Summary
Meningococcal disease causes great emotion and anxiety in the families and caregivers of patients. Numbers of such patients are usually small in industrialised countries, unlike those in many regions — especially in subsahelian Africa. Vaccines have been tried for more than 80 years; at present there are available polysaccharide vaccines against groups A, C, Y and W135, and a protein-based vaccine against group B. A property common to all is their relative efficacy (75 to 100%) at school age and after, and an acceptably short persistence of antibodies. Small children pose the major challenge, in whom there is essentially evidence of clinical protection only against group A and C diseases. With vaccines against other serogroups protection is possible, but not yet proven in controlled clinical studies. The search is on for help from various modifications, including the conjugation technique, to transform the independent nature of polysaccharide response towards T cell dependence, as was done earlier in Haemophilus influezae type b vaccines. First trials along this path are encouraging although, again, group B meningococci pose special problems. The next few years will probably see a new generation of meningococcal vaccines.
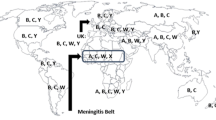
Generally speaking, the incidence of meningococcal disease is too low to indicate vaccinations for the whole population, or even children, but some risk groups and epidemics are important exceptions. To date, bivalent group A + C or tetravalent group A + C + Y + W135 polysaccharides, or an outer membrane protein-based group B vaccine, are the products to be used when the indications, that may vary from country to country, are considered met. A strong herd immunity effect, demonstrated with group A and C vaccinations, facilitates extinction of an epidemic since large-scale vaccinations can be restricted only in the major risk groups, children and in various schools. Prompt intervention demands, however, a functioning mechanism which detects very early on a pending epidemic.Unfortunately, such a mechanism is often lacking in countries often hit by this deadly disease.
Article PDF
Similar content being viewed by others
References
Hume SE. Mass voluntary immunization campaigns for menin-gococcal disease in Canada: media hysteria. JAMA 1992; 267: 1833–8
Gemmill I. An outbreak of meningococcal disease in Ottawa-Carleton December 1991-February 1992. Can J Publ Health 1992; 135: 134–7
Jackson LA, Schuchat A, Reeves MW, et al. Serogroup C meningococcal outbreaks in the United States: an emerging threat. JAMA 1995; 273: 383–9
WHO, Foundation Marcel Mérieux. Control of epidemic meningococcal disease: WHO practical guidelines. Lyon: Editions Fondation Marcel Mérieux, 1995
Riedo FX, Plikaytis BD, Broome CV. Epidemiology and prevention of meningococcal disease. Pediatr Infect Dis J 1995; 14: 643–57
Meningitis in the WHO African region: update, January-April 1997 [editorial]. Wkly Epidemiol Rec 1997; 72: 131
Peltola H. Meningococcal disease — still with us. Rev Infect Dis 1982; 5: 71–91
Peltola H, Kataja MA, Mäkelä PH. Shift in the age-distribution of meningococcal diseases as predictor of an epidemic? Lancet 1982; II: 595–7
Artenstein MS, Gold R, Zimmerly JG, et al. Prevention of meningococcal meningitis by group C polysaccharide vaccine. N Engl J Med 1970; 282: 417–20
Salmi I, Pettay O, Simula I. An epidemic due to sulphonamide-resistant group A meningococci in the Helsinki area (Finland): epidemiological and clinical observations. Scand J Infect Dis 1976; 8: 249–54
