Abstract
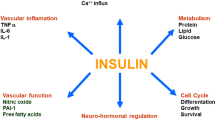
Resistance to the metabolic actions of insulin is thought to play a determining role in the aetiology of a great variety of disorders, including essential hypertension, accelerated atherosclerosis and cardiomyopathies. ACE inhibitors are recognised as being highly effective therapy for hypertension and cardiac insufficiency, and have a more beneficial effect on survival rate than expected on the basis of known mechanisms of action.
The mechanism responsible for these extremely positive effects are just beginning to be understood and appear to be linked to the effects these drugs have on metabolism. The relationship between the insulin and angiotensin II (Ang II) signalling pathways needs to be fully clarified in order to prevent or correct the target organ damage resulting from changes in the cross-talk of these two hormonal systems.
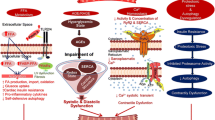
In recent years, Ang II has been shown to play a central role in cardiovascular and neuroendocrine physiology as well as in cellular cycle control. Moreover, the fact that Ang II utilises the insulin-receptor substrate (IRS)-1 to relay signals towards their intracellular destination, provides the biochemical explanation of how these two systems interact in a healthy organism and in a diseased one.
Since it is overactivity of the renin-angiotensin system that seems to impair the intracellular response to insulin signalling, cardiovascular drugs that modulate the cellular transmission of Ang II have attracted particular interest. As well as the already widely-used ACE inhibitors, selective blockers of the Ang II type 1 receptor (AT1) have been shown to be clinically effective in the control of haemodynamic parameters, but with perhaps a less striking effect on glucose homeostasis.
Many trials have investigated the effect of Ang II blockade on systemic glucose homeostasis. The inhibition of Ang II by ACE-inhibitors frequently showed a positive effect on glycaemia and insulin sensitivity, while information on the effects of AT1 receptor antagonists on glucose homeostasis is more limited and controversial. An important limitation of these studies has been the short treatment and follow-up periods, even for the “so called” long-term studies which were only 6 months.
Several investigators have focused on the effects of the nuclear factors involved in gene transcriptions, especially with respect to the agonists/antagonists of peroxisome proliferator-activated receptors (PPARs) and their intriguing interconnections with the insulin and Ang II subcellular pathways. In fact, in vitro and in vivo experimental studies have shown that thiazolidinediones (selective PPAR-γ ligands) are not only powerful insulin sensitisers, but also have anti-hypertensive and anti-atherosclerotic properties.
In addition to conventional pharmacological approaches, attempts have been made to use genetic transfer in the treatment of cardiovascular and metabolic disorders. The development of powerful viral vectors carrying target genes has allowed us to restore the expression/function of specific proteins involved in the cellular mechanism of insulin resistance, and research now needs to move beyond animal models.
Although a clearer picture is now emerging of the pathophysiological interaction between insulin and Ang II, especially from pre-clinical studies, there is much to be done before experimental findings can be used in daily clinical practice.










