetes. Basel: Karger, 2005: 715–26
Dormandy J, Heeck L, Vig S. Predicting which patients will develop chronic critical leg ischemia. Semin Vasc Surg 1999; 12: 138–41
Aboyans V. Criqui MH, Denenberg JO, et al. Risk factors for progression of peripheral arterial disease in large and small vessels. Circulation 2006; 6: 2623–9
Moss SE, Klein R, Klein BE. The 14-year incidence of lowerextremity amputation in a diabetic population: the Wisconsin Epidemiologic Study of Diabetic Retinopathy. Diabetes Care 1999; 22: 951–9
Lehto S, Niskanen L, Suhonen M, et al. Medial artery calcification: a neglected harbinger of cardiovascular complications in non-insulin-dependent diabetes mellitus. Arterioscler Thromb Vasc Biol 1996; 16: 978–83
Tentolouris N, Al-Sabbagh S, Walker MG, et al. Mortality in diabetic and nondiabetic patients after amputations performed from 1990 to 1995: a 5-year follow-up study. Diabetes Care 2004; 27: 1598–604
Jones EW, Mitchell JRA. Venous thrombosis in diabetes mellitus. Diabetologia 1983; 25: 502–5
Bergqvist D, Arnadottir M, Bergentz SE, et al. Juvenile diabetes mellitus a risk factor for postoperative venous thromboembolism? Acta Med Scand 1985; 217: 307–8
Petrauskiene V, Falk M, Waernbaum I, et al. The risk of venous thromboembolism is markedly elevated in patients with diabetes. Diabetologia 2005; 48: 1017–21
Heit JA, Silverstein MC, Mohr DN, et al. The epidemiology of venous thromboembolism in the community. Thromb Haemost 2001;86: 452–63
White RH, Gettner S, Newman JM, et al. Predictors of rehospitalization for symptomatic venous thromboembolism after total hip arthroplasthy. N Engl J Med 2000; 343: 1758–64
Kahn SR, Kearon C, Yulian JA, et al. Predictors of the post-thrombotic syndrome during long-term treatment of proximal deep vein thrombosis. J Thromb Haemost 2005; 3: 718–23
Bo S, Ciccone G, Rosato R, et al. Renal damage in patients with type 2 diabetes: a strong predictor of mortality. Diabet Med 2005; 22: 258–65
Spranger J, Pfeiffer AFH. Diabetic microvascular complications In: Morgensen CE, editor. Hypertension and diabetes. London: Lippincott Williams & Wilkins, 2002
Tarchini R, Bottini E, Botti P, et al. Type 2 diabetic nephropathy: clinical course and prevention, proposal, 2004. G Ital Nefrol 2005; 22: S15–19
Gambaro G, van der Woude FJ. Glycosaminoglycans: use in treatment of diabetic nephropathy. J Am Soc Nephrol 2000; 11: 359–68
Gimeno Orna JA, Boned Juliani B, Lou Arnal LM, et al. Microalbuminuria and clinical proteinuria as the main predicting factors of cardiovascular morbidity and mortality in patients with type 2 diabetes. Rev Clin Esp 2003; 203: 526–31
Trevisan R, Viberti G. Pathophysiology of diabetic nephropathy. In: LeRoith D, Taylor SI, Olefsky JM, editors. Diabetes mellitus: a fundamental and clinical text. Philadelphia (PA): Lippincott Williams & Wilkins, 2000
Koch M, Kutkuhn B, Grabensee B, et al. Apolipoprotein A, fibrinogen, age and history of stroke are predictors of death in dialyzed diabetic patients: a prospective study in 412 subjects. Nephrol Dial Transplant 1997; 12: 2603–11
Scheen AJ. Prevention of type 2 diabetes mellitus through inhibition of the renin-angiotensin system. Drugs 2004; 64: 2537–65
Jandeleit-Dahm KA, Tikellis C, Reid CM, et al. Why blockade of the renin-angiotensin system reduces the incidence of new-onset diabetes? Hypertension 2005; 23: 463–73
Scheen AJ. Renin-angiotensin system inhibition prevents type 2 diabetes mellitus, part 2: overview of physiological and biochemical mechanisms. Diabetes Metab 2004; 30: 498–505
Klein R, Klein BE, Moss SE. Epidemiology of proliferative diabetic retinopathy. Diabetes Care 1992; 15: 1875–91
Lyons TJ, Jenkins AJ, Zheng D, et al. Diabetic retinopathy and serum lipoprotein subclasses in the DCCT/EDIC cohort. Invest Ophthalmol Vis Sci 2004; 45: 910–8
Moss SE, Klein R, Klein BE. Ten-year incidence of visual loss in a diabetic population. Ophthalmology 1994; 101: 1061–70
Klein R, Klein BE, Moss SE, et al. The Wisconsin Epidemiologic Study of Diabetic Retinopathy, XV: the long-term incidence of macular oedema. Ophthalmology 1995; 102: 7–16
Juutilianen A, Lehto S, Ronnemaa T, et al. Retinopathy predicts cardiovascular mortality in type 2 diabetic men and women. Diabetes Care 2007; 30: 292–9
