Summary
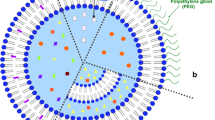
Encapsulation in polyethylene glycol-coated (pegylated; Stealth®) liposomes alters the pharmacokinetic characteristics, and hence the safety and tolerability profile, of doxorubicin.
Pegylated liposomal doxorubicin administered as a single agent is generally well tolerated. Grade III/IV leucopenia, stomatitis and palmar-plantar erythrodysaesthesia affected 16, 6 and 18% of solid tumour patients, respectively. Other adverse effects included nausea and vomiting and alopecia. Acute hypersensitivity infusion reactions have been reported in up to 9% of patients in some studies. Recently published data from a phase II trial in patients with refractory ovarian cancer showed no alopecia or cardiotoxicity and little nausea and vomiting after pegylated liposomal doxorubicin. Unlike free doxorubicin, pegylated liposomal doxorubicin is not a vesicant.
Preliminary data, not yet confirmed in comparative studies, suggest that the pegylated liposomal formulation may be less cardiotoxic than free doxorubicin. Mucositis, however, appears to be increased. Studies to determine optimal dosing schedules and safety of pegylated liposomal doxorubicin in combination with other agents are ongoing.
Similar content being viewed by others
References
Hortobagyi GN. Anthracyclines in the treatment of cancer: an overview. Drugs 1997; 54Suppl. 4: 1–7
Gabizon AA, Martin F. Polyethylene glycol-coated (pegylated) liposomal doxorubicin: rationale for use in solid tumours. Drugs 1997; 54Suppl. 4: 15–21
SEQUUS Pharmaceuticals Inc. Doxil safety report (07 Apr 1997). Menlo Park, California, USA
Dezube BJ. Safety assessment: Doxil® (doxorubicin HCl liposome injection) in refractory AIDS-related Kaposi’s sarcoma. Doxil® Clinical Series Vol. 1, No. 2; SEQUUS Pharmaceuticals, Inc., Menlo Park, California, 1996
Muggia FM, Amantea M, Jeffers S, et al. DOXIL (doxorubicin in stealth liposomes): a drug with favorable therapeutic index in the management of refractory epithelial ovarian cancer [abstract no. 31]. Cancer Invest 1996; 14Suppl. 1: 39–41
Uziely B, Jeffers S, Isacson R, et al. Liposomal doxorubicin: antitumor activity and unique toxicities during two complementary phase I studies. J Clin Oncol 1995; 13(7): 1777–85
Muggia FM, Hainsworth J, Jeffers S, et al. Phase II study of liposomal doxorubicin in refractory ovarian cancer: antitumor activity and toxicity modification by liposomal encapsulation. J Clin Oncol 1997; 15: 987–993
Coukell AJ, Spencer CM. Polyethylene glycol-liposomal doxorubicin: a review of its pharmacodynamic and pharmacokinetic properties, and therapeutic efficacy in the management of AIDS-related Kaposi’s sarcoma. Drugs 1997; 53(3): 520–38
Working PK, Dayan AD. Pharmacological-toxicological expert report: Caelyx™ (Stealth® liposomal doxorubicin HCl). Hum Exp Toxicol 1996; 15: 752–85
Lokich JJ, Moore C. Chemotherapy-associated palmar plantar erythrodysesthesia syndrome. Ann Int Med 1984; 101: 798–800
Ranson M, O’Byrne K, Carmichael J, et al. Phase II dose-finding trial of DOX-SL™ (Stealth® liposomal doxorubicin HCl) in the treatment of advanced breast cancer [abstract no. 161]. Proc Am Soc Clin Oncol 1996; 15: 124
Venook AP, Amantea M, Bonnem E, et al. Pegylated liposome-encapsulated doxorubicin (Doxil®) in patients with hepatocellular carcinoma (HCC) [abstract no. 1494]. Proc Am Soc Clin One 1996; 15: 473
Olver IN, Aisner J, Hament A, et al. A prospective study of topical dimethyl sulfoxide for treating anthracycline extravasation. J Clin Oncol 1988; 6(11): 1732–5
Bertelli G, Gozza A, Forno GB, et al. Topical dimethyl sulfoxide for the prevention of soft tissue injury after extravasation of vesicant cytotoxic drugs: a prospective clinical study. J Clin Oncol 1995; 13: 2851–5
Madhavan S, Northfelt DW. Lack of vesicant injury following extravasation of liposomal doxorubicin [letter]. J Natl Cancer Inst 1995; 87(20): 154–5
Hengge UR, Brockmeyer NH, Rasshofer R, et al. Fatal hepatic failure with liposomal doxorubicin [letter]. Lancet 1993; 341: 383–4
Coker RJ, James ND, Stewart JSW. Hepatic toxicity of liposomal encapsulated doxorubicin [letter]. Lancet 1993; 341: 756
Saltiel E, McGuire W. Doxorubicin (adriamycin) cardiomyopathy: a critical review. West J Med 1983; 139: 332–41
Bielack SS, Erttmann R, Kempf-Bielack B, et al. Impact of scheduling on toxicity and clinical efficacy of doxorubicin: what do we know in the mid-nineties? Eur J Cancer 1996; 32A(10): 1652–60
Madhavan S, Northfelt DW. Lack of vesicant injury following extravasation of liposomal doxorubicin [abstract]. Cancer Res Treat 1996; 37 Suppl.: 77
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Alberts, D.S., Garcia, D.J. Safety Aspects of Pegylated Liposomal Doxorubicin in Patients with Cancer. Drugs 54 (Suppl 4), 30–35 (1997). https://doi.org/10.2165/00003495-199700544-00007
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-199700544-00007