Abstract
Background and objective
The calcimimetic cinacalcet hydrochloride (cinacalcet) is used for treatment of patients with chronic kidney disease with secondary hyperparathyroidism, a population that commonly receives multiple concurrent medications. Cinacalcet is eliminated primarily via oxidative metabolism mediated, in part, through cytochrome P450 (CYP) 3A4. Thus, the potential for an inhibitor of CYP3A4 to alter the pharmacokinetics of cinacalcet is of clinical importance. The objective of this study was to evaluate the pharmacokinetics of cinacalcet during treatment with a potent CYP3A4 inhibitor, ketoconazole.
Subjects and methods
Twenty-four healthy subjects were enrolled in an open-label, crossover, phase I study to receive a single oral dose of cinacalcet (90mg) alone and with 7 days of ketoconazole (200mg twice daily). Blood samples for pharmacokinetics were collected for up to 72 hours postdose. Cinacalcet plasma concentration-time data were analysed by noncompartmental methods. Pharmacokinetic parameters were analysed using a crossover ANOVA model that included subjects who completed both treatment arms.
Results
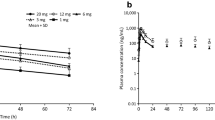
Twenty subjects completed both treatment arms. The mean area under the plasma concentration-time curve of cinacalcet increased 2.3-fold (90% CI 1.92, 2.67) [range 1.15- to 7.12-fold] and the mean maximum plasma concentration increased 2.2-fold (90% CI 1.67, 2.78) [range 0.904- to 10.8-fold] when administered with ketoconazole, relative to when administered alone. The time to reach the maximum plasma concentration was not significantly affected, and the terminal elimination half-lives were similar between treatments.
Conclusions
Co-administration of a potent CYP3A4 inhibitor moderately increased cinacalcet exposure in study subjects. This suggests that clinicians should monitor parathyroid hormone and calcium concentrations when a patient receiving cinacalcet initiates or discontinues therapy with a strong CYP3A4 inhibitor.



Similar content being viewed by others
Notes
The use of trade names is for product identification purposes only and does not imply endorsement.
References
Block GA, Klassen PS, Lazarus JM, et al. Mineral metabolism, mortality, and morbidity in maintenance hemodialysis. J Am Soc Nephrol 2004; 15(8): 2208–18
Moe SM, Chertow GM, Coburn JW, et al. Achieving NKF-K/DOQI™ bone metabolism and disease treatment goals with cinacalcet HCl. Kidney Int 2005; 67(2): 760–71
National Kidney Foundation. K/DOQI clinical practice guidelines for bone metabolism and disease in chronic kidney disease. Am J Kidney Dis 2003; 42(4 Suppl. 3): S1–201
Silverberg SJ, Faiman C, Bilezikian JP, et al. Cinacalcet HCl effectively treats hypercalcemia in patients with parathyroid carcinoma [abstract]. J Bone Miner Res 2004; 19 Suppl. 1: S103
Kumar GN, Sproul C, Poppe L, et al. Metabolism and disposition of calcimimetic agent cinacalcet HCl in humans and animal models. Drug Metab Dispos 2004; 32(12): 1491–500
Data on file, Amgen Inc., 2003
Amgen Inc. Sensipar® prescribing information [online]. Thousand Oaks (CA): Amgen Inc., 2006. Available from URL: http://www.sensipar.com/professional/prescribing_information/prescribing_information.jsp (Accessed 2007 Apr 24)
Dresser GK, Spence JD, Bailey DG. Pharmacokinetic-pharmacodynamic consequences and clinical relevance of cytochrome P450 3A4 inhibition. Clin Pharmacokinet 2000; 38(1): 41–57
Zhou S, Chan E, Lim LY, et al. Therapeutic drugs that behave as mechanism-based inhibitors of cytochrome P450 3A4. Curr Drug Metab 2004; 5(5): 415–42
Bjornsson TD, Callaghan JT, Einolf HJ, et al. The conduct of in vitro and in vivo drug-drug interaction studies: a PhRMA perspective. J Clin Pharmacol 2003; 43(5): 443–69