Similar content being viewed by others
References
Dzau VJ. Cell biology and genetics of angiotensin in cardiovascular disease. J Hypertens Suppl 1994; 12(4): S3–S10
Frossard M, Joukhadar C, Steffen G, et al. Paracrine effects of angiotensi-converting-enzyme and angiotensin-receptor-inhibiton on transcapillary glucose transport in human. Life Sci 2000; 66(10): 147–54
Velloso LA, Folli F, Sun XJ, et al. Cross-talk between the insulin and angiotensin signaling systems. Proc Natl Acad Sci U S A 1996; 93: 12490–5
Folli F, Kahn CR, Hansen H, et al. Angiotensin II inhibits insulin signaling in aortic smooth muscle cells at multiple levels. J Clin Invest 1997; 100: 2158–69
Buchanan TA, Thawani H, Kades W, et al. Angiotensin II increases glucose utilization during acute hyperinsulinemia via a hemodynamic mechanism. J Clin Invest 1993; 92: 720–6
Fliser D, Keller C, Bahrmann P, et al. Altered action of Angiotensin II in patients with type 2 diabetes mellitus of recent onset. J Hypertens 1997; 15(3): 293–9
Lithell HO, Polar T, Berne C. Insulin sensitivity in newly detected hypertensive patients: influence of captopril and other antihypertensive agents on insulin sensitivity and related biological parameters. J Cardiovasc Pharmacol 1990; 15: S46–52
Velasquez MT, Bhathena SJ, Striffler JS, et al. Role of angiotensin-converting-enzyme inhibition in glucose metabolism and renal injury in diabetes. Metabolism 1998; 47 (12 Suppl. 1); 7–11
Donnelly R. Angiotensin-converting-enzyme inhibitors and insulin sensitivity: metabolic effects in hypertension, diabetes and heart failure. J Cardiovasc Pharmacol 1992; 20: S38–44
Herings RM, de Boer A, Strieker BH, et al. Hypoglycaemia associated with use of inhibitors of angiotensin-converting-enzyme. Lancet 1995; 345:1195–8
Moan A, Risanger T, Eide I, et al. The effect of angiotensin II receptor blockade on insulin sensitivity and nervous system activity in primary hypertension. Blood Press 1994; 3(3): 185–8
Zavaroni I, Bonora E, Pagliara M, et al. Risk factors for coronary artery disease in healthy persons with hyperinsulinemia and normal glucose tolerance. N Engl J Med 1989; 320:702–6
Linseman DA, Benjamin CW, Jones DA. Convergence of angiotensin II and platelet-derived growth factor receptor signalling cascade in vascular smooth muscle cells. J Biol Chem 1995; 270(21): 12563–8
Nickening G, Roling J, Strehlow K, et al. Insulin induces up-regulation of vascular AT1 receptor gene expression by post-trascriptional mechanism. Circulation 1998; 98: 2453–60
Langendorff O. Untersuchungen am uberlebenden Saugethiertherzen. Arch Ges Physiol Menschen Tiere 1895; 61: 291–332
Opie LH. Metabolism of the heart in health and disease. Am Heart J 1968; 76: 685–9
Taegtmeyer H. Energy metabolism of the heart: from basic concepts to clinical applications. Curr Probl Cardiol 1994; 19: 59–113
Stanley WC, Lopaschuk GD, Hall JL, et al. Regulation of myocardial carbohydrate metabolism under normal and ischemic conditions: potential for pharmacological interventions. Cardiovasc Res 1997; 33: 243–57
Wolkowicz PE, Urthaler F, Forrest C, et al. 2-Tetradecylglycidic acid, an inhibitor of carnitine palmitoyltransferase-1, induces myocardial hypertrophy via the AT1 receptor. J Mol Cell Cardiol 1999 Aug; 31(8): 1405–12
Depre C, Vanoverschelde JLJ, Taegtmeyer H. Glucose for the heart. Circulation 1999; 99: 578–88
Goodwin GW, Taylor CS, Taegtmeyer H. Regulation of energy metabolism of the heart during acute increase in heart work. J Biol Chem 1998; 273: 29530–9
Allard MF, Schonekess BO, Henning SL, et al. Contribution of oxidative metabolism and glycolysis to ATP production in hyperthrophied hearts. Am J Physiol 1994; 267: H742–50