Vinik A, Mitchell B, Maser R, et al. Diabetic autonomie neuropathy. Diabetes Care 2003; 26: 1553–62
Wackers FJT, Young LH, Inzucchi SE, et al. Detection of silent myocardial ischemia in asymptomatic diabetic subjects: the DIAD Study. Diabetes Care 2004; 27: 1954–61
Sampson MJ, Wilson S, Karagiannis P, et al. Progression of diabetic autonomie neuropathy over a decade in insulin-dependent diabetics. Q J Med 1990; 75: 635–46
Deckert T, Feldt-Rasmussen D, Borch-Johnsen K, et al. Albuminuria reflects widespread vascular damage: the Steno hypothesis. Diabetologia 1989; 32: 219–26
Sun M, Yokoyama M, Ishiwata T, et al. Deposition of advanced glycation end-products (AGE) and expression of the receptor for AGE in cardiovascular tissue of the diabetic rat. Int J Exp Pathol 1998; 79: 207–22
Zee RYL, Romero JR, Gould JL, et al. Polymorphisms in the advanced glycosylation end product-specific receptor gene and risk of incident myocardial infarction or ischemic stroke. Stroke 2006; 37: 1686–90
Cahuana GM, Tejedo JR, Jimenez J, et al. Involvement of advanced lipoxidation end products (ALE) and protein oxidation in the apoptotic actions of nitric oxide in insulin secreting RINm5F cells. Biochem Pharmacol 2003; 15: 1963–71
Simionescu N, Popov D. Endothelial dysfunction in diabetes. In: Cheta D, editor. Vascular involvement in diabetes. Basel: Karger, 2005: 15–34
Gaede P, Vedel P, Larsen N, et al. Multifactorial intervention and cardiovascular disease in patients with type 2 diabetes. N Engl J Med 2003; 348: 383–93
Goldsmith DJA, Smith A, Bakri K, et al. Endothelial dysfunction, atherosclerosis and arteriosclerosis in diabetic patients with and without renal involvement. In: Cheta D, editor. Vascular involvement in diabetes. Basel: Karger, 2005: 289–301
Giugliano D, Ceriello A, Paolisso G. Oxidative stress and diabetic vascular complications. Diabetes Care 1996; 19: 257–67
Hogikyan RV, Galecki AT, Pitt B, et al. Specific impairment of endothelium-dependent vasodilation in subjects with type 2 diabetes independent of obesity. J Clin Endocrinol Metab 1998; 83: 1946–52
O’Neill MS, Veves A, Zanobetti A, et al. Diabetes enhances vulnerability to particulate air pollution-associated impairment in vascular reactivity and endothelial function. Circulation 2005; 111: 2913–20
Chappey O, Dosquet C, Wautier PM, et al. AGE products, oxidant stress and vascular lesions. Eur J Clin Invest 1997; 27: 97–108
Schmidt AM, Yan SD, Wautier JL, et al. Activation of receptor for advanced glycation end-products: a mechanism for chronic vascular dysfunction in diabetic vasculopathy and atherosclerosis. Circ Res 1999; 84: 489–97
Shoelson SE, Lee J, Goldfine AB. Inflammation and insulin resistance. J Clin Invest 2006; 116: 1793–801
Semenkovich CF. Insulin resistance and atherosclerosis. J Clin Invest 2006; 116: 1813–22
Viles-Gonzales JF, Fuster V, Badimon JJ. Links between inflammation and thrombogenicity in atherosclerosis. Curr Mol Med 2006; 6: 489–99
Madonna R, Pandolfi A, Massaro M, et al. Insulin enhances vascular cell adhesion molecule (VCAM-1) expression in human cultured endothelial cells through a pro-atherogenic p38MAP-kinase-mediated pathway. Diabetologia 2004; 47: 532–6
Walcher D, Aleksic M, Jerg V, et al. C-peptide induces chemotaxis of human CD4 positive cells: involvement of pertussis toxin-sensitive G-proteins and phosphoinositide 3-kinase. Diabetes 2004; 53: 1664–70
Marx N, Walcher D, Raichle C, et al. C-peptide colocalizes with macrophages in early arteriosclerotic lesions of diabetic subjects and induces monocyte chemotaxis in vitro. Arterioscler Thromb Vasc Biol 2004; 24: 540–5
Festa A, D’Agostino R Jr, Howard G, et al. Chronic subclinical inflammation as part of the insulin resistance syndrome: the Insulin Resistance Atherosclerosis Study (IRAS). Circulation 2000; 102: 42–7
Sjoholm A, Nystrom T. Endothelial inflammation in insulin resistance. Lancet 2005; 365: 610–2
Nechifor M. Involvement of eicosanoid metabolism in vascular complications of diabetes mellitus. In: Cheta D, editor. Vascular involvement in diabetes. Basel: Karger, 2005: 63–80
