Summary
Due in the main to a genetically determined difference in the activity of the liver N-acelyltransferase, a number of primary amine drugs or drug metabolites such as dapsone, isoniazid, hydrallazine, phenelzine, procainamide, sulphadimidine, sulphapyridine and nitrazepam are subject to a bimodal acetylation in man.
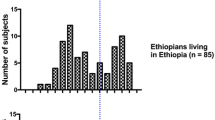
The population ratio of rapid versus slow acetylators varies widely between ethnic groups throughout the world, apparently being highest in those of an Eastern Asian origin and lowest in Egypt and some Western European countries. With some exceptions, the general clinical consequences of these differences in acetylator phenotype are, if any, that when patients are given a standard dose of the drugs mentioned, the slow acetylators are those who develop most adverse reactions while the rapid acetylators seem more prone to show an inadequate or lack of response to the standard dose.
The rationale behind dose adjustments based upon acetylator phenotyping is discussed, and it is tentatively concluded that more precise knowledge about the acetylator phenotype of the therapy. However, the possibility that a variety of pathophysiological factors and the concomitant therapy. However, the possibility that a variety of pathophysiological factors and the concomitant use of other drugs may interfere with acetylator phenotyping has to be considered, as well as some recent evidence that the acetylator phenotype may in itself represent a determinant for the development of certain diseases, like systemic lupus erythematosus and renal failure.
Similar content being viewed by others
References
Alarcon-Segovia, D.: Drug-induced antinuclear antibodies and lupus syndromes. Drugs 12: 69–77 (1976).
Alarcon-Segovia, D.; Fishbein, E. and Alcola, H.: Isoniazid acetylation rate and development of antinuclear antibodies upon isoniazid treatment. Arthritis and Rheumatism 14: 748–752 (1971).
Amlie, J.P.; Nesje, O.A.; Frislid, K.; Lunde, P.K.M. and Landmark, K.H.: Electrophysiological effects of N-acetylprocainamide as compared with procainamide in the dog heart in situ. Acta Pharmacologica et Toxicologica. In press (1977).
Armstrong, A.R. and Peart, H.E.: Comparison between the behaviour of eskimoes and non-eskimoes to the administration of isoniazid. American Review of Respiratory Diseases 81: 588–594 (1960).
Atkinson, A.J. Jr.; Parker, M. and Strong, J.: Rapid gas chromatographic measurement of plasma procainamide concentration. Clinical Chemistry 18: 643–646 (1972).
Berg, T. and Morland, J.: Induction of tryptophan oxygenase by dexamethasone in isolated hepatocytes. Biochimica et Biophysica Acta 392: 233–241 (1975).
Boxenbaum, H.G. and Reigelman, S.: Determination of isoniazid and metabolites in biological fluids. Journal of Pharmaceutical Sciences 63: 1191–1197 (1974).
Boxenbaum, H.G. and Riegelman, S.: Pharmacokinetics of isoniazid and some metabolites in man. Journal of Pharmacokinetics and Biopharmaceutics 4: 287–325 (1976).
Bonicke, R. and Reif, W.: Enzymatische Inaktiverung von Isonicotinsaure-hydrazid in menschlichen und tierischen Organismus. Archiv fur experimentelle Pathologie und Pharmakologie 220: 321–333 (1953).
Campbell, W.; Tikstone, W.J.; Lawson, D.H.; Hulton, I. and Lawrie, T.D.W.: Acetylator phenotype and the clinical pharmacology of slow release procainamide. British Journal of Clinical Pharmacology 3: 1023–1026 (1976).
Dahl, S.G.; Fremstad, D.; Jacobsen, S.; Kvaleid, I.; Landmark, K.H.; Lunde, P.K.M.; Marthinsen, A. Aa.; Nadland, K.J. and Waaler, T.: Pharmaceutical and pharmacokinetic principles in the formulation of a sustained-release procainamide tablet. Pharmaceutica Acta Helvetiae 51: 204–208 (1976).