Olson AL, Pessin J. Structure, function and regulation of the mammalian facilitative glucose transporter gene family. Ann Rev Nutr 1996; 16:235–56
Doege H, Schurmann A, Baharenberg G, et al. Glut 8 a novel member of the sugar transport facilitator family with glucose transport activity. J Biol Chem 2000; 275(21): 16275–80
Doege H, Bocianski A, Joost HG, et al. Activity and genomic organization of human glucose transporter 9 (GLUT 9) a novel member of family of sugar-transport facilitators predominantly expressed in brain and leucocytes. J Biol Chem 2000; 350(3): 771–6
McVie-Wylie AJ, Lamson DR, Chen YT. Molecular cloning of anovel memberof the GLUT family of transporters SLC2a10 (GLUT 10), localized on chromosome 20ql3.1: a candidate gene for NIDDM susceptibility. Genomics 2001; 72(1): 113–7
Mueckler M. Facilitative glucose transporters. Eur J Biochem 1994; 219: 713–25
Zaninetti D, Greco-Perotto R, Jeanrenaud B. Heart glucose transport and transporters in rat heart: regulation by insulin, workload and glucose. Diabetologia 1988; 31: 108–13
Russell RR, Yin R, Caplan MJ, et al. Additive effects of hyperinsulinemia and ischemia on myocardial GLUT 1 and GLUT 4 translocation in vivo. Circulation 1998; 98: 2180–6
Neely JR, Morgan HE. Relationship between carbohydrate and lipid metabolism and the energy balance of heart muscle. Annu Rev Physiol 1974; 36: 413–59
Stanley WC, Lopaschuk GD, McCormack JG. Regulation of energy substrate metabolism in the diabetic heart. Cardiovasc Res 1997; 34: 25–33
Shapiro LM. Specific heart disease in diabetes mellitus. BMJ 1982; 284: 140–1
Tahiliani AG, McNeill JH. Diabetes-induced abnormalities in the myocardium. Life Sci 1986; 38: 959–74
Dhalla NS, Elimban V, Rupp H. Paradoxical role of lipid metabolism in heart function and dysfunction. Mol Cell Biochem 1992; 116:3–9
Kannel WB, Muller JE, Hartwell T, et al. The effect of diabetes mellitus on prognosis and serial left ventricular function after acute myocardial infarction: contribution of both coronary disease and diastolic left ventricular dysfunction to the adverse prognosis. J Am Coll Cardiol 1989; 14: 49–57
Lopaschuk GD, Belke DD, Gamble J, et al. Regulation of fatty acid oxidation in the mammalian heart in health and disease. Biochem Biophys Acta 1994; 1213: 263–76
Liu B, El Aloui-Talibi Z, Clanachan AS, et al. Uncoupling of contractile function from mithocondrial TCA cycle activity and MVO2 during reperfusion of ischemic hearts. Am J Physiol 1996; 260: H72–80
Liu B, Clanachan AS, Schulz R, et al. Cardiac efficiency is improved after ischemia by altering both the source and the fate of protons. Circ Res 1996; 79: 940–8
Lee J, Pilch PF. The insulin receptor: structure, function and signalling. Am J Physiol 1994; 266(35): C319–34
Myers MG, White ME Insulin signal transduction and the IRS proteins. Annu Rev Pharmacol Toxicol 1996; 36: 615–58
Moule KS, Denton RM. Multiple signalling pathways involved in the metabolic effects of insulin. Am J Cardiol 1997; 80(3): 41A-9A
Denton RM, McCormack JG, Rutter GA, et al. The hormonal regulation of pyruvate-dehydrogenase complex. Adv Enzyme Regul 1996; 36: 183–98
Cheatham B, Vlahos CJ, Cheatham L, et al. Phosphatidylinositol 3-kinase activation is required for insulin stimulation of pp70 S6 kinase, DNA synthesis, and glucose transporter translocation. Mol Cell Biol 1994; 14(7): 4902–11
Baker KM, Aceto JF. Angiotensin II stimulation of protein synthesis and cell growth in chick heart cells. Am J Physiol 1990; 259:H610–8
Baker KM, Booz GW, Dostal DE. Cardiac actions of angiotensin II: role of an intracardiac renin-angiotensin system. Annu Rev Physiol 1992; 54: 227–41