Davi G, Catalano I, Averna M. Thromboxane biosynthesis and platelet function in type 2 diabetes mellitus. N Engl J Med 1990; 322: 1769–74
Hanasaki K, Nakano K, Kasai H, et al. Specific receptors for thromboxane A2 in cultured vascular endothelial cells in rat aorta. Biochem Biophys Res Commun 1988; 151: 1352–7
Ford-Hutchinson AW. Leukotrienes: their formation and role as inflammatory mediators. Fed Proc 1985; 44: 25–45
Brezinsky ME, Gimbrone MA, Nicolaou KC. Lipoxins stimulate prostacyclin generation by human endothelial cells. FEBS Lett 1989; 245: 167–72
Liu T, Stern A, Roberts LJ, et al. Isoprostanes: novel prostaglandin-like products of the free radical-catalysed peroxidation of arachidonic acid. J Biomed Sci 1999; 6: 226–35
Ostermann H, Van de Loo J. Factors of the haemostatic system in diabetic patients: a survey of controlled studies. Heamostasis 1986; 16: 386–416
Di Minno G, Silver MJ, Cerbone AM, et al. Trial of repeated low-dose aspirin in diabetic angiopathy. Blood 1986; 68: 886–91
Tan KT, Tayebjee MH, Lim HS, et al. Clinically apparent atherosclerotic disease in diabetes is associated with an increase in platelet microparticle levels. Diabet Med 2005; 22: 1657–62
Bavbek N, Kargili A, Kaftan O, et al. Elevated concentrations of soluble adhesion molecules and large platelets in diabetic patients: are they markers of vascular disease and diabetic nephropathy? Clin Appl Thromb Haemost 2007. In press
Tschoepe D, Roesen P, Schwippert B, et al. Platelets in diabetes: the role in the haemostatic regulation and atherosclerosis. Sem Thromb Haemost 1993; 19: 122–8
Lecrubier C, Scarabin PY, Grauso F, et al. Platelet aggregation related to age in diabetes mellitus. Haemostasis 1980; 9: 43–51
Ferroni P, Basili S, Falco A, et al. Platelet activation in type 2 diabetes mellitus. J Thromb Haemost 2004; 2: 1282–91
Ceriello A. Coagulation activation in diabetes mellitus: the role of hyperglycaemia and therapeutic prospects. Diabetologia 1991; 34: 457–62
Ceriello A. Fibrinogen and diabetes mellitus: is it time for intervention trials? Diabetologia 1997; 33: 731–4
Sommeijer DW, Hansen HR, van Oerle R, et al. Soluble tissue factor is a candidate marker for progression of microvascular disease in patients with type 2 diabetes. J Thromb Haemost 2006; 4: 574–80
Sauls DL, Banini AE, Boyd LC, et al. Elevated prothrombin level and shortened clotting times in subjects with type 2 diabetes. J Thromb Haemost 2007; 5: 638–9
Ceriello A, Giugliano D, Quatraro A, et al. Evidence for a hyperglycaemia-dependent decrease of antithrombin III: thrombin complex formation in humans. Diabetologia 1990; 33: 163–7
Gonzalez J, Colwell JA, Sarji KE, et al. Effect of metabolic control with insulin on plasma von Willebrand factor activity (VIII R: WF) in diabetes mellitus. Thromb Res 1980; 17: 261–6
Juhan-Vague I, Roul C, Alessi MC, et al. Increased plasma plasminogen activator inhbitor-1 levels: a possible link between insulin resistance and atherothrombosis. Diabetologia 1991; 34: 457–62
Hornsby WG, Boggess KA, Lyons TJ, et al. Hemostatic alterations with exercise conditioning in NIDDM. Diabetes Care 1990; 13: 87–92
Jokl R, Klein RL, Lopes-Virella MF, et al. Release of platelet plasminogen activator inhibitor 1 in whole blood is increased in patients with type 2 diabetes. Diabetes Care 1995; 18: 1150–5
Schneider DJ, Nordt TK, Sobel BE. Stimulation by proinsulin of expression of plasminogen activator inhibitor type 1 in endothelial cells. Diabetes 1992; 41: 890–5
Seljeflot I, Larsen JR, Dahl-Jorgensen K, et al. Fibrinolytic activity is highly influenced by long-term glycemie control in type 1 diabetic patients. J Thromb Haemost 2006; 4: 686–8
Machin A, Silverman SH, Lip GYH. Peripheral vascular disease and Virchow’s triad for thrombogenesis. Q J Med 2002; 95: 199–210
Carr ME. Diabetes mellitus: a hypercoagulable state. J Diabetes Complications 2001; 15: 44–54
Adler AI, STratton IM, Neil HA, et al. Association of systolic blood pressure with macrovascular and microvascular complications of type 2 diabetes (UKPDS 36): prospective observational study. BMJ 2000; 321: 412–9
Rossi R, Turco V, Origliani G, et al. Type 2 diabetes mellitus is a risk factor for the development of hypertension in postmenopausal women. J Hyperten 2006; 24: 2017–22.