Das, K.M. and Dubin, R.: Clinical pharmacokinetics of sulphasalazine. Clinical Pharmacokinetics 1: 406–425 (1976).
Das, K.M. and Eastwood, M.A.: Acetylation polymorphism of sulphapyridine in patients with ulcerative colitis and Crohns disease. Clin. Pharmacol. Ther. 18: 514–520 (1975).
Das, K.M.; Eastwood, M.A.; McManus, J.P.A. and Sircus, W.: Adverse reactions during salicylazosulfapyridine therapy and the relation with drug metabolism and acetylator phenotype. New England Journal of Medicine 289: 491–495.(1973).
Davies, D.M.; Beedie, M.A. and Rawlins, M.D.: Antinuclear antibodies during procainamide treatment and during acetylation. British Medical Journal 3: 682–683 (1975).
Devadatta, S.; Gangadharam, P.R.J.; Andrews, R.H.; Fox, W.; Ramakrishnan, C.V.; Selhon, J.B. and Veru, S.: Peripheral neuritis due to isoniazid. Bulletin of the World Health Organization 23: 587–598 (1960).
Drayer, D.E.; Reidenberg, M.M. and Sevy, R.W.: N-acetylprocainamide: An active metabolite of procainamide. Proceedings of the Society for Experimental Biology and Medicine 146: 358–363 (1974).
Ellard, G.A. and Gammon, P.T.: Pharmacokinetics of isoniazid metabolism in man. Journal of Pharmacokinetics and Biopharmaceutics 4: 83–113 (1976).
Ellard, G.A. and Gammon, P.T.: Acetylator phenotyping of tuberculosis patients using matrix isoniazid or sulphadimidine and its prognostic significance for treatment with several intermittent isoniazid-containing regimens. British Journal of Clinical Pharmacology 4: 5–14 (1977).
Ellard, G.A.; Gammon, P.T.; Helmy, H.S. and Rees, R.J.W.: Dapsone acetylation and the treatment of leprosy. Nature 239: 159–160 (1972).
Ellard, G.A.; Gammon, P.T.; Savin, J.A. and Tan, R.S.H.: Dapsone acetylation in dermatitis herpetiformis. British Journal of Dermatology 90: 441–444 (1974).
Elson, J.; Strong, J.M. and Atkinson, A.J. Jr.: Antiarrhythmic potency of N-acetylprocainamide. Clinical Pharmacology and Therapeutics 17: 134–140 (1975).
Evans, D.A.P.: Pharmacogenetique. Medecine et hygiene (Geneve) 20: 905 (1962).
Evans, D.A.P.: Conjoint clinic on drugs and genes. Journal of Chronic Diseases 18: 59–76 (1965).
Evans, D.A.P.: Genetic variations in acetylation of isoniazid and other drugs. Annals of New York Academy of Sciences 151(2): 723–733 (1968).
Evans, D.A.P.: An improved and simplified method of detecting the acetylator phenotype. Journal of Medical Genetics 6: 405–407 (1969).
Evans, D.A.P.; Davison, K. and Pratt, R.T.C.: The influence of acetylator phenotype on the effects of treating depression with phenelzine. Clinical Pharmacology and Therapeutics 6: 430–435 (1965).
Evans, D.A.P.; Manley, K.A. and McKusick, V.A.: Genetic control of isoniazid metabolism in man. British Medical Journal 2: 485–491 (1960).
Evans, D.A.P. and White, T.A.: Human acetylation polymorphism. Journal of laboratory and Clinical Medicine 63: 394–403 (1964).
Fine, A. and Sumner, D.J.: Determination of acetylator status in uremia. Brit. J. Clin. Pharmacol. 2: 475–476 (1975).
Frislid, K.; Berg, M.; Hansteen, V. and Lunde, P.K.M.: Comparison of the acetylation of procainamide and sulfadimidine in man. European Journal of Clinical Pharmacology 9: 433–438 (1976).