Low BC, Ross IK, Grigor MR. Angiotensin II stimulates glucose transport activity in cultured vascular smooth muscle cell. J Biol Chem 1992; 267(29): 20740–5
Tang W, Richards EM, Raizada MK, et al. Angiotensin II increases glucose uptake and glucose transporter-1 mRNA levels in astroglia. Am J Physiol 1995; 268 (3 Pt 1): E384–90
Nascimben L, Bothwell JH, Dominguez DY, et al. Angiotensin II stimulates insulin-independent glucose uptake in hypertrophied rat hearts [abstract]. J Hypertens 1997; 15Suppl.4: S84
Natarajan R, Scott S, Wei B, et al. Angiotensin II signalling in vascular smooth muscle cells under high glucose conditions. Hypertension 1999; 33: 378–84
Hoenack C, Roesen P. Inhibition of angiotensin type 1 receptor prevents decline of glucose transporter (Glut4) in diabetic rat heart. Diabetes 1996; 45 Suppl. 1: S82–7
Marrero MB, Schieffer B, Paxton WG, et al. Direct stimulation of Jak/STAT pathway by the Angiotensin II AT1 receptor. Nature 1995; 375: 247–50
Schorb W, Peeler TC, Madigan NN, et al. Angiotensin II-induced protein tyrosine phosphorylation in neonatal rat. J Biol Chem 1994; 269: 19626–32
Wan J, Kurosaki T, Huang XY, et al.; Tyrosine kinases in activation of the MAP-kinase cascade by G protein-coupled receptors. Nature 1996; 380: 541–4
Saad MJA, Velloso LA, Carvalho CRO. Angiotensin II induces tyrosine phosphorylation of insulin receptor substrate 1 and its association with phosphatidylinositol 3-kinase in rat heart. Biochem J 1995; 310: 741–4
Servant MJ, Giasson E, Meloche S. Inhibition of growth factor-induced protein synthesis by a selective MEK inhibitor in aortic smooth muscle cells. J Biol Chem 1996; 271:16047–52
Giles TD, Ouyang J, Kerut EK, et al. Changes in protein kinase C in early cardiomyopathy and in gracilis muscle in the BB/Wor diabetic rat. Am J Physiol 1998; 274 (1 Pt 2): H295–307
Liu X, Wang J, Takeda N, et al. Changes in protein kinase C activities and isozymes in streptozotocin-induced diabetes. Am J Physiol 1999; 277 (5 Pt 1): E798–804
Malhotra A, Barinder PS, Cheung S, et al. Angiotensin II promotes glucose-induced activation of cardiac protein kinase C isozymes and phosphorylation of troponin I. Diabetes 2001; 50: 1918–26
Pfeiffer A, Schatz H. Diabetic microvascular complications and growth factors. Exp Clin Endocrinol Diabetes 1995; 103(1): 7–14
Williams B. Glucose-induced vascular smooth muscle dysfunction: the role of protein kinase C. J Hypertens 1995; 18:477–86
Ishii H, Jirousek MR, Koya D, et al. Amelioration of vascular dysfunction in diabetic rats by an oral PKC beta inhibitor. Science 1996; 272(5262): 728–31
Fiordaliso F, Li B, Latini R, et al. Myocyte death in streptozotocin-induced diabetes in rats is Angiotensin II-dependent. Lab Invest 2000; 80: 513–27
Kajstura J, Cigola E, Malhotra A, et al. Angiotensin II induces apoptosis in adult ventricular myocytes via a PKC-dependent mechanism. J Moll Cell Cardiol 1997; 29: S59–70
Schenee Jm, Hsueh WA. Angiotensin II, adhesion, and cardiac fibrosis. Cardiovasc Res 2000; 46(2): 264–8
Edwards CR, Padfield PL. Angiotensin-converting-enzyme inhibitors: past, presence and bright future. Lancet 1985; I(8419): 30–4
Weinberg MH. Blood pressure and metabolic responses to hydrochlorothiazide, captopril and the combination in black and white mild-to-moderate hypertensive patients. J Cardiovasc Pharmacol 1985; 7: S52–5
Captopril Research Group of Japan. Clinical effects of low-dose Captopril plus a thiazide diuretic on mild to moderate essential hypertension: a multicenter double blind comparison with propanolol. J Cardiovasc Pharmacol 1985; 7: 552–5
McMurray J, Fraser DM. Captopril, enalapril and blood glucose [letter]. Lancet 1986; I(8488): 1035