Zhang Y, Lee ET, Devereux RB, et al. Prehypertension, diabetes, and cardiovascular disease risk in a population-based sample: the Strong Heart Study. Hypertension 2006; 47: 410–4
Allemann S, Diem P, Egger M, et al. Fibrates in the prevention of cardiovascular disease in patients with type 2 diabetes mellitus: meta-analysis of randomised controlled trials. Curr Med Res Opin 2006 22; 617–23
Kahn HS. The lipid accumulation product is better than BMI for identifying diabetes: a population-based comparison. Diabetes Care 2006; 29: 151–3
Haire-Joshu D, Glassow RE, Tibbs TL. Smoking and diabetes. Diabetes Care 1999; 22: 1887–989
Lonn E, Yusuf S, Arnold MJ, et al. Homocysteine lowering with folic acid and B vitamins in vascular disease. N Engl J Med 2006; 354: 1567–77
Violi F, Criqui M, Longoni A, et al. Relation between risk factors and cardiovascular complications in patients with peripheral vascular disease: results from the ADEP study. Atherosclerosis 1996; 120: 25–35
Despres JP, Lamarche B, Mauriege P, et al. Hyperinsulinemia as an independent risk factor for ischemic heart disease. N Engl J Med 1996; 334: 952–7
Zethelius B, Lithel H, Hales CN, et al. Insulin sensitivity, pro insulin and insulin as predictors of coronary heart disease: a population-based 10-year follow-up study in 70-year old men using the euglycaemic insulin clamp. Diabetologia 2005; 48: 862–7
Bonora E, Formentini G, Calcaterra F, et al. HOMA-estimated insulin resistance is an independent predictor of cardiovascular disease in type 2 diabetic subjects: prospective data from the Verona Diabetes Complications Study. Diabetes Care 2002; 25: 1135–41
Stratton IM, Adler AI, Neil HA, et al. Association of glycaemia with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35): prospective observational study. BMJ 2000; 321: 405–12
Meerwaldt R, Lutgers HL, Links TP, et al. Skin autofluorescence is a strong predictor of cardiac mortality in diabetes. Diabetes Care 2007; 30: 107–12
Leelawattana R, Rattarasarn C, Lim A, et al. Causes of death, incidence and risk factors of cardiovascular disease in Thai type 2 diabetic patients: a 5 year follow-up study. Diabetes Res Clin Pract 2003; 60: 183–9
Chiodini I, Adda G, Scillitani A, et al. Cortisol secretion in patients with type 2 diabetes: relationship with chronic complications. Diabetes Care 2007; 30: 83–8
Linneman B, Janka HU. Prolonged QTc interval and elevated heart rate identify the type 2 diabetic patient at high risk for cardiovascular death: the Bremen Diabetes Study. Exp Clin Endocrinol Diabetes 2003; 111: 215–22
Banskota NK, Taub R, Zeller K, et al. Insulin, insulin-like growth factor I, and platelet-derived growth factor interact additively in the induction of the protoncogene c-myc and cellular proliferation in cultured bovine aortic smooth muscle cells. Mol Endocrinol 1989; 8: 1183–90
Cimminiello C, Milani M. Diabetes mellitus and peripheral vascular disease: is aspirin effective in preventing vascular events? Diabetologia 1996; 39: 1402–4
Sivenius J, Laakso M, Riekkeinen P, et al. European stroke prevention study: effectiveness of antiplatelet therapy in diabetic patients in secondary prevention of stroke. Stroke 1992; 23: 851–4
Cimminiello C. Is aspirin effective in diabetic patients? No. J Thromb Haemost 2005; 3: 2615–6
Sacco M, Pellegrini F, Roncaglioni MC, et al., on behalf of the PPP Collaborative Group. Primary prevention of cardiovascular events with low-dose aspirin and vitamin E in type 2 diabetic patients: results of the Primary Prevention Project (PPP) trial. Diabetes Care 2003; 26: 3264–72
ETDRS Investigators. Aspirin effects on mortality and morbidity in patients with diabetes mellitus: Early Treatment Diabetic Retinopathy Study report 14. JAMA 1992; 268: 1292–300
The DAMAD Study Group. Effect of aspirin alone and aspirin plus dipyridamole in early diabetic retinopathy: a multicenter randomized controlled clinical trial. Diabetes 1989; 38: 491–8
Antithrombotic Trialists’ Collaboration. Collaborative Meta-analysis of randomised trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients. BMJ 2002; 324: 71–86
Colwell JA, Bingham SF, Abraira C, et al. Veterans Administration Cooperative Study on antiplatelet agents in diabetic patients after amputation for gangrene, II: effects of aspirin and dipyridamole on atherosclerotic vascular disease rates. Diabetes Care 1986; 9: 140–8
Diener HC, Bogousslavsky J, Brass LM, et al. Aspirin and clopidogrel compared with clopidogrel alone after recent ischaemic stroke or transient ischaemic attack in high-risk patients (MATCH): randomized, double-blind, placebo-controlled trial. Lancet 2004; 364: 331–7
Bhatt DL, Fox KA, Hacke W, et al. Clopidogrel and aspirin versus aspirin alone for the prevention of atherothrombotic events. N Engl J Med 2006; 354: 1706–17