Frislid, K.; Bredesen, J.E. and Lunde, P.K.M.: Fluorometric or gas liquid chromatographic determination of procainamide? Clinical Chemistry 21: 1180–1181 (1975).
Forstrom, L.; Mattila, M.J. and Mustakallio, K.K.: Acetylator phenotype minimal maintenance dose and haemolytic effect of dapsone in dermatitis herpetiformis. Annals of Clinical Research 6: 308–310 (1974).
Gangadharam, P.R.J.; Bhatia, A.L.; Radhkrishna, S. and Selkon, J.B.: Rate of inactivation of isoniazid in South-Indian patients with pulmonary tuberculosis. Bulletin of the World Health Organization 25: 765–777 (1961).
Gelber, R.; Peters, J.H.; Gordon, G.R.; Glazko, A.J. and Levy, L.: The polymorphic acetylation of dapsone in man. Clinical Pharmacology and Therapeutics 12: 225–238 (1971).
Gibson, T.P.; Matusik, J.; Matusik, E.; Nelson, H.A.; Wilkinson, J. and Briggs, W.A.: Acetylation of procainamide in man and its relationship to isonicotinic acid hydrazide acetylation phenotype. Clinical Pharmacology and Therapeutics 17: 395–399 (1975).
Gilles, H.M.K. and Clyde, D.F.: Acetylator phenotype in sulphonamide resistant falciparum malaria. Annals of Tropical Medicines and Parasitology 68: 367–368 (1974).
Greenblatt, P.J. and Koch-Weser, J.: Clinical pharmacokinetics. N. Engl. J. Med. 293: 702-705, 964–970 (1975).
Govier, W.C.: Reticuloendothelial cells as the site of sulphanilamide acetylation in the rabbit. Journal of Pharmacology and Experimental Therapeutics 150: 305–308 (1965).
Hahn, B.H.; Sharp, G.C.; Irvin, W.S.; Kantor, O.S.; Gardner, C.A.; Babby, M.K.; Perry, H.M. and Osterland, C.K.: Immune responses to hydralazine and nuclear antigens in hydralazine-induced lupus erythematosus. Annals of Internal Medicine 76: 365–374 (1972).
Hanngren, A.; Borga, O. and Sjoqvist, F.: Inactivation of isoniazid (INH) in Swedish tuberculous patients before and during treatment with para-aminosalicylic acid (PAS). Scandinavian Journal of Respiratory Diseases 51: 61–69 (1970).
Hansteen, V.; Frislid, K. and Lunde, P.K.M.: Immunological side effects of drugs subjected to bimodal acetylation. In preparation (1977).
Hansteen, V.; Landmark, K.H.; Fremstad, D.; Dahl, S.G.; Jacobsen, S.; Marthinsen, A.Aa.; Waaler, T.; Frislid, K. and Lunde, P.K.M.: Maintenance therapy with a new retard tablet preparation of procainamide. American Heart Journal 92: 47–56 (1976).
Hashem, N.; Khalifa, S. and Nour, A.: The frequency of isoniazid acetylase enzyme deficiency among Egyptians. American Journal of Physical Anthropology 31: 97–102 (1969).
Hayward, G.A.: Human acetylation polymorphism in Polynesians. Proceedings of the University of Otago Medical School 53: 67–68 (1975).
Henningsen, N. Chr.; Cederberg, A.; Hanson, A. and Johansson, B.W.: Effects of long-time treatment with procainamide. Acta Medica Scandinavica 198: 475–482 (1975).
Hughes, H.B.; Biehl, J.P.; Jones, A.P. and Schmidt, L.H.: Metabolism of isoniazid in man as related to the occurrence of peripheral neuritis. American Review of Tuberculosis and Pulmonary Diseases 70: 266–273 (1954).
Irias, J.J.: Hydralazine-induced lupus erythematosus-like syndrome. American Journal of Diseases of Children 129: 862–864 (1975).