Lederle RM. Captopril and hydrochlorothiazide in the fixed combination multicenter trial. J Cardiovasc Pharmacol 1985; 7: S63–9
Paolisso G, Gambardella A, Verza M, et al. ACE inhibition improves insulin-sensitivity in aged insulin-resistant hypertensive patients. J Hum Hypertens 1992 Jun; 6(3): 175–9
Bak JF, Gerdes LU, Sorensen NS, et al. Effects of perindopril on insulin sensitivity and plasma lipid profile in hypertensive non-insulin-dependent diabetic patients. Am J Med 1992; 92(4B): 69S–72S
Santoro D, Galvan AQ, Natali A, et al. Some metabolic aspects of essential hypertension and its treatment. Am J Med 1993; 94(4A): 32S–9S
Paolisso G, Balbi V, Gambardella A, et al. Lisinopril administration improves insulin action in aged patients with hypertension. J Hum Hypertens 1995 Jul; 9(7): 541–6
Thurig C, Bohlen L, Schneider M, et al. Lisinopril is neutral to insulin sensitivity and serum lipoproteins in essential hypertensive patients. Eur J Clin Pharmacol 1995; 49(1-2): 21–6
Vuorinen-Markolla H, Yki-Jarvinen H. Antihypertensive therapy with enalapril improves glucose storage and insulin sensitivity in hypertensive patients with non-insulin-dependent diabetes mellitus. Metabolism 1995 Jan; 44(1): 85–9
Ferri C, Laurenti O, Bellini C, et al. Circulating endothelin-1 levels in lean non-insulin-dependent diabetic patients: influence of ACE inhibition. Am J Hypertens 1995 Jan; 8(1): 40–7
Falkner B, Canessa M, Anzalone D. Effect of angiotensin converting enzyme inhibitor (lisinopril) on insulin sensitivity and sodium transport in mild hypertension. Am J Hypertens 1995 May; 8 (5 Pt 1): 454–60
Bohlen L, Bienz R, Doser M, et al. Metabolic neutrality of perindopril: focus on insulin sensitivity in overweight patients with essential hypertension. J Cardiovasc Pharmacol 1996 Jun; 27(6): 770–6
De Mattia G, Ferri C, Laurenti O, et al. Circulating catecholamines and metabolic effects of captopril in NIDDM patients. Diabetes Care 1996 Mar; 19(3): 226–30
Wiggam MI, Hunter SJ, Atkinson AB, et al. Captopril does not improve insulin action in essential hypertension: a double-blind placebo-controlled study. J Hypertens 1998 Nov; 16(11): 1651–7
Petrie JR, Morris AD, Ueda S, et al. Trandolapril does not improve insulin sensitivity in patients with hypertension and type 2 diabetes: a double-blind, placebo-controlled crossover trial. J Clin Endocrinol Metab 2000 May; 85(5): 1882–9
Jamerson KA, Nesbitt SD, Amerena JV, et al. Angiotensin mediates forearm glucose uptake by hemodynamic rather than direct effects. Hypertension 1996; 27(4): 854–8
Jauch KW, Hartl W, Guenther B, et al. Captopril enhances insulin responsiveness of forearm muscle tissue in non insulin-dependent diabetes mellitus. Eur J Clin Invest 1987; 17: 448–54
Ferriere M, Lachkar H, Richard JL, et al. Captopril and insulin sensitivity. Ann Intern Med 1985; 102: 134–5
Hulthen L, Hoekfelt B. The effect of converting-enzyme inhibitor SQ20881on kinins, renin-angiotensin-aldosterone and catecholamines in relation to blood pressure in hypertensive patients. Acta Med Scand 1978; 204: 497–502
Chow L, De Gasparo M, Levens N. Improved glucose metabolism following blockade of angiotensin converting enzyme but not Angiotensin AT1 receptors. Eur J Pharmacol 1995; 282(1–3): 77–86
Chen S, Kashiwabara H, Kosegawa I, et al. Bradykinin may not be involved in improvement of insulin resistance by angiotensin-converting-enzyme inhibitor. Clin Exp Hypertens 1996; 18(5): 625–36
Rosenthal T, Erlich Y, Rosenmann E, et al. Effects of enalapril, losartan and verapamil on blood pressure and glucose metabolism in the Cohen-Rosenthal diabetic hypertensive rats. Hypertension 1997; 29(6): 1260–4
Moan A, Hoieggen A, Nordby G, et al. Effects of losartan on insulin sensitivity in severe hypertension: connection with sympathetic nervous system activity? J Hum Hypertens 1995; 9: S45–50