Novelli VM, Lewis RG, Dawood ST. Epidemic group A meningococcal disease in Hajj pilgrims [letter]. Lancet 1987; II: 863
Spiegel A, Greindl Y, Lippevald T, et al. Effet de deux stratégies de vaccination sur l’évolution de l’épidémie de méningite à méningocoque a survenue à N’Djamena (Tchad) en 1988. Bull WHO 1993; 71: 311–5
Peltola H. Early meningococcal disease: advising the public and the profession. Lancet 1993; 342: 509–10
Whalen CM, Hockin JC, Ryan A, et al. The changing epidemiology of invasive meningococcal disease in Canada, 1985–1992: emergence of a virulent clone of Neisseria meningitidis. JAMA 1995; 273: 390–4
Flexner S. The results of serum treatment in 1300 cases of epidemic meningitis. J Exp Med 1913; 17: 553–76
Goldschneider I, Gotschlich EC, Artenstein MS. Human immunity to the meningococcus: I. The role of humoral antibodies. J Exp Med 1969; 129: 1307–26
Gold R, Goldschneider I, Lepow ML, et al. Carriage of Neisseria meningitidis and Neisseria lactamica in infants and children. J Infect Dis 1978; 137: 112–21
Mäkelä PH, Peltola H, Käyhty H, et al. Polysaccharide vaccines of group A Neisseria meningitidis and Haemophilus influenzae type b: a field trial in Finland. J Infect Dis 1977; 136 Suppl.: S43–S50
Peltola H, Käyhty H, Kuronen T, et al. Meningococcus group A vaccine in children three months to five years of age. J Pediatrics 1978; 92: 818–22
Sivonen A. Effect of Neisseria meningitidis group A polysaccharide vaccine on nasopharyngeal carrier rates. J Infect 1981; 3: 266–72
Goldschneider I, Gotschlich EC, Artenstein MS. Human immunity to the meningococcus: II. The development of natural immunity. J Exp Med 1969; 129: 1327–48
Wyle FA, Artenstein MS, Brandt BL, et al. Immunologic response of man to group B meningococcal vaccines. J Infect Dis 1972; 126: 514–22
Gotschlich EC, Rey M, Triau R, et al. Quantitative determination of the human immune response to immunization with meningococcal vaccines. J Clin Invest 1972; 51: 89–96
Finne J, Leinonen M, Mäkelä PJ. Antigenic similarities between brain components and bacteria causing meningitis implications for vaccine development and pathogenesis. Lancet 1983; II: 355–7
Lifely MR, Moreno C. Vaccine against meningococcal group B disease. Lancet 1986; I: 214–5
Mandrell RE, Azmi FH, Granoff DM. Complement-mediated bactericidal activity of human antibodies to poly α2→8 N-acetylneuraminic acid, the capsular polysaccharide of Neisseriameningitidis serogroup B. J Infect Dis 1995; 172: 1279–89
Triau R. La méningite cérébro-spinale: son traitement et sa prévention modernes. Séminaires intenational sur les vaccinations en Afrique. Lyon, France: Editions Fondation Mérieux, 1974: 135
Lapeyssonnie L. La méningite cérébro-spinale en Afrique. Bull WHO 1963: 28 Suppl.: 1–114
Delville J. Sérothérapie de la méningite cérébrospinale. Arch Belges Med Soc 1973; 6-7: 424
Riding D, Corkill NL. Prophylactic vaccination in epidemic meningococcal meningitis. J Hyg 1932; 32: 258
Saleun G, Ceccaldi J. Étude des méningocoques en Afrique Équatoriale Française et vaccination antiméningococcique. Bull Soc Path Exot 1936; 29: 996–1006
Greenberg L, Cooper MY. A somatic antigen vaccine for the prevention of meningococcal cerebrospinal meningitis. Bull WHO 1965; 33: 21–6
Heidelberger M, MacLeod CM, Lapi MM. The human antibody response to simultaneous injection of six specific polysaccharides of pneumococci. J Exp Med 1948; 88: 369–72
MacLeod CM, Hodges RG, Heidelberger M, et al. Prevention of pneumococcal pneumonia by immunization with specific capsular polysaccharides. J Exp Med 1945; 82: 445–65
