Abstract
Background
Clinical trials in children are being encouraged by regulatory authorities in light of the immense off-label and unlicensed use of drugs in the paediatric population. The use of in silico techniques for pharmacokinetic prediction will aid in the development of paediatric clinical trials by guiding dosing regimens, ensuring efficient blood sampling times, maximising therapeutic effect and potentially reducing the number of children required for the study. The goal of this study was to extend an existing physiologically based pharmacokinetic (PBPK) model for adults to reflect the age-related physiological changes in children from birth to 18 years of age and, in conjunction with a previously developed age-specific clearance model, to evaluate the accuracy of the paediatric PBPK model to predict paediatric plasma profiles.
Methods
The age-dependence of bodyweight, height, organ weights, blood flows, interstitial space and vascular space were taken from the literature. Physiological parameters that were used in the PBPK model were checked against literature values to ensure consistency. These included cardiac output, portal vein flow, extracellular water, total body water, lipid and protein. Five model compounds (paracetamol [acetaminophen], alfentanil, morphine, theophylline and levofloxacin) were then examined by gathering the plasma concentration-time profiles, volumes of distribution and elimination half-lives from different ages of children and adults. First, the adult data were used to ensure accurate prediction of pharmacokinetic profiles. The model was then scaled to the specific age of children in the study, including the scaling of clearance, and the generated plasma concentration profiles, volumes of distribution and elimination half-lives were compared with literature values.
Results
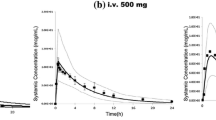
Physiological scaling produced highly age-dependent cardiac output, portal vein flow, extracellular water, total body water, lipid and protein values that well represented literature data. The pharmacokinetic profiles in children for the five compounds were well predicted and the trends associated with age were evident. Thus, young neonates had plasma concentrations greater than the adults and older children had concentrations less than the adults. Eighty-three percent, 97% and 87% of the predicted plasma concentrations, volumes of distribution and elimination half-lives, respectively, were within 50% of the study reported values. There was no age-dependent bias for term neonates to 18 years of age when examining volumes of distribution and elimination half-lives.
Conclusion
This study suggests that the developed paediatric PBPK model can be used to scale pharmacokinetics from adults. The accurate prediction of pharmacokinetic parameters in children will aid in the development of dosing regimens and sampling times, thus increasing the efficiency of paediatric clinical trials.















Similar content being viewed by others
Notes
The use of trade names is for product identification purposes only and does not imply endorsement.
References
Best Pharmaceuticals for Children Act, January 4, 2002 (Public Law No. 107–109). USA 2005
Roberts R, Rodriguez W, Murphy D, et al. Pediatric drug labeling: improving the safety and efficacy of pediatric therapies. JAMA 2003; 290(7): 905–11
Choonara I. Unlicensed and off-label drug use in children: implications for safety. Expert Opin Drug Saf 2004; 3(2): 81–3