Colwell JA. Is aspirin effective in diabetic patients? Yes. J Thromb Haemost 2005; 3: 2612–4
American Diabetes Association. Aspirin therapy in diabetes. Diabetes Care 2004; 27: S72–3
American Heart Association. AHA scientific statement: AHA guidelines for primary prevention of cardiovascular disease and stroke -2002 update. Circulation 2002; 106: 288–91
US Preventive Services Task Force. Aspirin for the primary prevention of cardiovascular events: summary of the evidence. Ann Intern Med 2002; 136: 161–72
The Task Force on the Use of Antiplatelet Agents in Patients with Atherosclerotic Cardiovascular Disease of the European Society of Cardiology. Expert consensus document on the use of antiplatelet agents. Eur Heart J 2004; 25: 166–81
Song SH, Brown PM. Coronary heart disease risk assessment in diabetes mellitus: comparison of the UKPDS risk engine with Framingham risk assessment function and its clinical implications. Diabetic Med 2004; 21: 238–45
Colwell JA. Aspirin therapy in diabetes is underutilized. Diabetes Care 2001; 24: 195–6
Neri Serneri GG, Coccheri S, Marubini E, et al., on behalf of the the Drug evaluation in Atherosclerotic Vascular disease In Diabetics (DAVID) Study Group. Picotamide, a combined inhibitor of thromboxane A2 synthase and receptor, reduces 2-year mortality in diabetics with peripheral arterial disease: the DAVID Study. Eur Heart J 2004; 25: 1845–52
Cleland JGF. Preventing atherosclerotic events with aspirin. BMJ 2002; 324: 103–5
Bhatt DL, Marso SP, Hirsh AT, et al. Amplified benefit of clopidrogrel versus aspirin in patients with diabetes mellitus. Am J Cardiol 2002; 90: 625–8
Angiolillo DJ, Bernardo E, Ramirez C, et al. Insulin therapy is associated with platelet dysfunction in patients with type 2 diabetes mellitus on dual oral antiplatelet treatment. J Am Coll Cardiol 2006; 48: 298–304
Sibbing D, von Beckerath O, Schomig A, et al. Diabetes mellitus and platelet function after administration of aspirin and a single 600mg dose of clopidogrel. J Thromb Hemost 2006; 4: 2566–8
Angiolillo DJ, Shoemaker SB, Desai B, et al. Randomized comparison of a high clopidogrel maintenance dose in patients with diabetes mellitus and coronary artery disease: results of the Optimizing Antiplatelet Therapy in Diabetes Mellitus (OP-TIMUS) study. Circulation 2007; 115: 708–16
Roffi M, Chew DP, Mukherjee D, et al. Platelet glycoprotein IIb-IIIa inhibitors reduce mortality in diabetic patient with non-ST-segment-elevation acute coronary syndromes. Circulation 2001; 104: 2767–71
Colwell JA. Antiplatelet agents for the prevention of cardiovascular disease in diabetes mellitus. Am J Cardiovasc Drugs 2004; 4: 87–106
TIMAD Study Group. Ticlopidine treatment reduces the progression of non-prolipherative diabetic retinopathy. Arch Ophthalmol 1990; 108: 1577–83
Early Treatment Diabetic Retinopathy Study Research Group. Effects of aspirin treatment on diabetic retinopathy: ETDRS report no. 8. Ophthalmology 1991; 98: 757–65
Otsuki M, Hashimoto K, Morimoto Y, et al. Circulating vascular cell adhesion molecule-1 (VCAM-1) in atherosclerotic NIDDM patients. Diabetes 1997; 46: 2096–101
Ceriello A, Motz E. Prevention of vascular events in diabetes mellitus: which ‘antithrombotic’ therapy? Diabetologia 1996; 39: 1405–6
Meade TW, Brennan PJ. Determination of who may derive most benefit from aspirin in primary prevention: subgroup results from a randomised controlled trial. BMJ 2000; 321: 13–7
Pulcinelli FM, Riondino S, Celestini A, et al. Persistent production of platelet thromboxane A2 in patients chronically treated with aspirin. J Thromb Haemost 2005; 3: 2784–9
Watala C, Golansky J, Pluta J, et al. Reduced sensitivity of platelets from type 2 diabetes patients to acetylsalicylic acid (aspirin): its relation to metabolic control. Thromb Res 2004; 113: 101–13
Di Minno G, Violi F. Aspirin resistance and diabetic angiopathy: back to the future. Thromb Res 2004; 113: 97–9
Fateh-Moghadam S, Plockinger U, Cabeza N, et al. Prevalence of aspirin resistance in patients with type 2 diabetes. Acta Diabetol 2005; 42: 99–103
Pulcinelli FM, Biasucci LM, Giubilato S, et al. Reduced platelet sensitivity to aspirin in type 2 diabetic subjects compared to nondiabetics. American College of Cardiology 55th Annual Scientific Session; 2006 Mar 11–14; Atlanta (GA): presentation 1023-165
Watala C, Pluta J, Golansky J, et al. Increased protein glycation in diabetes mellitus is associated with decreased aspirin-mediated protein acetylation and reduced sensitivity of blood platelets to aspirin. J Mol Med 2005; 83: 148–58
Mullarkey CJ, Edelstein D, Brownlee M. Free radical generation by early glycation products: a mechanism for accelerated atherogenesis in diabetes. Biochem Biophys Res Commun 1990; 173: 932–9