Kabat EA, Kaiser H, Sikorski H. Preparation of the type specific polysaccharide of the type I meningococcus and a study of its effectiveness as an antigen in human beings. J Exp Med 1945; 80: 229–307
Liu T-Y, Gotschlich EC, Jonseen EK, et al. Studies on the meningococcal polysaccharides: I. Composition and chemical properties of the group A polysaccharide. J Biol Chem 1971; 246: 2849–58
Gotschlich EC, Liu T-Y, Artenstein MS. Human immunity to the meningococcus: III. Preparation and biochemical properties of the group A, group B and group C meningococcal polysaccharide. J Exp Med 1969; 129: 1349–65
Sanborn WR, Bencic Z, Cvjetanovic B, et al. Trial of a serogroup A meningococcus polysaccharide vaccine in Nigeria. Progr Immunobiol Stand 1972; 5: 497–505
Greenwood BM, Hassan-King M, Whittle HC. Prevention of secondary cases of meningococcal disease in household contacts by vaccination. BMJ 1978; 1: 1317–9
Gold R, Lepow ML, Goldschneider I, et al. Clinical evaluation of group A and group C meningococcal polysaccharide vaccines in infants. J Clin Invest 1975; 56: 1536–47
Farquhar JD, Hankins WA, Desanctis AN. Clinical and serological evaluation of a meningococcal polysaccharides vaccine groups A, C, and Y. Proc Soc Exp Biol Med 1978; 157: 79–82
Steinhoff MC, Lewin EB, Gotschlich EC, et al. Group C Neisseria meningitidis variant polysaccharide vaccines in children. Infect Immun 1981; 34: 144–6
McLeod Griffiss J, Brandt BL, Allied PL, et al. Safety and immunogenicity of group Y and group W135 meningococcal capsular polysaccharide vaccines in adults. Infect Immun 1981; 34: 725–32
Hankins WA, Gwaltney JM, Hendly JO, et al. Clinical and serological evaluation of a meningococcal polysaccharide vaccine groups A, C, Y, and W135 (41306). Proc Soc Exp Biol Med 1982; 169: 54–7
Peltola H, Safary A, Käyhty H. Evaluation of two tetravalent (ACYW135) meningococcal vaccines in infants and small children: a clinical study comparing immunogenicity of 0-acetyl-negative and 0-acetyl-positive group C polysaccharides. Pediatrics 1985; 76: 91–6
Cadoz M, Armand J, Arminjon F, et al. Tetravalent (A, C, Y, W) meningococcal vaccine in children: immunogenicity and safety. Vaccine 1985; 3: 340–2
Granoff DM, Gupta RK, Belshe RB, et al. Induction of immunologic tolerance in adults by meningococcal C (MenC) polysaccharide (PS) vaccination [abstract no. 417]. 35th Annual Meeting of the Infectious Diseases Society of America (IDSA); 1997 Sep 13–16: San Francisco
Wahdan MH, Rizk F, el-Akkad AM. A controlled field trial of a serogroup A meningococcal polysaccharide vaccine. Bull WHO 1973; 48: 667–73
Erwa HH, Haseeb MA, Idris AA, et al. A serogroup A meningococcal polysaccharide vaccine. Bull WHO 1973; 49: 301–5
Lepow ML. Meningococcal vaccines. In: Plotkin SA, Mortimer Jr EA, editors. Vaccines. Philadelphia: WB Saunders, 1994: 503–15
Mäkelä PH, Käyhty H, Weckström P, et al. Effect of group A meningococcal vaccine in army recruits in Finland. Lancet 1975; II: 883–6
Barnett ED, Breña AE, McNamara ER, et al. Response to quadrivalent meningococcal vaccine in children less than 2 years of age [abstract no. 983]. Pediatr Res 1996; 39 (Pt 2): 166
Artenstein MS, Brandt BL. Immunologic hyporesponsiveness in man to group C meningococcal polysaccharide. J Immunol 1975; 115: 5–7
Gold R, Lepow ML, Goldschneider I, et al. Immune response of human infants to polysaccharide vaccines of groups A and C Neisseria meningitidis. J Infect Dis 1977; 136: S31–5