Conroy S, Choonara I, Impicciatore P, et al. Survey of unlicensed and off label drug use in paediatric wards in European countries. European Network for Drug Investigation in Children. BMJ 2000; 320(7227): 79–82
Horen B, Montastruc JL, Lapeyre-Mestre M. Adverse drug reactions and off-label drug use in paediatric outpatients. Br J Clin Pharmacol 2002; 54(6): 665–70
Turner S, Nunn AJ, Fielding K, et al. Adverse drug reactions to unlicensed and off-label drugs on paediatric wards: a prospective study. Acta Paediatr 1999; 88(9): 965–8
Kauffman RE, Kearns GL. Pharmacokinetic studies in paediatric patients: clinical and ethical considerations. Clin Pharmacokinet 1992; 23(1): 10–29
Gobburu JV, Marroum PJ. Utilisation of pharmacokinetic-pharmacodynamic modelling and simulation in regulatory decision-making. Clin Pharmacokinet 2001; 40(12): 883–92
Bhattaram VA, Booth BP, Ramchandani RP, et al. Impact of pharmacometrics on drug approval and labeling decisions: a survey of 42 new drug applications. AAPS J 2005; 7(3): E503–12
Nestorov I. Whole body pharmacokinetic models. Clin Pharmacokinet 2003; 42(10): 883–908
Alcorn J, McNamara PJ. Pharmacokinetics in the newborn. Adv Drug Deliv Rev 2003; 55(5): 667–86
Alcorn J, McNamara PJ. Ontogeny of hepatic and renal systemic clearance pathways in infants: part I. Clin Pharmacokinet 2002; 41(12): 959–98
Ginsberg G, Hattis D, Miller RM, et al. Pediatric pharmacokinetic data: implications for environmental risk assessment for children. Pediatrics 2004; 113(4): 973–83
Edginton AN, Schmitt W, Voith B, et al. A mechanistic approach for the scaling of clearance in children. Clin Pharmacokinet 2006; 45(7): 683–704
Johnson TN. Modelling approaches to dose estimation in children. Br J Clin Pharmacol 2005; 59(6): 663–9
Bjorkman S. Prediction of drug disposition in infants and children by means of physiologically based pharmacokinetic (PBPK) modelling: theophylline and midazolam as model drugs. Br J Clin Pharmacol 2005; 59(6): 691–704
Ginsberg G, Hattis D, Russ A, et al. Physiologically based pharmacokinetic (PBPK) modeling of caffeine and theophylline in neonates and adults: implications for assessing children’s risks from environmental agents. J Toxicol Environ Health A 2004; 67(4): 297–329
Willmann S, Lippert J, Schmitt W. From physicochemistry to absorption and distribution: predictive mechanistic modelling and computational tools. Expert Opin Drug Metab Toxicol 2005; 1(1): 159–68
Willmann S, Lippert J, Sevestre M, et al. PK-Sim®: a physiologically based pharmacokinetic ‘whole-body’ model. Biosilico 2003; 1(4): 121–4
PK-Sim® software manual [online]. Available from URL: http://www.pk-sim.com [Accessed 2006 August 21]
Syracuse Research Corporation. SRC PhysProp database [online]. Available from URL: http://www.syrres.com/esc/phys-demo.htm [Accessed 2005 Aug 10]
Bailey DN, Briggs JR. The binding of selected therapeutic drugs to human serum [alpha]-1 acid glycoprotein and to human serum albumin in vitro. Ther Drug Monit 2004; 26(1): 40–3
Hardman JG, Limbird LE, Gilman A. Goodman and Gilman’s: the pharmacological basis of therapeutics. 10th ed. New York: McGraw Hill, 2001
Roure P, Jean N, Leclerc AC, et al. Pharmacokinetics of alfentanil in children undergoing surgery. Br J Anaesth 1987; 59(11): 1437–40
Macfie AG, Magides AD, Reilly CS. Disposition of alfentanil in burns patients. Br J Anaesth 1992; 69(5): 447–50
Kart T, Christrup LL, Rasmussen M. Recommended use of morphine in neonates, infants and children based on a literature review: Part 1 — pharmacokinetics. Paediatr Anaesth 1997; 7(1): 5–11