Neri Serneri GG, Abbate R, Gensini GF, et al. TXA2 production by human arteries and veins. Prostaglandins 1983; 25: 753–65
Samama M, Conard J. Antithrombotic therapy in unique patient cohorts: diabetes, pregnancy, hypercoagulable syndromes, renal insufficiency. In: Kristensen SD, De Caterina R, Moliterno DJ, editors. Therapeutic strategies in thrombosis. Oxford: Clinical Publishing, 2006
Bueller HR, Agnelli G, Hull RD, et al. Antithrombotic therapy for venous thromboembolic disease: the 7th ACCP Conference on antithrombotic and thrombolytic therapy. Chest 2004; 126 (3 Suppl.): 401-28S
Atrial Fibrillation Investigators. Risk factors for stroke and efficacy of antithrombotic therapy in atrial fibrillation: analysis of pooled data from five randomised control trials. Arch Intern Med 1994; 154: 1449–57
Varughese GI, Patel JV, Tomson J, et al. The prothrombotic risk of diabetes mellitus in atrial fibrillation and heart failure. J Thromb Haemost 2005; 3: 2811–3
Wehinger C, Stollberger C, Langer T, et al. Evaluation of risk factors for stroke/embolism and of complications due to anticoagulant therapy in atrial fibrillation. Stroke 2001; 32: 2246–52
Wittkowsky AK, Warfarin and other coumadin derivatives: pharmacokinetics, pharmacodynamics and drug interactions. Sem Vasc Med 2003; 3: 221–30
Gambaro G, Baggio B. Growth factors and the kidney in diabetes mellitus. Crit Rev Clin Lab Sci 1998; 35: 117–51
Wolf G, Ziyadeh FN, Zahner G, et al. Angiotensin II-stimulated expression of transforming growth factor beta in renal proximal tubular cells: attenuation after stable transfection with cmass oncogene. Kidney Int 1995; 48: 1818–27
Lovell HG. Angiotensin converting enzyme inhibitors in normotensive diabetic patients with microalbuminuria. Coch-rane Database Syst Rev 2001; 1: CD002183
Vernier RL, Steffes MW, Sissons-Ross S, et al. Heparan-sulphate proteoglycan in the glomerular membrane basement in type 1 diabetes mellitus. Kidney Int 1992; 41: 1070–80
Wasty F, Alavi MZ, Moore S. Distribution of glycosaminogly-cans in the intima in the human aorta: changes in atherosclerosis and diabetes mellitus. Diabetologia 1993; 36: 316–22
Myrup B, Hansen PM, Jensen T, et al. Effect of low-dose heparin on urinary albumin excretion in insulin-dependent diabetes mellitus. Lancet 1995; 345: 421–2
Van der Pijl JW, van der Woude FJ, Geelhoed-Duijvestijn PHLM, et al. Danaparoid sodium lowers proteinuria in diabetic nephropathy. J Am Soc Nephrol 1997; 8: 456–62
Gambaro G, Skrha J, Ceriello A. Glycosaminoglycan therapy for long-term diabetic complications? Diabetologia 1998; 41: 975–9
Gambaro G. Cavazzana AO, Luzi P, et al. Glycosaminoglycans prevent morphological renal alterations and albuminuria in diabetic rats. Kidney Int 1992; 42: 285–91
Gambaro G, Kinalska I, Oksa A, et al. Oral sulodexide reduces albuminuria in microalbuminuric and macroalbuminuric type I and type 2 diabetic patients: the Di.N.A.S. randomized trial. J Am Soc Nephrol 2002; 13: 1615–25
Achour A, Kacem M, Dibej K, et al. One year course of oral sulodexide in the management of diabetic nephropathy. J Nephrol 2005; 18: 568–74
Condorelli M, Chiariello M, Dagianti A, et al. IPO-V2: a prospective multicenter, randomized, comparative clinical investigation of effects of sulodexide in preventing cardiovascular accidents in the first year after acute myocardial infarction. J Am Coll Cardiol 1994; 23: 27–34
Coccheri S, Scondotto G, Agnelli G, et al. Sulodexide in the treatment of intermittent claudication: results of a randomized, double-blind, multicentre, placebo-controlled study. Eur Heart J 2002; 23: 1057–65
Boeri D, Maiello M, Lorenzi M. Increased prevalence of microthromboses in retinal capillaries of diabetic individuals. Diabetes 2001; 50: 1432–9
Cimponeriu D, Apostol P, Radu I, et al. Genetic bases of vascular complications in diabetes mellitus. In: Cheta D, editor. Vascular involvement in diabetes. Basel: Karger, 2005: 81–119
Henricsson M, Berntorp K, Fernlud P, et al. Progression of retinopathy in insulin-treated type 2 diabetic patients. Diabetes Care 2002; 25: 381–5
Negrisanu G, Diaconu L. Glycosaminoglycans in diabetic vasculopathy: hypotheses and current evidence. In: Cheta D, editor. Vascular involvement in diabetes. Basel: Karger, 2005: 625–44
Van der Pijl JW, van der Woude FJ, Swart W, et al. Effect of danaparoid sodium on hard exudates in diabetic retinopathy. Lancet 1997; 350: 1743–5
Buse JB, Ginsberg HN, Bakris GL, et al. Primary prevention of cardiovascular disease in people with diabetes mellitus: a scientific statement from the American Heart Association and the American Diabetes Association. Circulation 2007; 115: 114–26
Ryden L, Standl E, Bartnik M, et al., on behalf of Task Force Members. Guidelines on diabetes, pre-diabetes, and cardiovascular disease: executive summary. Eur Heart J 2007; 28: 88–136