Center for Drug Evaluation and Research, Food and Drug Administration, US Department of Health and Human Services. Guidance for industry: in vivo drug metabolism/drug interaction studies — study design, data analysis, and recommendations for dosing and labeling [online]. Rockville (MD): Center for Drug Evaluation and Research, 1999. Available from URL: http://www.fda.gov/cder/guidance/ (Accessed 2007 Mar 20)
Daneshmend TK, Warnock DW, Ene MD, et al. Influence of food on the pharmacokinetics of ketoconazole. Antimicrob Agents Chemother 1984; 25(1): 1–3
Rowland M, Tozer TN. Clinical pharmacokinetics: concepts and applications. Media, PA: Williams & Wilkins, 1995: 271–2
Olkkola KT, Backman JT, Neuvonen PJ. Midazolam should be avoided in patients receiving the systemic antimycotics ketoconazole or itraconazole. Clin Pharmacol Ther 1994; 55(5): 481–5
Varhe A, Olkkola KT, Neuvonen PJ. Oral triazolam is potentially hazardous to patients receiving systemic antimycotics ketoconazole or itraconazole. Clin Pharmacol Ther 1994; 56(6 Pt 1): 601–7
Boxenbaum H. Cytochrome P450 3A4 in vivo ketoconazole competitive inhibition: determination of Ki and dangers associated with high clearance drugs in general. J Pharm Pharm Sci 1999; 2(2): 47–52
Yun CH, Okerholm RA, Guengerich FP. Oxidation of the antihistaminic drug terfenadine in human liver microsomes: role of cytochrome P-450 3A (4) in N-dealkylation and C-hydroxylation. Drug Metab Dispos 1993; 21(3): 403–9
Kronbach T, Mathys D, Umeno M, et al. Oxidation of midazolam and triazolam by human liver cytochrome P450IIIA4. Mol Pharmacol 1989; 36(1): 89–96
Tsunoda SM, Velez RL, von Moltke LL, et al. Differentiation of intestinal and hepatic cytochrome P450 3A activity with use of midazolam as an in vivo probe: effect of ketoconazole. Clin Pharmacol Ther 1999; 66(5): 461–71
Venkatakrishnan K, von Moltke LL, Greenblatt DJ. Effects of the antifungal agents on oxidative drug metabolism: clinical relevance. Clin Pharmacokinet 2000; 38(2): 111–80
Takano M, Hasegawa R, Fukuda T, et al. Interaction with P-glycoprotein and transport of erythromycin, midazolam and ketoconazole in Caco-2 cells. Eur J Pharmacol 1998; 358(3): 289–94
Chang T, Benet LZ, Hebert MF. The effect of water-soluble vitamin E on cyclosporine pharmacokinetics in healthy volunteers. Clin Pharmacol Ther 1996; 59(3): 297–303
Block GA, Martin KJ, de Francisco AL, et al. Cinacalcet for secondary hyperparathyroidism in patients receiving hemodialysis. N Engl J Med 2004; 350(15): 1516–25
Lindberg JS, Culleton B, Wong G, et al. Cinacalcet HCl, an oral calcimimetic agent for the treatment of secondary hyper-parathyroidism in hemodialysis and peritoneal dialysis: a randomized, double-blind, multicenter study. J Am Soc Nephrol 2005; 16(3): 800–7
Charytan C, Coburn JW, Chonchol M, et al. Cinacalcet hydrochloride is an effective treatment for secondary hyperparathyroidism in patients with CKD not receiving dialysis. Am J Kidney Dis 2005; 46(1): 58–67
Harris RZ, Padhi D, Marbury TC, et al. Pharmacokinetics, pharmacodynamics, and safety of cinacalcet hydrochloride in hemodialysis patients at doses up to 200mg once daily. Am J Kidney Dis 2004; 44(6): 1070–6
Acknowledgements
This study (Study 20010206) was sponsored by Amgen Inc. and was conducted at MDS Pharma Services (Lincoln, NB, USA). Drs Harris, Sullivan and Padhi and Ms Salfi are employees and stockholders of Amgen Inc. Dr William Stark (also an employee and stockholder of Amgen Inc.) and Dr J. Megginson Hollister contributed to the writing of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Harris, R.Z., Salfi, M., Sullivan, J.T. et al. Pharmacokinetics of Cinacalcet Hydrochloride When Administered with Ketoconazole. Clin Pharmacokinet 46, 495–501 (2007). https://doi.org/10.2165/00003088-200746060-00003
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-200746060-00003