Valko K, Nunhuck S, Bevan C, et al. Fast gradient HPLC method to determine compounds binding to human serum albumin. Relationships with octanol/water and immobilized artificial membrane lipophilicity. J Pharm Sci 2003; 92(11): 2236–48
Dobashi A. Levofloxacin. Department of Pharmaceutical Information Science, Tokyo University of Pharmacy and Life Science [online]. Available from URL: http://www.pharmis.org/database/structure/l_group/levofloxacin.html [Accessed 2005 Aug 23]
Fish DN, Chow AT. The clinical pharmacokinetics of levofloxacin. Clin Pharmacokinet 1997; 32(2): 101–19
International Commission on Radiological Protection (ICRP). Basic anatomical and physiological data for use in radiological protection: reference values. ICRP publication 89. Amsterdam: Elsevier Science, 2002
Segel SA, Fanelli CG, Dence CS, et al. Blood-to-brain glucose transport, cerebral glucose metabolism, and cerebral blood flow are not increased after hypoglycemia. Diabetes 2001; 50(8): 1911–7
Epstein HT. Stages of increased cerebral blood flow accompany stages of rapid brain growth. Brain Dev 1999; 21(8): 535–9
Raynaud C, Chiron C, Maziere B, et al. Followup of regional CBF in children from birth to 18 years with Xe-133 [abstract]. J Nucl Med 1990; 31S: 892
Wintermark M, Lepori D, Cotting J, et al. Brain perfusion in children: evolution with age assessed by quantitative perfusion computed tomography. Pediatrics 2004; 113(6): 1642–52
Visser MOJM, Leighton JO, van de Bor M, et al. Renal blood flow in neonates: quantification with color flow and pulsed doppler US. Radiology 1992; 183(2): 441–4
Scholbach T. Color doppler sonographic determination of renal blood flow in healthy children. J Ultrasound Med 1999; 18(8): 559–64
Scholbach T. Changes of renal blood flow volume in the hemolytic-uremic syndrome-color Doppler sonographic investigations. Pediatr Nephrol 2001; 16(8): 644–7
Raitakari M, Nuutila P, Ruotsalainen U, et al. Evidence for dissociation of insulin stimulation of blood flow and glucose uptake in human skeletal muscle: studies using [15O]H2O, [18F]fluoro-2-deoxy-D-glucose, and positron emission tomography. Diabetes 1996; 45(11): 1471–7
Skovranek J, Samanek M. Chronic impairment of leg muscle blood flow following cardiac catheterization in childhood. AJR Am J Roentgenol 1979; 132(1): 71–5
Wu PYK, Wong WH, Guerra G, et al. Peripheral blood flow in the neonate: 1. changes in total, skin, and muscle blood flow with gestational and postnatal age. Pediatr Res 1980; 14(12): 1374–8
Goetzova J, Skovranek J, Samanek M. Muscle blood flow in children, measured by 133Xe clearance method. Cor Vasa 1977; 19(2): 161–4
Mikasa H, Sakuragi T, Higa K, et al. Skin blood flow and plasma catecholamine concentrations during removal of a phaeochromocytoma in a child. Br J Anaesth 2004; 92(5): 757–60
Chimoskey JE. Skin blood flow by 133Xe disappearance validated be venous occlusion plethysmography. J Appl Physiol 1972; 32(3): 432–4
Irazuzta JE, Berde CB, Sethna NF. Laser Doppler measurements of skin blood flow before, during, and after lumbar sympathetic blockade in children and young adults with reflex sympathetic dystrophy syndrome. J Clin Monit 1992; 8(1): 16–9
Moustogiannis AN, Raju TN, Roohey T, et al. Intravenous morphine attenuates pain induced changes in skin blood flow in newborn infants. Neurol Res 1996; 18(5): 440–4
Leggett RW, Williams LR. A proposed blood circulation model for reference man. Health Phys 1995; 69(2): 187–201