McCormick JB, Gusmao HH, Nakamura S, et al. Antibody response to serogroup A and C meningococcal vaccines in infants born to mothers vaccinated during pregnancy. J Clin Invest 1980; 65: 1141–4
MacDonald N, Halperin S, Law B, et al. Immunization of toddlers with Chiron® conjugated meningococcal C (MenC) vaccine induces immunologic memory while plain Men polysaccharide (PS) vaccine induces tolerance [abstract no. G-3]. 37th Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC); 1997 Sep 28–Oct 1: Toronto
Peltola H, Mäkelä PH, Käyhty H, et al. Clinical efficacy of meningococcus group A capsular polysaccharide vaccine in children three months to five years of age. N Engl J Med 1977; 297: 686–91
Gotschlich ED. Development of polysaccharide vaccines for the prevention of meningococcal diseases. Monogr Allergy 1975; 9: 245–58
Gotschlich EC, Goldschneider I, Artenstein MS. Human immunity to the meningococcus: IV Immunogenicity of group A and group C meningococcal polysaccharides in human volunteers. J Exp Med 1969; 129: 1367–84
Käyhty H, Karanko V, Peltola H, et al. Serum antibodies to capsular polysaccharide vaccine of group A Neisseria meningitidis followed for three years in infants and children. J Infect Dis 1980; 142: 861–8
el Bushra HE, Mawlawi MY, Fontaine RE, et al. Meningococcal meningitis group A: a successful control of an outbreak by mass vaccination. E Afr Med J 1995; 72: 715–8
Reingol AL, Broome CV, Hightower AW, et al. Age-specific differences in duration of clinical protection after vaccinations with meningococcal polysaccharide A vaccine. Lancet 1985; II: 114–8
Ceesay SJ, Allen SJ, Menon A, et al. Decline in meningococcal antibody levels in African children 5 years after vaccination and the lack of an effect of booster immunization. J Infect Dis 1993; 167: 1212–6
Greenwood AM, Greenwood BM, Bradley AK, et al. Enhancement of the immune response to meningococcal polysaccharide vaccine in a malaria endemic area by administration of chloroquine. Ann Trop Med Parasitol 1981; 75: 261–3
Williamson AW, Greenwood BM. Impairment of the immune response to vaccination after malaria. Lancet 1978; I: 1328–9
Lepow M, Goldschneider I, Gold R, et al. Persistence of antibody following immunization of children with groups A and C meningococcal polysaccharide vaccines. Pediatrics 1977; 60: 673–80
King J, MacDonald N, Huang J, et al. 3 year follow-up and booster response to quadrivalent meningococcal polysaccharide vaccine (QMPV) [abstract no. 622]. Pediatr Res 1996; 39 (Pt 2): 106
King WJ, MacDonald N, Wells G, et al. Total and functional antibody response to a quadrivalent meningococcal polysaccharide vaccine among children. J Pediatr 1996; 128: 196–202
Brandt BL, Artenstein MS. Duration of antibody responses after vaccination with group C Neisseria meningitidis polysaccharide. J Infect Dis 1975; 131: 569–75
Mohammed I, Onyemelukwe GC, Obineche EN, et al. Control of epidemic meningococcal meningitis by mass vaccination: II. Persistence of antibody four years after vaccination. J Infect 1984; 9: 197–202
Beuvery EC, Leussink AB, van Delft RW, et al. Immunoglobulin M and G antibody responses and persistence of these antibodies in adults after vaccination with a combined meningococcal group A and group C polysaccharide vaccine. Infect Immun 1982; 37: 579–85
Zangwill KM, Stout RW, Carlone GM, et al. Duration of antibody response after meningococcal polysaccharide vaccination in US Air Force personnel. J Infect Dis 1994; 169: 847–52
Scheifele DW, Bjornson G, Boraston S. Local adverse effects of meningococcal vaccine. Can Med Assoc J 1994; 150: 1033–6
