Summary
The pharmacokinetics of oral gold (auranofin) in some respects resemble, and in other respects differ from, those of existing parenteral gold compounds such as gold sodium thiomalate (GST). This may in part relate to physicochemical differences as GST is a water-soluble polymeric compound in vitro whereas auranofin is lipid-soluble and characteristically monomeric. Furthermore, intramuscularly administered gold is > 95% bioavailable, whereas only 20 to 30% of an orally administered dose of auranofin is absorbed.
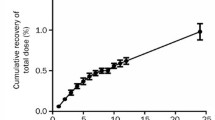
Following a standard 50mg intramuscular injection of GST, serum gold concentrations rise sharply, peaking between 4 and 8 mg/L in approximately 2 hours and declining to an average of 3 mg/L by 7 days. With repeated injections of GST stable serum concentrations of gold (3 to 5 mg/L) are eventually achieved (usually within 5 to 8 weeks) although absolute concentrations may vary widely between patients. On the other hand, long term treatment with auranofin is associated with lower and more stable serum concentrations of gold (0.5 to 0.7 mg/L), on the standard dosing regimen of 6mg daily. Both compounds are retained within the body for prolonged periods. However, the amount of gold retained with auranofin is significantly less compared with GST (less than 5% of a tracer dose of auranofin — about 20% of the absorbed dose — is retained by 100 days whereas the retention for a single labelled dose of GST over a similar interval is greater than 50%).
Excretory patterns of GST and auranofin also differ. Most of an absorbed dose of GST (> 70%) is excreted by the kidneys whereas only 50% of an absorbed (15% of an administered) dose of auranofin is excreted in the urine. Both compounds are avidly bound by plasma proteins and auranofin shows a particularly strong association with circulating cellular elements. In human subjects, parenterally administered gold is widely distributed among bodily tissues, showing a predilection for tissues of the reticuloendothelial system as well as the kidney and adrenal cortex. Comparable studies in humans are not available for auranofin but animal studies have shown comparatively less affinity for the liver, kidney and spleen.
Valuable insight has been gained in analysing the comparative pharmacokinetics of oral and injectable gold compounds. Unfortunately, attempts to correlate pharmacokinetic findings with clinical response or pharmacodynamic changes, as a whole, remain largely unsuccessful with these agents.
Similar content being viewed by others
References
Arrigoni-Martelli E, Branum E, Binderup L. D-penicillamine-like activity of thiols. EBRA Symposium on anti-inflammatory and anti-rheumatic agents, Paris, July 22, 1978
Berglof FE, Berglof K, Walz DT. Auranofin — an oral chrysotherapeutic agent for the treatment of rheumatoid arthritis. Journal of Rheumatology 5: 68–74, 1978
Blau SPL. Metabolism of gold during lactation. Arthritis and Rheumatism 16: 777–778, 1973
Blocka KLN, Furst DE, Landaw EM, Droomgoole S, Blomberg AL, et al. Single dose pharmacokinetics of auranofin in rheumatoid arthritis. Journal of Rheumatology 9 (Suppl. 8): 110–119, 1982
Blocka KLN, Landaw EM. The organic gold compounds: comparative pharmacokinetics. Auranofin 1982 — Proceedings of a Smith Kline & French International Symposium, Excerpta Medica, pp. 32–44, 1983
Calin A, Saunders D, Bennett R, et al. Auranofin: 1 or 9mg? The search for the appropriate dose. Journal of Rheumatology 9 (Suppl. 8): 146–148, 1982
Chaffman M, Brogden RN, Heel RC, Speight TM, Avery GS. Auranofin. A preliminary review of its pharmacological properties and therapeutic use in rheumatoid arthritis. Drugs 27: 378–424, 1984
Champion GD, Bieri D, Browne CD, et al. Determination of response of individual patients with rheumatoid arthritis to auranofin. International symposium on auranofin, Netherlands, November, 1982