Kagimoto S, Fujitsuka S, Kinoshita K, et al. Study to establish normal values for portal vein blood flow in children using a duplex ultrasound system. Acta Paediatr Jpn 1991; 33(6): 693–6
Lopez Barrio AM, De Palma Gaston MA, Munoz Conde J. Evaluation of the portal blood flow in healthy children by doppler duplex echography. An Esp Pediatr 1996; 44(1): 45–9
Greenway CV, Stark RD. Hepatic vascular bed. Physiol Rev 1971; 51(1): 23–5
Winso O, Biber B, Gustavsson B, et al. Portal blood flow in man during graded positive end-expiratory pressure ventilation. Intensive Care Med 1985; 12(2): 80–8
Chang AC, Atz AM, Wernovsky G, et al. Milrinone: systemic and pulmonary hemodynamic effects in neonates after cardiac surgery. Crit Care Med 1995; 23(11): 1907–14
Sholler GF, Celermajer JM, Whight CM, et al. Echo Doppler assessment of cardiac output and its relation to growth in normal infants [published erratum appears in Am J Cardiol 1988; 61 (10): 872]. Am J Cardiol 1987; 60(13): 1112–9
Cecchetti C, Stoppa F, Vanacore N, et al. Monitoring of intrathoracic volemia and cardiac output in critically ill children. Minerva Anestesiol 2003; 69(12): 907–18
Shekerdemian LS, Bush A, Lincoln C, et al. Cardiopulmonary interactions in healthy children and children after simple cardiac surgery: the effects of positive and negative pressure ventilation. Heart 1997; 78(6): 587–93
Pastore E, Turchetta A, Attias L, et al. Cardiorespiratory functional assessment after pediatric heart transplantation. Pediatr Transplant 2001; 5(6): 425–9
Pianosi PT. Measurement of exercise cardiac output by thoracic impedance in healthy children. Eur J Appl Physiol 2004; 92(4–5): 425–30
Kardos A, Vereczkey G, Pirot L, et al. Use of impedance cardiography to monitor haemodynamic changes during laparoscopy in children. Paediatr Anaesth 2001; 11(2): 175–9
Hjortdal VE, Emmersten K, Stenbog E, et al. Effects of exercise and respiration on blood flow in total cavopulmonary connection: a real-time magnetic resonance flow study. Circulation 2003; 108(10): 1227–31
Kawai R, Lemaire M, Steimer J, et al. Physiologically based pharmacokinetic study on a cyclosporin derivative, SDZ IMM 125. J Pharmacokinet Biopharm 1994; 22(5): 327–3
Tsuji A, Yoshikawa T, Nishide K, et al. Physiologically based pharmacokinetic model for beta-lactam antibiotics I: tissue distribution and elimination in rats. J Pharm Sci 1983; 72(11): 1239–52
Baker GL. Human adipose tissue composition and age. Am J Clin Nutr 1969; 22(7): 829–35
Dickerson JWT, Widdowson EM. Chemical changes in skeletal muscle during development. Biochemistry 1960; 74: 247–57
Levitt DG. The pharmacokinetics of the interstitial space in humans. BMC Clin Pharmacol 2003; 3: 3
Boulton TJ, Dunlop M, Court JM. The growth and development of fat cells in infancy. Pediatr Res 1978; 12(9): 908–9
DeJongh J, Verhaar HJ, Hermens JL. A quantitative property-property relationship (QPPR) approach to estimate in vitro tissue-blood partition coefficients of organic chemicals in rats and humans. Arch Toxicol 1997; 72(1): 17–25
Poulin P, Theil FP. Prediction of pharmacokinetics prior to in vivo studies. 1. Mechanism-based prediction of volume of distribution. J Pharm Sci 2001; 91(1): 129–56
Friss-Hansen B. Water distribution in the foetus and newborn infant. Acta Paediatr Scand 1983; 305(S): 1–7
Battistini N, Virgili F, Severi S, et al. Relative expansion of extracellular water in obese vs. normal children. J Appl Physiol 1995; 79(1): 94–6