UK Prospective Diabetes Study (UKPDS) Group. Effect of intensive blood-glucose control with metformin on complica-tions in overweight patients with type 2 diabetes (UKPDS 34). Lancet 1998; 352: 854–65
Vosper H, Khoudoli GA, Graham TL, et al. Peroxisome prolipherator-activated receptors agonists, hyperlipidemia and atherosclerosis. Pharmacol Ther 2002; 95: 47–62
Dormandy JA, Charbonnel B, Eckland DJ, et al. Secondary prevention of macrovascular events in patients with type 2 diabetes in the PROactive Study (PROspective pioglitAzone Clinical Trial In macroVascular Events): a randomized controlled trial. Lancet 2005; 366: 1279–89
Richter B, Bandeira-Echtler E, Bergerhoff K, et al. Pioglitazone for type 2 diabetes mellitus. Cochrane Database Syst Rev 2006; 18: CD006060
Gerstein HC, Yusuf S, Bosch J, et al. DREAM (Diabetes REduction Assessment with ramipril and rosiglitazone Medication) Trial Investigators. Effect of rosiglitazone on the frequency of diabetes in patients with impaired glucose tolerance or impaired fasting glucose: a randomised controlled trial. Lancet 2006; 368: 1096–105
Wannamethee SG, Shaper AG, Walker M. Overweight and obesity and weight change in middle aged men: impact on cardiovascular disease and diabetes. J Epidemiol Community Health 2005; 59: 134–9
Grundy SM, Cleeman JI, Mertz CN, et al. Implications of recent clinical trials for the National Cholesterol Education Program adult treatment: panel III guidelines. Circulation 2004; 110: 227–39
Vijan S, Hayward RA, American College of Physicians. Pharmacologic lipid-lowering therapy in type 2 diabetes mellitus: background paper for the American College of Physicians. Ann Intern Med 2004; 140: 650–8
Collins R, Armitage J, Parish S, et al., on behalf of the Heart Protection Study Collaborative Group. MRC/BHF Heart Protection Study of cholesterol-lowering with simvastatin in 5963 people with diabetes: a randomised placebo-controlled trial. Lancet 2003; 361: 2005–16
Colhoun HM, Betteridge DJ, Durrington PN, et al. Primary prevention of cardiovascular disease with atorvastatin in type 2 diabetes in the Collaborative Atorvastatin Diabetes Study (CARDS): multicentre randomised placebo-controlled trial. Lancet 2004; 364: 685–96
Knopp RG, d’Emden M, Smilde JG, et al. Efficacy and safety of atorvastatin in the prevention of cardiovascular end points in subjects with type 2 diabetes: the Atorvastatin Study for Prevention of Coronary Heart Disease Endpoints in Non-Insulin-Dependent Diabetes Mellitus (ASPEN). Diabetes Care 2006; 29: 1478–85
Shepherd J, Barter P, Carmena R, et al. Effect of lowering LDL cholesterol substantially below currently recommended levels in patients with coronary heart disease and diabetes: the Treating to New Targets (TNT) study. Diabetes Care 2006; 29: 1220–6
Wolffenbuttel BH, Franken AA, Vincent HH, et al. Cholesterol-lowering effects of rosuvastatin compared with atorvastatin in patients with type 2 diabetes: CORRAL study. J Intern Med 2005; 257: 531–9
Betteridge DJ, Gibson JM. Effects of rosuvastatin on lipids, lipoproteins and apolipoproteins in the dyslipidemia of diabetes. Diabet Med 2007 Mar 15. Epub
Goldberg RB, Guyton JR, Mazzone T, et al. Ezetimibe/simvastatin vs atorvastatin in patients with type 2 diabetes mellitus and hypercholesterolemia: the VYTAL study. Mayo Clin Proc 2006; 81: 1579–88
Chu CS, Lee KT, Lee MY, et al. Effects of rosiglitazone alone and in combination with atorvastatin on nontraditional markers of cardiovascular disease in patients with type 2 diabetes mellitus. Am J Cardiol 2006; 97: 646–50
Keech A, Simes RJ, Barter P, et al. Effects of long-term fenofibrate therapy on cardiovascular events in 9795 people with type 2 diabetes mellitus (the FIELD study): randomised controlled trial. Lancet 2005; 26: 1849–61
Diabetes Atherosclerosis Intervention Study Investigators. Effect of fenofibrate on progression of coronary-artery disease in type 2 diabetes: the Diabetes Atherosclerosis Intervention Study, a randomised study. Lancet 2001; 357: 905–10
Keating GM, Croom KF. Fenofibrate: a review of its use in primary dyslipidemia, the metabolic syndrome and type 2 diabetes mellitus. Drugs 2007; 67 (1): 121–53
Wierzbicki AS. FIELDS of dreams, fields of tears: a perspective on the fibrate trial. Int J Clin Pract 2006; 60: 442–9
Sacco RL, Adams R, Albers G, et al. Guidelines for prevention of stroke in patients with ischemic stroke or transient ischemic attack: a statement for healthcare professionals from the American Heart Association/American Stroke Association Council on Stroke. Stroke 2006; 37: 577–617
UK Prospective Diabetes Study (UKPDS) Group. Tight blood pressure control and risk of macrovascular and microvascular complications in type 2 diabetes (UKPDS 38). BMJ 1998; 317: 703–13