Jamba G, Bytchenko B, Causse G, et al. Immunization during a cerebrospinal meningitis epidemic in the Mongolian People’s Republic 1974–1975. Bull WHO 1979; 57: 943–6
Mohammed I, Zaruba K. Control of epidemic meningococcal meningitis by mass vaccination. Lancet 1981; II: 80–3
Greenwood BM, Wali SS. Control of meningococcal infection in the African meningitis belt by selective vaccination. Lancet 1980; I: 729–32
Binkin N, Band J. Epidemic of meningococcal meningitis in Bamako, Mali: epidemiological features and analysis of vaccine efficacy. Lancet 1982; II: 315–8
Greenwood BM, Smith AW, Hassan-King M, et al. The efficacy of meningococcal polysaccharide vaccine in preventing group A meningococcal disase in the Gambia, West Africa. Trans R Soc Trop Med Hyg 1986; 80: 1006–7
Cochi SL, Markowitz LE, Joschi DD, et al. Control of epidemic group A meningococcal meningitis in Nepal. Int J Epidemiol 1987; 16: 91–7
Lennon D, Gellin B, Hood D, et al. Successful intervention in a group A meningococcal outbreak in Auckland, New Zealand. Pediatr Infect Dis J 1992; 11: 617–23
Al-Gahtani YM, El Bushra HE, Al-Qarawi SM, et al. Epidemiological investigation of an outbreak of meningococcal meningitis in Makkah (Mecca), Saudi Arabia, 1992. Epidemiol Infect 1995; 115: 399–409
Whitney CG, Dondoc N, Enktuja B, et al. Control of epidemic serogroup A meningococcal disease, Mongolia [poster 20.011]. 7th International Congress for Infectious Diseases; 1996 Jun 10–13: Hong Kong
Peltola H. Group A meningococcal polysaccharide vaccine and course of the A meningococcal epidemic in Finland. Scand J Infect Dis 1978; 10: 41–4
Haelterman E, Boelaert M, Suetens C, et al. Impact of a mass vaccination campaign against a meningitis epidemic in a refugee camp. Trop Med Int Health 1996; 1: 385–92
Gold R, Artenstein MS. Meningococcal infections: 2. Field trial of group C meningococcal polysaccharide vaccine in 1969–1970. Bull WHO 1971; 45: 279–82
Stroffolini T. Vaccination campaign against meningococcal disease in army recruits in Italy. Epidemiol Infect 1990; 105: 579–83
Masterton RG, Youngs ER, Wardle JC, et al. Control of an outbreak of group C meningococcal meningitis with a polysaccharide vaccine. J Infect Dis 1988; 17: 177–82
Feigin RD, Baker CJ, Herwaldt LA, et al. Epidemic meningococcal disease in an elementary-school classroom. N Engl J Med 1982; 307: 1255–7
Rønne T, Berthelsen L, Buhl LH, et al. Comparative studies on pharyngeal carriage of Neisseria meningitidis during a localized outbreak of serogroup C meningococcal disease. Scand J Infect Dis 1993; 25: 331–9
Rosenstein N, Levine O, Taylor J, et al. Persistent serogroup C meningococcal disease outbreak [abstract no. G84]. 36th Interscience Conference on Antimicrobial Agents and Chemotherapy; 1996 Sep 15–18: New Orleans
Taunay AE, Feldman RA, Bastos CO, et al. Avaliaçao do efeito protetor de vacina polissacaridica antimeningocócica do grupo C, em crianças de 6 a 36 meses. Rev Inst Adolfo Lutz 1978; 38: 77–82
Biselli R, Fattorossi A, Matricardi PM, et al. Dramatic reduction of meningococcal meningitis among military recruits in Italy after introduction of specific vaccination. Vaccine 1993; 11: 578–81
Pearce MC, Sheridan JW, Jones DM, et al. Control of group C meningococcal disease in Australian Aboriginal children by mass rifampicin chemoprophylaxis and vaccination. Lancet 1995; 346: 20–3
De Wals P, Dionne M, Douville-Fradet M, et al. Impact of a mass immunization campaign against serogroup C meningococcus in the province of Quebec, Canada. Bull WHO 1996; 74: 407–11