Hamadeh MJ, Robitaille L, Boismenu D, et al. Human extracellular water volume can be measured using the stable isotope Na2 34SO4 1, 2. J Nutr 1999; 129: 722–7
Haschke F, Fomon SJ, Zeigler EE. Body composition of a nine-year-old reference boy. Pediatr Res 1981; 15(5): 847–9
Fomon SJ, Haschke F, Ziegler EE, et al. Body composition of reference children from birth to age 10 years. Am J Clin Nutr 1982; 35(5S): 1169–75
Norberg A, Sandhagen B, Bratteby LE, et al. Do ethanol and deuterium oxide distribute into the same water space in healthy volunteers? Alcohol Clin Exp Res 2001; 25(10): 1423–30
Zeigler EE, O’Donnell AM, Nelson SE, et al. Body composition of the reference fetus. Growth 1976; 40(4): 329–41
Depre M, van Hecken A, Verbesselt R, et al. Tolerance and pharmacokinetics of propacetamol, a paracetamol formulation for intravenous use. Fundam Clin Pharmacol 1992; 6(6): 259–62
Bannwarth B, Netter P, Lapicque F, et al. Plasma and cerebrospinal fluid concentrations of paracetamol after a single intravenous dose of propacetamol. Br J Clin Pharmacol 1992; 34(1): 79–81
Egan TD, Minto CF, Hermann DJ, et al. Remifentanil versus alfentanil: comparative pharmacokinetics and pharmacodynamics in healthy adult male volunteers. Anesthesiology 1996; 84(4): 821–33
Kharasch ED, Russell M, Garton K, et al. Assessment of cytochrome P450 3A4 activity during the menstrual cycle using alfentanil as a noninvasive probe. Anesthesiology 1997; 87(1): 26–35
Baillie SP, Bateman DN, Coates PE, et al. Age and the pharmacokinetics of morphine. Age Aging 1989; 18(4): 258–62
Skarke C, Schmidt H, Geisslinger G, et al. Pharmacokinetics of morphine are not altered in subjects with Gilbert’s syndrome. Br J Clin Pharmacol 2003; 56(2): 228–31
Stringer KA, Mallet J, Clarke M, et al. The effect of three different oral doses of verapamil on the disposition of theophylline. Eur J Clin Pharmacol 1992; 43(1): 35–8
Gisclon LG, Curtin CR, Fowler CL, et al. Absence of a pharmacokinetic interaction between intravenous theophylline and orally administered levofloxacin. J Clin Pharmacol 1997; 37(8): 744–50
Chien S, Wells TG, Blumer JL, et al. Levofloxacin pharmacokinetics in children. J Clin Pharmacol 2005; 45(2): 153–60
McNamara PJ, Alcorn J. Protein binding predictions in infants. AAPS PharmSci 2002; 4(1): E4
Holford NHG, Hale M, Ko HC, et al., editors. Simulation in drug development: good practices. San Francisco (CA): Center for Drug Development Science, University of California San Francisco, 1999
Sheiner LB, Beal SL. Some suggestions for measuring predictive performance. J Pharmacokinet Biopharm 1981; 9(4): 503–12
Boyd E. The growth of the surface area of the human body. Minneapolis: The University of Minnesota Press, 1935
Tachibana M, Tanaka M, Masubuchi Y, et al. Acyl glucuronidation of fluoroquinolone antibiotics by the UDP-glucuronosyl-transferase 1A subfamily in human liver microsomes. Drug Metab Dispos 2005; 33(6): 803–11
de Wildt SN, Kearns GL, Leeder JS, et al. Glucuronidation in humans. Pharmacogenetic and developmental aspects. Clin Pharmacokinet 1999; 36(6): 439–52
Allegaert K, Van der Marel CD, Debeer A, et al. Pharmacokinetics of single dose intravenous propacetamol in neonates: effect of gestational age. Arch Dis Child Fetal Neonatal Ed 2004; 89(1): F25–8
Autret E, Dutertre JP, Breteau M, et al. Pharmacokinetics of paracetamol in the neonate and infant after administration of propacetamol chlorhydrate. Dev Pharmacol Ther 1993; 20(3–4): 129–34