Laasko M, Karjalainen L, Lempiainen-Kuosa P. Effects of losartan on insulin sensitivity in hypertensive subjects. Hypertension 1996; 28: 392–6
Paolisso G, Tagliamonte MR, Gambardella A, et al. Losartan mediated improvement in insulin action is mainly due to a non-oxidative glucose metabolism and blood flow in insulin-resistant hypertensive patients. J Hum Hypertens 1997; 11(5): 307–12
Fogari R, Zoppi A, Lazzari P, et al. ACE-inhibition but not angiotensin II antagonism reduces plasma fibrinogen and insulin resistance in overweight hypertensive patients. J Cardiovasc Pharmacol 1998; 32(4): 616–20
Fogari R, Zoppi A, Corradi L, et al. Comparative effects of lisinopril and losartan on insulin sensitivity in the treatment of non diabetic hypertensive patients. Br J Clin Pharmacol 1998; 46: 467–71
Trenkwalder P, Dahl K, Lehtovirta M, et al. Antihypertensive treatment with candesartan cilexitil does not affect glucose homeostasis or serum lipid profile in patients with mild hypertension and type II diabetes. Blood Press 1998; 7(3): 170–5
Higashiura K, Ura N, Miyazaki Y, et al. Effects of angiotensin II receptor antagonist, candesartan cilexitil, on insulin resistance and pressor mechanisms in essential hypertension. J Hum Hypertens 1999; 13 Suppl. 1: S71–4
Taegtmeyer H, Passmore JM. Defective energy metabolism of the heart in diabetes. Lancet 1985 Jan; I(8421): 139–41
Petersen S, Russ M, Renauer H, et al. Inverse regulation of the glucose transporter Glut 4 and G-protein Gs mRNA expression in cardiac myocytes from insulin resistant rats. FEBS Lett 1991; 286: 1–5
Garvey WT, Huecksteadt TP, Matthei S, et al. Role of glucose transporters in the cellular insulin resistance of type II non insulin-dependent diabetes mellitus. J Clin Invest 1988; 81: 1528–36
Garvey WT, Hardin D, Juhaszova M, et al. Effects of diabetes on myocardial glucose transport system in rats: implication for diabetic cardiomyopathy. Am J Physiol 1993; 264 (3 Pt 2): H837–44
Paternostro G, Clarke K, Heath J, et al. Decreased Glut4 mRNA content and insulin-sensitive deoxyglucose uptake show insulin resistance in the hypertensive rat heart. Cardiovasc Res 1995; 30: 205–11
Paternostro G, Pagano D, Gnecchi-Ruscone T, et al. Insulin resistance in patients with cardiac hypertrophy. Cardiovasc Res 1999; 42: 246–53
Koretsky AP. Insight into cellular energy metabolism from transgenic mice. Physiol Rev 1995; 75: 667–88
Katz E, Stenbit AE, Hatton K, et al. Cardiac and adipose tissue abnormalities but not diabetes in mice deficient in Glut 4. Nature 1995; 377: 151–5
Abel D, Kaulbach HC, Tiang R, et al. Cardiac hypertrophy with preserved contractile function after selective deletion of Glut 4 from the heart. J Clin Invest 1999; 101: 1703–14
Belke DD, Larsen TS, Gibbs EM, et al. Glucose metabolism in perfused mouse hearts overexpressing human Glut 4 glucose transporter. Am J Physiol Endocrinol Metab 2001; 280: E420–7
Yang HVT, Erdos EG, Levin Y. Adipeptidyl-carboxy-peptidase that converts Angiotensin I and inactivates bradykinin. Biochem Biophys Acta 1970; 214: 374–6
Lindpaitner K, Ganten D. The cardiac renin-angiotensin system: an appraisal of present experimental and clinical evidence. Circ Res 1991; 68: 905–21
Schoelkens BA, Linz W. Bradykinin-mediated metabolic effects in isolated perfused rat heart. Agents Actions Suppl 1992; 38(2): 36–42
Tomiyama H, Kushiro T, Abeta H, et al. Kinins contribute to the improvement of insulin sensitivity during treatment with ACE-inhibitors. Hypertension 1994; 23(4): 450–5