Finkelstein AE, Walz DT, Batista V, et al. Auranofin, a new oral gold compound for treatment of rheumatoid arthritis. Annals of the Rheumatic Diseases 35: 251, 1976
Furst DE, Levine S, Srinivasan R, et al. A double blind trial of high versus conventional doses of gold salts for rheumatoid arthritis. Arthritis and Rheumatism 20: 1473, 1977
Gerber RC, Paulus HE, Jennrich RI, Lederer M, Bluestone R, et al. Gold kinetics following aurothiomalate therapy: use of a whole body radiation counter. Journal of Laboratory and Clinical Medicine 83: 778, 1974
Gerber RC, Paulus HE, Bluestone R, Lederer M. Kinetics of aurothiomalate in serum and synovial fluid. Arthritis and Rheumatism 15: 6, 1972a
Gerber RC, Paulus HE, Bluestone R, Pearson CM. Clinical response and serum gold levels in chrysotherapy. Annals of the Rheumatic Diseases 31: 308, 1972b
Ghadially FN. The aurosome. Journal of Rheumatology 6 (Suppl. 5): 45–50, 1979
Gottlieb NL, Smith PM, Smith EM. Tissue gold concentration in a rheumatoid arthritic receiving chrysotherapy. Arthritis and Rheumatism 15: 16–22, 1972a
Gottlieb NL, Smith PM, Smith EM. Gold excretion correlated with clinical course during chrysotherapy in rheumatoid arthritis. Arthritis and Rheumatism 15: 582–591, 1972b
Gottlieb NL, Smith PM, Penneys NS. Gold concentrations in hair, nail, and skin during chrysotherapy. Arthritis and Rheumatism 17: 56–62, 1974a
Gottlieb NL, Smith PM, Smith EM. Pharmacodynamics of AU197 and 195-labeled aurothiomalate in blood. Correlation with course of rheumatoid arthritis, gold toxicity and gold excretion. Arthritis and Rheumatism 17: 2, 1974b
Gottlieb NL, Major JC. Ocular chrysiasis correlated with gold concentrations in the crystalline lens during chrysotherapy. Arthritis and Rheumatism 21: 704–708, 1978
Gottlieb NL. Gold excretion and retention during the auranofin treatment: a preliminary report. Journal of Rheumatology 6 (Suppl. 5): 61, 1979
Gottlieb NL. Comparative pharmacokinetics of parenteral and oral gold compounds. Journal of Rheumatology 9 (Suppl. 8): 99–109, 1982
Grahame R, Billings R, Lawrence M, et al. Tissue gold levels after chrysotherapy. Annals of Rheumatic Diseases 33: 536–539, 1974
Herrlinger JD, Alsen C, Beress R, Hecker U, Weikert W. Distribution of gold serum erythrocytes and white blood cells after in vitro incubation and during chrysotherapy with different gold compounds. Journal of Rheumatology 9 (Suppl. 8): 81–89, 1982
Intoccia AP, Flanagan TL, Walz DT, et al. Pharmacokinetics of auranofin in animals. Journal of Rheumatology 9 (Suppl. 8): 90–98, 1982
Jellum E, Munthe E, Guldal G, Aaseth J. Fate of the gold and the thiomalate part after intramuscular administration of aurothiomalate to mice. Annals of the Rheumatic Diseases 39: 155–158, 1980
Jeffrey MR, Freundlich HF, Bailey DM. Distribution and excretion of radiogold in animals. Annals of the Rheumatic Diseases 17: 52–60, 1958
Kamel H, Brown DH, Ottaway JM, Smith WE, Cottney J, et al. Comparison of gold levels and distribution in guinea pig serum. Arthritis nd Rheumatism 21: 441–446, 1978
Krusius FE, Markkanen A, Deltola P. Plasma levels and urinary excretion of gold during routine treatment of rheumatoid arthritis. Annals of Rheumatic Diseases 29: 232–235, 1979
Laurence JS. Studies with radioactive gold. Annals of Rheumatic Diseases 20: 341, 1961
Lorber A, Cohen RL, Chan CC, Anderson HE. Gold determination in biological fluids by atomic absorption spectrophotometry: application to chyrosotherapy in rheumatoid arthritis patients. Arthritis and Rheumatism 22: 170, 1968
Lorber A, Simon TM, Kunishima DH, Harralson AF. Unbound serum gold: in vivo and in vitro correlates. (Abstract.) Arthritis and Rheumatism 25 (Suppl.): 548, 1982