Granry JC, Rod B, Boccard E, et al. Pharmacokinetics and antipyretic effects of an injectable pro-drug of paracetamol (propacetamol) in children. Paediatr Anaesthesiol 1992; 2: 291–5
Mikkelsen S, Feilberg VL, Chistensen CB, et al. Morphine pharmacokinetics in premature and mature newborn infants. Acta Paediatr 1994; 83(10): 1025–8
Hain RD, Hardcastle A, Pinkerton CR, et al. Morphine and morphine-6-glucuronide in the plasma and cerebrospinal fluid of children. Br J Clin Pharmacol 1999; 48(1): 37–42
Loughnan PM, Sitar DS, Ogilvie RI, et al. Pharmacokinetic analysis of the disposition of intravenous theophylline in young children. J Pediatr 1976; 88(5): 874–9
Arnold JD, Hill GN, Sansom LN. A comparison of the pharmacokinetics of theophylline in asthmatic children in the acute episode and in remission. Eur J Clin Pharmacol 1981; 20(6): 443–7
Ellis EF, Koysooko R, Levy G. Pharmacokinetics of theophylline in children with asthma. Pediatrics 1976; 58(4): 542–7
Goresky GV, Koren G, Sabourin MA, et al. The pharmacokinetics of alfentanil in children. Anesthesiology 1987; 67(5): 654–9
Meistelman C, Saint-Maurice C, Lepaul M, et al. A comparison of alfentanil pharmacokinetics in children and adults. Anesthesiology 1987; 66(1): 13–6
Marlow N, Weindling AM, Van Peer A, et al. Alfentanil pharmacokinetics in preterm infants. Arch Dis Child 1990; 65(4): 349–51
Davis PJ, Killian A, Stiller RL, et al. Pharmacokinetics of alfentanil in newborn premature infants and older children. Dev Pharmacol Ther 1989; 13(1): 21–7
Killian A, Davis PJ, Stiller RL, et al. Influence of gestational age on pharmacokinetics of alfentanil in neonates Dev Pharmacol Ther 1990; 15(2): 82–5
den Hollander JM, Hennis PJ, Burm AG, et al. Alfentanil in infants and children with congenital heart defects. J Cardiothorac Anesth 1998; 2(1): 12–7
Bhat R, Chari G, Gulati A, et al. Pharmacokinetics of a single dose of morphine in preterm infants during the first week of life. J Pediatr 1990; 117(3): 477–81
Olkkola KT, Maunuksela EL, Korpela R, et al. Kinetics and dynamics of postoperative intravenous morphine in children. Clin Pharmacol Ther 1988; 44(2): 128–36
Pokela ML, Olkkola KT, Seppala T, et al. Age-related morphine kinetics in infants. Dev Pharmacol Ther 1993; 20(1–2): 26–34
Aranda JV, Sitar DS, Parsons WD, et al. Pharmacokinetic aspects of theophylline in premature newborns. N Engl J Med 1976; 295(8): 413–6
Simons FE, Simons KJ. Pharmacokinetics of theophylline in infancy. J Clin Pharmacol 1978; 18(10): 472–6
Vichyanond P, Aranyanark N, Visitsuntorn N, et al. Theophylline pharmacokinetics in Thai children. Asian Pac J Allergy Immunol 1994; 12(2): 137–43
Agbaba D, Pokrajac M, Varagic VM, et al. Dependence of the renal excretion of theophylline on its plasma concentrations and urine flow rate in asthmatic children. J Pharmacol 1990; 42(12): 827–30
Pohl HR, van Engelen JG, Wilson J, et al. Risk assessment of chemicals and pharmaceuticals in the pediatric population: a workshop report. Regul Toxicol Pharmacol 2005; 42(1): 83–95
Acknowledgements
No specific funding was received for this project. All authors are employed by Bayer Technology Services (BTS) GmbH.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Edginton, A.N., Schmitt, W. & Willmann, S. Development and Evaluation of a Generic Physiologically Based Pharmacokinetic Model for Children. Clin Pharmacokinet 45, 1013–1034 (2006). https://doi.org/10.2165/00003088-200645100-00005
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-200645100-00005