Roesen P, Eckel J, Reinauer H. Influence of bradykinin on glucose uptake and metabolism studied in isolated cardiac myocytes and isolated perfused hearts. Hoppe Seylers Z Physiol Chem 1983; 364(10): 1431–8
Taylor-McCabe KJ, Ersahin C, Simmons WH. Bradykinin metabolism in the isolated perfused heart. J Hypertens 2001; 19(7): 1295–9
Dietze G, Wicklmayr M. Evidence for a participation of the kallikrein-kinin system in the regulation of muscle metabolism during muscular work. FEBS Lett 1977; 74: 205–8
Henriksen EJ, Jacob S, Augustin HJ, et al. Glucose transport activity in insulin resistant rat muscle: effect of ACE-inhibitors and bradykinin antagonism. Diabetes 1996; 45 Suppl. 1: S125–8
Erdos EG, Marcic BM. Kinins, receptors, kininases and inhibitors: where did they lead us? J Biol Chem 2001; 382(1): 43–7
Rett K, Wicklmayr M, Dietze GJ, et al. Insulin-induced glucose transporters (Glut 1 and Glut 4) translocation in cardiac muscle tissue is mimicked by bradykinin. Diabetes 1996; 45 Suppl. l:S66–9
Rabito SF, Minshall RD, Nakamura F, et al. Bradykinin B2 receptors on skeletal muscle are copled to inositol 1,4,5 trisphosphate formation. Diabetes 1996; 45 Suppl. 1: S29–33
Rett K, Maerker E, Renn W, et al. Perfusion-independent effect of bradykinin and fosinoprilat on glucose transport in Langendorff rat heart. Am J Cardiol 1997; 80(3A): 143A–7A
Henriksen EJ, Jacob S, Tyson R, et al. ACE-inhibition and glucose transport in insulin-resistant muscle: roles of bradykinin and nitric oxide. Am J Physiol 1999; 277(1): R332–6
Carvalho CR, Throne AC, Jose AR, et al. Effect of captopril, losartan and bradykinin on early steps of insulin action. Diabetes 1997; 46: 1950–7
Arbin V, Claperon N, Fournie-Zaluski MC, et al. Acute effect of the dual angiotensin converting enzyme and neutral endopeptidase 24-11 inhibitor Mixanpril on insulin sensitivity in obese Zucker rats. Br J Pharmacol 2001; 133(4): 495–502
Tontonoz P, Hu E, Spiegelman BM, et al. Stimulation of adipogenesis in fibroblasts by PPAR-γ2, a lipid-activated transcription factor. Cell 1994; 79: 1147–56
Schoonjans K, Martin G, Staels B, et al. Peroxisome proliferator-activated receptors, orphans with ligands and functions. Curr Opin Lipidol 1997; 8: 159–166
Staels B, Koening W, Habib A, et al. Activation of human aortic smooth muscle cells is inhibited by PPAR-α but not by PPAR-γ activators. Nature 1998; 393: 790–3
Young PW, Cawthorne MA, Coyle PJ, et al. Repeated treatment of obese mice with BRL 49653, a new potent insulin sensitizer, enhances insulin action in white adipocytes: association with increased insulin binding and cell-surface Glut 4 as measured by photoaffmity labelling. Diabetes 1995; 44(9): 1087–92
Auwerx J, Schoonjans K, Fruchart JC, et al. Regulation of triglyceride metabolism by PPARs: fibrates and thiazolid-inediones. J Atheroscler Thromb 1996; 3(2): 81–9
Ding ST, Schinckel AP, Weber TE, et al. Expression of porcine transcription factors and genes related to fatty acid metabolism in different tissues and genetic populations. J Anim Sci 2000; 78(8): 2127–34
Goetze S, Xi XP, Graf K, et al. Troglitazone inhibits angiotensin II-induced extracellular signal-regulated kinases 1/2 nuclear translocation and activation in vascular smooth muscle cells. FEBS Lett 1999; 452: 277–82
Hattori Y, Akimoto K, Kasai K. The effects of thiazolid-inediones on vascular smooth muscle cell activation by angiotensin II. Biochem Biophys Res Commun 2000; 273(3): 1144–9
Plosker GL, Faulds D. Troglitazone: a review of its use in the management of type 2 diabetes mellitus. Drugs 1999; 57(3): 409–38
Barman Balfour JA, Plosker GL. Rosiglitazone. Drugs 1999; 57(6): 921–30
Mooradian AD, Wolffenbuttel BHR. Rosiglitazone: viewpoints. Drugs 1999; 57(6): 931–2
Gillies PS, Dunn CJ. Pioglitazone. Drugs 2000; 60(2): 333–43
