Summary
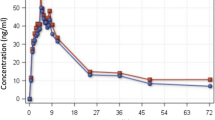
Stereoselective pharmacokinetic properties of chloroquine were investigated in humans after a single oral dose of the separate enantiomers. The study was carried out according to a crossover experimental design with a washout period between the administration of each enantiomer. Total blood chloroquine concentrations were measured using an achiral high performance liquid chromatography method.
Terminal half-life (t½λz z) and mean residence time (MRT) were longer for (R)-chloroquine (294h and 388h, respectively) than for (S)-chloroquine (236h and 272h, respectively). The total body clearance was lower for the (R)-enantiomer [136 ± 38 ml/min (8.16 ± 2.28 L/h)] than for the (S)-enantiomer [237 ± 71 ml/min (14.22 ± 4.26 L/h)]. Although the (R)-stereoisomer remained longer in the body, its volume of distribution (3410 ± 720L) was lower than that of (S)-chloroquine (4830 ± 1490L). Protein binding was different for both chloroquine stereoisomers, with opposite preferential binding to human albumin and α1-acid glycoprotein. Binding to total human plasma amounted to 66.6 ± 3.3% for (S)-chloroquine and to 42.7 ± 2.1% for the (R)-enantiomer.
De-ethyl-chloroquine concentrations were also different for both enantiomers, resulting in a statistically significant increase in the AUC of (S)-de-ethyl-chloroquine (12.9 ± 7.4 mg/L · h) compared with (R)-de-ethyl-chloroquine (6.29 ± 2.18 mg/L · h).
With a daily dosage regimen, the divergent pharmacokinetic behaviour of chloroquine enantiomers generates a calculated R: S ratio of blood concentrations amounting to 1: 0.7 at steady-state. Insufficient information about stereoselective activity and toxicity of chloroquine stereoisomers prevent further conclusions about the clinical consequences of these pharmacokinetic differences.
Similar content being viewed by others
References
Augustijns P, Verbeke N. HPLC method for the determination of chloroquine and its main metabolite in biological samples. Journal of Liquid Chromatography 13: 1203–1213, 1990
Blaschke G, Kraft HP, Schwanghart AD. Chloroquin-Enantiomere durch chromatographische Racemattrennung und Synthese. Chemische Berichte 111: 2732–2734, 1978
Conings L. Pharmacokinetics and metabolism of tocainide and its enantiomers in man, Ph.D Thesis, K.U. Leuven, Belgium, 1986
Drake NL, Creech HJ, Draper D, Garman JA, Haywood S, et al. Synthetic antimalarials: the preparation and properties of 7-cnloro-4-(4-diethylamino-1-methyl-butylamino)-quinoline (SN-7618). Journal of the American Chemical Society 68: 1214–1216, 1946
Essien EE, Ette EI, Thomas WOA, Brown-Awala EA. Chloroquine disposition in hypersensitive and non-hypersensitive subjects and its significance in chloroquine-induced pruritus. European Journal of Drug Metabolism and Pharmacokinetics 14: 71–77, 1989
Ette EI, Essien EE, Thomas WOA, Brown-Awala EA. Pharmacokinetics of chloroquine and some of its metabolites in healthy volunteers: a single dose study. Journal of Clinical Pharmacology 29: 457–462, 1989
Fink F, Minet G, Nickel P. Chloroquin-Enantiomere. Arzneimittel-Forschung 29: 163–164, 1979
Fu S, Björkman A, Wahlin B, Ofori-Adjei D, Ericsson Ö. In vitro activity of chloroquine, the two enantiomers of chloroquine, desethyl-chloroquine and pyronaridine against Plasmodium falciparum. British Journal of Clinical Pharmacology 22: 93–96, 1986
Gibaldi M, Perrier D. Pharmacokinetics, Marcel Dekker, Inc., New York, 1982
Haberkorn A, Kraft HP, Blaschke G. Antimalarial activity in animals of the optical isomers of chloroquine diphosphate. Tropical Medicine and Parasitology 30: 308–312, 1979
McLachlan AS, Tett SE, Cutler DS. High-performance liquid Chromatographic separation of the enantiomers of hydroxychloroquine and its major metabolites in biological fluids using an α1-acid glycoprotein stationary phase. Journal of Chromatography 570: 119–127, 1991
Ofori-Adjei D, Ericsson O, Lindström B, Hermansson J, Adjepon-Yamoah K, et al. Enantioselective analysis of chloroquine and desethyl-chloroquine after oral administration of racemic chloroquine. Therapeutic Drug Monitoring 8: 457–461, 1986a
Ofori-Adjei D, Ericsson O, Lindström B, Sjöqvist F. Protein binding of chloroquine enantiomers and desethyl-chloroquine. British Journal of Clinical Pharmacy 22: 356–358, 1986b
Titus ED. Recent developments in the understanding of the pharmacokinetics and mechanism of action of chloroquine. Therapeutic Drug Monitoring 11: 369–379, 1989
Tucker GT, Lennard MS. Enantiomer specific pharmacokinetics. Pharmacological Therapy 45: 309–329, 1990
Vanhoutte F, Vereecke J, Verbeke N, Carmeliet E. Stereoselective effects of the enantiomers of bupivacaine on the electrophysiological properties of the guinea-pig papillary muscle. British Journal of Clinical Pharmacology 103: 1275–1281, 1991
Veng-Pedersen P, Gillespie WR. A method for evaluating the mean residence times of metabolites in the body, systemic circulation, and the peripheral tissue not requiring separate IV administration of metabolite. Biopharmacy and Drug Disposition 8: 395–401, 1987
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Augustijns, P., Verbeke, N. Stereoselective Pharmacokinetic Properties of Chloroquine and De-Ethyl-Chloroquine in Humans. Clin. Pharmacokinet. 24, 259–269 (1993). https://doi.org/10.2165/00003088-199324030-00007
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-199324030-00007