Lorber A, Simon T, Leeb J, et al. Effect of chrysotherapy on parameters of the immune response. Journal of Rheumatology 6 (Suppl. 5): 82–90, 1979a
Lorber A, Simon T, Wilcox S. In vivo gold kinetics: lymphocyte binding and effect on lymphocyte responsiveness. (Abstract). Arthritis and Rheumatism 20: 126, 1977
Lorber A, Wilcox SA, Vibert GJ, Simon T. Carob rod atomization analysis: application to in vivo lymphocyte gold quantitation. Journal of Rheumatology 6 (Suppl. 5): 31, 1979b
Mascarhenas BR, Granda JL, Freyberg RH. Gold metabolism in patients with rheumatoid arthritis treated with gold compounds reinvestigated. Arthritis and Rheumatism 15: 4, 1972
Mason RW, Sharma RP, McQueen EG. Pharmacokinetics and subcellular interactions of gold complexes: significance in relation to therapeutic and side effects. In Rainsford KD et al. (Eds) Trace elements in the pathogenesis and treatment of inflammation, pp. 489–507, Birkhauser, Basel, 1981
McQueen EG, Dykes PW. Transport of gold in the body. Annals of the Rheumatic Diseases 28: 437, 1969
Norton WL, Lewis DC, Ziff M. Elecgron-dense deposits following injection of gold sodium thiomalate and thiomalic acid. Arthritis and Rheumatism 11: 436–443, 1968
Penneys NS, Kramer K, Gottlieb NL. The quantitative distribution of gold in skin during chrysotherapy. Journal of Investigative Dermatology 65: 331–333, 1975
Rubinstein HM, Dietz AA. Serum gold. II. Levels in rheumatoid arthritis. Annals of the Rheumatic Diseases 32: 128–132, 1973
Sadler PJ. The comparative evaluation of the physical and chemical properties of gold compounds. Journal of Rheumatology 9 (Suppl. 8): 71–78, 1982
Smith RT, Peak WP, Kron KM, et al. Increasing the effectiveness of gold therapy in rheumatoid arthritis. Journal of the American Medical Association 167: 1197–1204, 1958
Sutton BM, Hempel JC, Hill DT, Auranofin: its chemical development and unique molecular properties. International symposium on auranofin, Netherlands, November, 1982
Sutton BM, McCusty E, Walz DT, et al. Oral gold: anti-arthritic properties of alkylphosphine gold coordination complexes. Journal of Medical Chemistry 15: 1095, 1972
Thomas I, Ghadially FN. Aurosomes produced in the synvoial membrane by the oral administration of a gold compound SK&F 36914. Virchows Archiv Abt B Cell Pathology (Berlin) 105–109, 1977
Vernon-Roberts B, Jessop JD, Dore JL, et al. Selective concentration and localization of gold in macrophages of synovial and other tissues during and after chrysotherapy in rheumatoid patients. Annals of the Rheumatic Diseases 35: 477–486, 1976
Waller ES, Massarella JW, Crout JE, Yakatan GJ. The half of gold sodium thiomalate. (Letters.) Arthritis and Rheumatism 22: 1418–1419, 1979
Walz DT, DiMartino MJ, Chakrin LW, et al. Antiarthritis properties and unique pharmacologic profile of a potential chryosotherapeutic agent: SKF D-39162. Journal of Pharmacology and Experimental Therapeutics 197: 142, 1976
Walz DT, DiMartino MJ, Griswold DE. Comparative pharmacology and biological effects of different gold compounds. Journal of Rheumatology 9 (Suppl. 8): 54–60, 1982
Walz DT, Griswold DE, DiMartino MJ, Bumbier EE. Distribution of gold in blood following administration of auranofin (SKF D-39162). Journal of Rheumatology 6 (Suppl. 5): 56, 1979
Walz DT, Griswold DE, DiMartino MJ, Bumbier EE. Effect of auranofin dose regimen change upon cell-associated gold in rheumatoid arthritic patients. Journal of Rheumatology 8: 829–832, 1981
Weisman MH, Hardison WGM, Walz DT. Studies of the intestinal metabolism of oral gold. Journal of Rheumatology 7: 633–638, 1980
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Blocka, K.L.N., Paulus, H.E. & Furst, D.E. Clinical Pharmacokinetics of Oral and Injectable Gold Compounds. Clin-Pharmacokinet 11, 133–143 (1986). https://doi.org/10.2165/00003088-198611020-00003
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-198611020-00003