Sugawara A, Takeuchi X, Uruno A, et al. Differential effects among thiazolidinediones on the transcription of tromboxane receptor and angiotensin II type 1 receptor genes. Hypertens Res 2001; 24: 229–33
Ogihara T, Rakugi H, Ikegami H, et al. Enhancement of insulin sensitivity by troglitazone lowers blood pressure in diabetic hypertensives. Am J Hypertens 1995; 8: 316–20
Ghazzi MN, Perez JE, Antonucci TK, et al. Cardiac and glycemic benefits of troglitazone treatment in NIDDM. The Troglitazone Study Group. Diabetes 1997; 46(3): 433–9
Cominacini L, Garbin U, Fratta Pasini A, et al. Troglitazone reduces LDL oxidation and lowers plasma E-selectin concentration in NIDDM patients. Diabetes 1998; 47: 130–3
Berk BC, Corson MA. Angiotensin II signal transduction in vascular smooth muscle: role of tyrosine kinases. Circ Res 1997; 80: 607–16
Paradis P, Dalf-Youcef N, Paradis FN, et al. Overexpression of Angiotensin II type 1 receptor in cardiomyocytes induces cardiac hypertrophy and remodelling. Proc Natl Acad Sci U S A 2000; 97:931–6
Shinohara E, Kihara S, Ouchi N, et al. Troglitazone suppresses intimai formation following balloon injury in insulin-resistant Zucker fatty rats. Atherosclerosis 1998; 136: 275–9
Takeda K, Ichiki T, Tokunov T, et al. Peroxisome proliferator-activated receptor-γ activators downregulate Angiotensin II type 1 receptor in vascular smooth muscle cells. Circulation 2000; 102: 1834–9
Sugawara A, Takeuchi X, Uruno A, et al. Transcriptional suppression of type 1 Angiotensin II receptor gene expression by PPAR-γ in vascular smooth muscle cells. Endocrinology 2001; 142: 3125–34
Benbow A, Steward M, Yeoman G. Thiazolidinediones for type 2 diabetes: all glitazones may exacerbate heart failure [letter]. BMJ 2001; 322(7280): 326
Thomas Ml, Lloyd SJ. Pulmonary edema associated with rosiglitazone and troglitazone. Ann Pharmacother 2001; 35(1): 123–4
Gnudi L, Jensen DR, Tozzo E, et al. Adipose-specific over-expression of Glut 4 in transgenic mice alters lipoprotein-lipase activity. Am J Physiol 1996; 270: R785–92
Broznick JT, McCoid SC, Reynolds TH, et al. Regulation of cell surface Glut 4 in skeletal muscle of transgenic mice. Biochem J 1997; 321: 75–81
Treadway JL, Hargrove DM, Nardone NA, et al. Enhanced peripheral glucose utilisation in transgenic mice overexpressing the human Glut 4 gene. J Biol Chem 1994; 269: 29956–61
Lin Z, Weinberg JM, Malhotra R, et al. Glut 1 reduces hypoxia-induced apoptosis and JNK pathway activation. Am J Physiol Endocrinol Metab 2000; 278: E958–66
McCormack JG, Barr RL, Wolff AA. Ranolazine stimulates glucose oxidation in normoxic, ischemic and reperfused ischemic rat hearts. Circulation 1996; 93: 135–42
Rupp H, Elimban V, Dhalla NS. Modification of subcellular organelles in pressure-overload heart by etomoxir, a carnitine-palmitoyltransferase I inhibitor. FASEB J 1992; 6: 2349–53
Reaves PY, Wang HW, Katovch MJ, et al. Attenuation of hypertension by systemic delivery of retroviral vector containing type 1 angiotensin II receptor antisense cDNA. Methods 2000; 22: 211–8
Natali A, Buzzigoli G, Taddei S, et al. Effects of insulin on hemodynamics and metabolism in human forearm. Diabetes 1990; 39: 490–500
Amiri F, Venema VJ, Wang X, et al. Hyperglycemia enhances Angiotensin II-induced Janus-activated Kinase/STAT signaling in vascular smooth muscle cells. J Biol Chem 1999; 274(45): 32382–6
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bernobich, E., de Angelis, L., Lerin, C. et al. The Role of the Angiotensin System in Cardiac Glucose Homeostasis. Drugs 62, 1295–1314 (2002). https://doi.org/10.2165/00003495-200262090-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-200262090-00002