Kriz P, Vlckova J, Bobak M. Targeted vaccination with meningococcal polysaccharide vaccine in one district of the Czech Republic. Epidemiol Infect 1995; 115: 411–8
Milagres LG, Lemos APS, Meles CEAA, et al. Antibody response after immunization of Brazilian children with serogroup C meningococcal polysaccharide noncovalently complexed with outer membrane proteins. Braz J Med Biol Res 1995; 28: 981–9
Sáez-Nieto JA, Perucha M, Casamayor H, et al. Outbreak of infection caused by Neisseria meningitidis group C type 2 in nursery. J Infection 1984; 8: 49–55
Bjune G, Højby EA, Grønnesby JK, et al. Effect of outer membrane vesicle vaccine against group B meningococcal disease in Norway. Lancet 1991; 338: 1093–6
Sierra GVG, Campa HC, Varcacel NM, et al. Vaccine against group B Neisseria meningitidis: protection trial and mass vaccination results in Cuba. NIPH Annals 1991; 14: 195–210
de Moraes JC, Perkins BA, Camargo MCC, et al. Protective efficacy of a serogroup B meningococcal vaccine in Sao Paulo, Brazil. Lancet 1992; 340: 1074–8
Costa E de A. On the controversy about the efficacy of the antimeningococcal B vaccine: methodological pitfalls. Cadernos de Saúde Pública (Rio de Janeiro) 1995; 11: 332–5
Noronha CP, Struchiner CJ, Halloran ME. Assessment of the direct effectiveness of BC meningococcal vaccine in Rio de Janeiro, Brazil: a case-control study. Int J Epidemiol 1995; 24: 1050–7
Costa E de A, Martins H, Klein CH. Avaliação da proteção conferidda pela vacina antimenigocócica BC no Estado de Santa Catarina, Brazil, 1990/ 92. Rev Saúde Pública 1996; 30: 460–70
Frasch C, Coetzee G, Zahradnik JM, et al. Development and evaluation of group B serotype 2 protein vaccines: report of a group B field trial. Med Trop 1983; 43: 177–80
Zollinger WD, Boslego J, Moran E, et al. Meningococcal serogroup B vaccine protection trial and follow-up studies in Chile. NPHI Annals 1991; 14: 211–2
Boslego J, Garcia J, Cruz C, et al. Efficacy, safety, and immunogenicity of a meningococcal group B (15: P1.3) outer membrane protein vaccine in Iquique, Chile. Vaccine 1995; 13: 821–9
Fine PEM. Meningococcal vaccine trial in Norway. Lancet 1991; 338: 1456–7
Sierra GVG, Campa HC, Garcia IL, et al. Efficacy evaluation of the Cuban vaccine VA-MENGOC-BC against disease caused by serogroup B Neisseria meningitidis. In: Achtman M, Marchai C, Morelli G, et al., editors. Neisseriae 1990. Berlin: Walter de Gruyter, 1991: 129–34
Nishioka S de A. Effectiveness of BC meningococcal vaccine in Brazil. Int J Epidemiol 1996; 25: 1102–3
Costa E de A. Effectiveness of meningococcal vaccine in Brazil. Int J Epidemiol 1997; 26: 681–4
Cordeiro OR, Colls CP, Fernandez AA. Eficacia poslicen-ciamiento de VA-MENGOC-BC en menores de 6 años en Holguín, Cuba: primer año de observación. Rev Cubana Med Trop 1995; 47: 59–64
Romero JD, Outschoorn IM. Current status of meningococcal group B vaccine candidates: capsular or noncapsular? Clin Microb Rev 1994; 7: 559–75
Perkins B, Jonsdottir K, Briem H, et al. Immunogenicity of two outer membrane protein-based serogroup B meningococcal vaccines among young adults in Iceland [abstract no. G81]. 34th Interscience Conference on Antimicrobial Agents and Chemotherapy; 1994 Oct 4–7: Orlando (FL)
Carlone GM, Williams D, Dykes J, et al. Comparison of serum bactericidal results using vaccine type-strains and heterologous target strains to evaluate immunogenicity of two meningococcal serogroup B vaccines in Iceland [poster 178]. 9th International Pathogenic Neisseria Conference; 1994 Sep 26–30: Winchester