Jeanes, C.W.L.; Schaefer, O. and Eidus, L: Inactivation of isoniazid by Canadian eskimoes and indians. Canadian Medical Association Journal 106: 331 (1972).
Jeanes, C.W.L.; Schaefer, O. and Eidus, L.: Comparative blood levels and metabolism of INH and an INH-matrix preparation in fast and slow inactivators. Canadian Medical Association Journal 109: 483–487 (1973).
Jenne, J.W.: Partial purification and properties of the isoniazid transacetylase in human liver. Its relationship to the acetylation of p-aminosalicylic acid. Journal of Clinical Investigation 44: 1992–2002 (1965).
Jessamine, A.G.; Hamilton, E.J. and Eidus, L.: A clinical study of isoniazid inactivation. Canadian Medical Association Journal 89: 1214–1217 (1963).
Jounela, A.J.; Pasanen, M. and Mattila, J.J.: Acetylation phenotype and the antihypertensive response to hydralazine. Acta Medica Scandinavica 197: 303–306 (1975).
Johnstone, E.C. and Marsh, W.: Acetylator status and response to phenelzine in depressed patients. Lancet 1: 567–570 (1973).
Karim, A.K.M.B. and Evans, D.A.P.: Polymorphic acetylation of nitrazepam. Journal of Medical Genetics 13: 17–19 (1976).
Karlsson, E. and Molin, L.: Polymorphic acetylation of procainamide in healthy subjects. Acta medica Scandinavica 197: 299–302 (1975).
Karlsson, E.; Molin, L.; Norlander, B. and Sjoqvist, F.: Acetylation of procainamide in man studied with a new gas chromatographic method. British Journal of Clinical Pharmacology 1: 467–475 (1975).
Knight, R.A.; Selin, M.J. and Harris, H.W.: Genetic factors influencing isoniazid blood levels in humans. Veterans Administration 18th Conference of Chemotherapy of Tuberculosis, p.52 (Ed. by V.A. Department of Medicine and Surgery Central Office, Washington D.C. 1959).
Koch-Weser, J.: Hydralazine. New England Journal of Medicine 295: 320–323 (1976).
Koch-Weser, J. and Klein, S.W.: Procainamide dosage schedules, plasmaconcentrations and clinical effects. Journal of the American Medical Association 215: 1454–1460 (1971).
Kutt, H.; Brennan, R.; Dehejia, H. and Verebely, K.: Diphenyl-hydantoin intoxication. A complication of isoniazid therapy. American Review of Respiratory Disease 101: 377–384 (1970).
La Du, B.N.: Isoniazid and pseudocholinesterase polymorphism. Federation Proceedings 31: 1276–1285 (1972).
Larsson, R.; Karlsson,- E. and -Molin,- L.: Spontaneous-systemic lupus erythematosus and acetylator phenotype. Acta Medica Scandinavica. In press (1977).
Menon, N.K.: Madras study of supervised once-weekly chemotherapy. Bulletin of the International Union against Tuberculosis 41: 316–321 (1968).
Mitchell, J.R.; Nelson, S.D.; Thorgeirsson, U.P.; McMurthy, R.J. and Dybing, E.: Metabolic activation: Biochemical basis for many drug-induced liver injuries; in Popper and Schaffner (Eds): Progress in Liver Diseases p.259–297, vol. 5 (Grune and Straiten, New York 1976b).
Mitchell, J.R.; Thorgeirsson, U.P.; Black, M.; Timbroll, J.A.; Snodgrass, W.R.; Potter, W.Z.; Jollow, D.J. and Kelser, H.R.: Increased incidence of isoniazid hepatitis in rapid acetylators: possible relation to hydralazine metabolites. Clinical Pharmacology and Therapeutics 18: 70–79 (1975).