Hansson L, Lindholm LH, Niskanen L, et al. Effect of angiotensin-converting-enzyme inhibition compared with conventional therapy on cardiovascular morbidity and mortality in hypertension: the Captopril Prevention Project (CAPP) randomized trial. Lancet 1999; 353: 611–6
Tatti P, Pahor M, Byington RP, et al. Outcome results of the Fosinopril versus Amlodipine Cardiovascular Events randomized Trial (FACET) in patients with hypertension and NIDDM. Diabetes Care 1998; 21: 597–603
Goff DC. Landmark study: the ALLHAT Study. Clin Diabetes 2003; 21: 102–4
Heart Outcomes Prevention Evaluation Study Investigators. Effects of ramipril on cardiovascular and microvascular outcomes in people with diabetes mellitus: results of the HOPE study and MICRO-HOPE substudy. Lancet 2000; 355: 253–9
Verma S, Leiter LA, Lonn EM, et al. Perindopril in diabetes: perspective from the EUROPA substudy, PERSUADE. Eur Heart J 2005; 26: 1347–9
Solomon SD, Rice MM, A Jablonski K, et al. Renal function and effectiveness of angiotensin-converting enzyme inhibitor therapy in patients with chronic stable coronary disease in the Prevention of Events with ACE inhibition (PEACE) trial. Circulation 2006; 114: 26–31
Ruggenenti P, Fassi A, Ilieva AP, et al. Preventing microalbuminuria in type 2 diabetes: Bergamo Nephrologie Diabetes Complications Trial (BENEDICT) Investigators. N Engl J Med 2004; 351: 1941–51
Strippoli GF, Craig M, Schena FP, et al. Antihypertensive agents for primary prevention of diabetic nephropathy. J Am Soc Nephrol 2005; 16: 3081–91
Deferrari G, Ravera M, Deferrari L, et al. Renal and cardiovascular protection in type 2 diabetes mellitus: angiotensin II receptor blockers. J Am Soc Nephrol 2002; 3: S224–9
Lindholm LH, Ibsen H, Dahlof B. Devereux RB, et al. Cardiovascular morbidity and mortality in patients with diabetes in the Losartan Intervention For Endpoint reduction in hypertension study (LIFE): a randomised trial against atenolol. Lancet 2002; 359: 1004–10
Strippoli GF, Craig M, Deeks JJ, et al. Effects of angiotensin converting enzyme inhibitors and angiotensin II receptor antagonist on mortality and renal outcomes in diabetic nephropathy: systematic review. BMJ 2004; 329: 828
Fardoun RZ. Carvedilol versus cardioselective beta-blockers for the treatment of hypertension in patients with type 2 diabetes mellitus. Pharmacotheraphy 2006; 26: 1491–500
Poierer L, Cleroux J, Nadeau A, et al. Effects of nebivolol and atenolol on insulin sensitivity and haemodynamics in hypertensive patients. Hypertens 2001; 19: 1429–35
Scheen AJ. Renin-angiotensin system inhibition prevents type 2 diabetes mellitus, part 1: a meta-analysis of randomised clinical trials. Diabetes Metab 2004; 30: 487–96
McCall KL, Craddock D, Edwards K. Effect of angiotensin-converting enzyme inhibitors and angiotensin II type 1 receptor blockers on the rate of new-onset diabetes mellitus: a review and pooled analysis. Pharmacotherapy 2006; 29: 1297–306
Dahlof B, Sver PS, Poulter NR, et al. ASCOT Investigators. Prevention of cardiovascular events with an antihypertensive regimen of amlodipine adding perindopril as required versus atenolol adding bendroflumethiazide as required, in the Anglo-Scandinavian Cardiac Outcomes Trial-Blood Pressure Lowering Arm (ASCOT-BPLA): a multicentre randomised controlled trial. Lancet 2005; 366: 895–906
The DREAM Trial Investigators, Bosch J, Yusuf S, et al. Effect of ramipril on the incidence of diabetes. N Engl J Med 2006; 355: 1551–62
Elliot WJ, Meyer PM. Incident diabetes in clinical trials of antihypertensive drugs: a network meta-analysis. Lancet 2007; 369: 201–7
Williams ME, Tuttle KR. The next generation of diabetic nephropathy therapies: an update. Adv Chronic Kidney Dis 2005; 12: 212–22
Signorelli S, Neri S, Di Pino L, et al. Effect of PLC on functional parameters and oxidative profile in type 2 diabetes-associated PAD. Diabetes Res Clin Pract 2006; 72: 231–7
Ekberg K, Brismar T, Johansson BL, et al. C-peptide replacement therapy and sensory nerve function in type 1 diabetic neuropathy. Diabetes Care 2007; 30: 71–6
Acknowledgements
The author did not receive any specific funding for the preparation of this manuscript. The author has not received any research grants during the last 5 years from pharmaceutical companies that manufacture the drugs discussed in this review. The author would like to acknowledge the collaboration of Dr Donatella Orlando in the literature search and organisation of this review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Coccheri, S. Approaches to Prevention of Cardiovascular Complications and Events in Diabetes Mellitus. Drugs 67, 997–1026 (2007). https://doi.org/10.2165/00003495-200767070-00005
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-200767070-00005