Milagres LG, Ramos SR, Sacchi CT, et al. Immune response of Brazilian children to Neisseria meningitidis serogroup B outer membrane protein vaccine: comparison with efficacy. Infect Immun 1994; 62: 4419–24
Rosenqvist E, Høiby EA, Wedege E, et al. Human antibody responses to meningococcal outer membrane antigens after three doses of Norwegian group B meningococcal vaccine. Infect Immun 1995; 63: 4642–52
Debbag R, Rüttimann R, Stamboulian D. Evaluation of adverse reactions associated to antimeningococcal BC vaccination in 16,700 children [abstract no. 420]. 33rd Annual Meeting of the Infectious Disease Society of America; 1995 Oct 16–17: San Francisco
Ala’aldeen DA, Stevenson P, Griffiths E, et al. Immune response in humans and animals to meningococcal transferrin-binding proteins: implications for vaccine design. Infect Immun 1993; 62: 2984–90
van der Ley P, Poolman JT. Construction of a multivalent meningococcal vaccine strain based on the class 1 outer membrane protein. Infect Immun 1992; 60: 3156–61
Peeters CCAM, Riimke HC, Sundermann LC, et al. Phase I clinical trial with hexavalent PorA containing meningococcal outer membrane vesicle vaccine. Vaccine 1996; 14: 1009–15
Muttilainen S, Idänpäänn-Heikkilä I, Whalström E, et al. The Neisseria meningitidis outer membrane protein PI produced in Bacillus subtilis and reconstituted into phospholipid vesicles elicits antibodies to native P1 epitopes. Microb Pathogen 1995; 18: 423–36
Idänpään-Heikkilä I, Wahlström W, Muttilainen S, et al. Immunization with meningococcal class 1 outer membrane protein produced in Bacillus subtilis and reconstituted in the presence of Zwittergent or Triton X-100. Vaccine 1996; 14: 886–91
Peltola H, Kilpi T, Anttila M. Rapid disappearance of Haemophilus influenzae type b meningitis after routine childhood immunisation with conjugate vaccines. Lancet 1992; 340: 592–4
Jennings HJ, Lugosski C. Immunochemistry of groups A, B, and C meningococcal polysaccharide-tetanus toxoid conjugates. J Immunol 1981; 127: 1011–8
Devi SJN, Robbins JB, Schneerson R. Antibodies to poly[2-8)-a-N-acetylneuraminic acid] and poly[2-9)-a-N-acetylneuraminic acid] are elicited by immunization of mice with Escherichia coli K92 conjugates: potential vaccines for groups B and C meningococci and E. coli Kl. Proc Natl Acad Sci USA 1991; 88: 7175–9
Granoff DM, Forrest B, Rappuoli R. Meningococcal polysaccharide-protein conjugate vaccines. Int J Infect Dis 1997; 1: 152–7
Costantino P, Viti S, Podda A, et al. Development and phase 1 clinical testing of a conjugate vaccine against meningococcus A and C. Vaccine 1992; 10: 691–8
Anderson EL, Bowers T, Mink CM, et al. Safety and immunogenicity of meningococcal A and C polysaccharide conjugate vaccine in adults. Infect Immun 1994; 62: 3391–5
Fairley CK, Begg N, Borrow R, et al. Conjugate meningococcal serogroup A and C vaccine: reactogenicity and immunogenicity in United Kingdom infants. J Infect Dis 1996; 174: 1360–3
Twumasi Jr PA, Kumah S, Leach A, et al. A trial of a group A plus group C meningococcal polysaccharide-protein conjugate vaccine in African infants. J Infect Dis 1995; 171: 632–8
Shackley FM, Heath PT, Flamank C, et al. Immunogenicity and reactogenicity of a group C meningococcal conjugate vaccine in British children [abstract no. 284]. Clin Infect Dis 1992; 23: 912
Maclennan J, Shackley F, Heath P, et al. Induction of immunological memory by a Neisseria meningitidis group C conjugate vaccine [abstract no. 19]. 15th Annual Meeting of the European Society for Paediatric Infectious Diseases; 1997 May 21–23: Paris
Leach A, Twumasi PA, Kumah S, et al. Induction of immunologic memory in Gambian children by vaccination in infancy with a group A plus group C meningococcal polysaccharide-protein conjugate vaccine. J Infect Dis 1997; 175: 200–4
Campagne G, Garba A, Fabre P, et al. Safety and immunogenicity of three doses of a N. meningitidis A/C diphtheria conjugate vaccine in infants in Niger [abstract no. G-1]. 37th Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC); 1997 Sep 28–Oct 1: Toronto
Bartoloni A, Norelli F, Ceccarini C, et al. Immunogenicity of meningococcal B polysaccharide conjugated to tetanus toxoid or CRM197 via adipic acid dihydrazide. Vaccine 1995; 13: 463–70
Fusco PC, Michon F, Tai JY, et al. Preclinical evaluation of a novel group B meningococcal conjugate vaccine that elicits bactericidal activity in both mice and nonhuman primates. J Infect Dis 1997; 175: 364–72
Tai JY, Michon PC, Fusco PC. Combination conjugate vaccine against Neisseria meningitidis serogroups A, B and C [poster 123.008]. 7th International Congress for Infectious Diseases; 1996 Jun 10–13: Hong Kong
Gray C. Meningococcal disease: was Ottawa’s mass-vaccination program necessary? Can Med Assoc J 1992; 146: 1033–7
Jackson LA, Schuchat A, Gorsky RD, et al. Should college students be vaccinated against meningococcal disease? A cost-benefit analysis. Am J Public Health 1995; 85: 843–5
Varaine F, Caugan DA, Riou JY, et al. Meningitis outbreaks and vaccination strategy. Trans R Soc Trop Med Hyg 1997; 91: 3–7
American Academy of Pediatrics. Meningococcal disease prevention and control strategies for practice-based physicians. Pediatrics 1996; 97: 404–11
Recommendations of the Advisory Committee on Immunization Practices (ACIP). Control and prevention of meningococcal disease, and control and prevention of serogroup C meningococcal disease: evaluation and management of suspected outbreaks. MMWR Morb Mortal Wkly Rep 1977; 46(RR-5): 13–21
Torres EM, Alvarez GA, Rodriguez RM. Los cambios en la distribucion segun la edad, como factor de prediccion epidemica en la enfermedad meningococica. Rev Cub Pediatr 1987; 59: 773–83
Jones DM, Mallard RH. Age incidence of meningococcal infection in England and Wales, 1984–1991. J Infect 1993; 27: B3–8
Centers for Disease Control. Serogroup B meningococcal disease — Oregon, 1994. MMWR Morb Mortal Wkly Rep 1995; 44: 121–4
Zangwill KM, Schuchat A, Riedo FX, et al. School-based clusters of meningococcal disease in the United States: descriptive epidemiology and a case-control analysis. JAMA 1997; 227: 389–5
Hubert B, Caugant DA. Evolution récente des infections à méningocoque en Europe. Eurosurveillance 1997; 2: 69–71
Kaiser AB, Hennekens CH, Saslaw MS, et al. Seroepidemiology and chemoprophylaxis of diseases due to sulfonamide-resistant Neisseria meningitidis in a civilian population. J Infect Dis 1974; 130: 217–24
Jacobson JA, Camargos PAM, Ferreira JT, et al. The risk of meningitis among classroom contacts during an epidemic of meningococcal disease. Am J Epidemiol 1976; 104: 552–5
Miller MA, Wenger J, Rosenstien N, et al. Evaluation of meningococcal meningitis control strategies for the Meningitis Belt in Africa [abstract no. K127]. 37th Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC); 1997 Sep 28–Oct 1: Toronto
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Peltola, H. Meningococcal Vaccines. Drugs 55, 347–366 (1998). https://doi.org/10.2165/00003495-199855030-00003
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-199855030-00003