Mitchell, J.R.; Zimmermann, H.J.; Ishak, K.G.; Thorgeirsson, U.P.; Timbrell, J.A.; Snodgrass, W.R. and Nelson, S.D.: Isoniazid liver injury: Clinical spectrum, pathology and probable pathogenesis. Annals of Internal Medicine 84: 181–192 (1976a).
Mitchell, R.S.; Bell, J.C. and Riemensneider, D.K.: Further observations with isoniazid inactivation tests. Veterans administration 19th Conference of chemotherapy of Tuberculosis, p.62 (Ed. by VA Department of Medicine and Surgery Central Office, Washington D.C. 1960).
Mitchison, D.A.: Clinical applications of antibiotic and chemotherapeutic agents. Proceedings of the Royal Scoiety of Medicine 64: 537–540 (1971).
Molin, L.; Larsson, R. and Karlsson, E.: Evaluation of the sulfapyridine acetylator phenotyping test in healthy subjects and in patients with cardiac and renal diseases. Acta Medica Scandinavica. In press (1977).
Morland, J. and Olsen, H.: Personal communication (1976).
Pharmacogenetics: Report of a WHO Scientific Group. World Health Organization Technical Report Series, No.524, p. 19 (1973).
Perry, H.M. Jr.: Late toxicity to hydralazine resembling systemic lupus erythematosus or rheumatoid arthritis. American Journal of Medicine 54: 58–72 (1973).
Perry, H.M. Jr.; Tan, E.M.; Cordody, S. and Sahamato, A.: Relationship of acetyl transferase activity to antinuclear antibodies and toxic symptoms on hypertensive patients treated with hydralazine. Journal of Laboratory and Clinical Medicine 76: 114–125 (1970).
Peters, J.H.; Gordon, G.R. and Brown, P.: The relationship between the capacities of human subjects to acetylate isoniazid, sulphanilamid and sulphamethazin. Life Sci. 4: 99–107 (1965).
Peters, J.H.; Gordon, R.G.; Ghoul, D.C., Tolentino, J.G.; Walsh, G.P. and Levy, L.: The disposition of the antileprotic drug dapsone in Philippine subjects. American Journal of Tropical Medicine and Hygiene 21: 450–457 (1972).
Peters, J.H.; Gordon, G.R. and Karat, A.B.A.: Polymorphic acetylation of the antibacterials sulphamethazine and dapsone, in South Indian subjects. American Journal of Tropical Medicine and Hygiene 24: 641–648 (1975).
Peters, J.H. and Levy, L.: Dapsone acetylation in man. Another example of polymorphic acetylation. Annals of the New York Academy of Sciences 179: 660–666 (1971).
Peters, J.H.; Gordon, G.R.; Levy, L.; Storkan, M.A.; Jacobson, R.R.; Enna, C.D. and Kirchheimer, W.F.: Metabolic disposition of dapsone in patients with dapsone-resistant leprosy. American Journal of Tropical Medicine and Hygiene 23: 222–230 (1974).
Reidenberg, M.M.; Drayer, D.; DeMarco, A.L. and Bellow, C.T.: Hydralazine elimination in man. Clinical Pharmacology and Therapeutics 14: 970–977 (1973).
Reidenberg, M.M.; Drayer, D.D.; Levy, M. and Warner, H.: Polymorphic acetylation of procainamide in man. Clinical Pharmacology and Therapeutics 17: 722–730 (1975).
Reidenberg, M.M. and Martin, J.H.: The acetylator phenotype of patients with systemic lupus erythematosus. Drug Metabolism and Disposition 2: 71–73 (1974).
Remmer, H.: Induction of drug metabolizing enzyme system in the liver. European Journal of Clinical Pharmacology 5: 116–136 (1972).
Schroder, H. and Campbell, D.T.S.: Absorption, metabolism and excretion of salicylazosulphapyridine in man. Clinical Pharmacology and Therapeutics 13: 539–551 (1972).
Schroder, H. and Evans, D.A.P.: The polymorphic acetylation of sulphapyridine in man. Journal of Medical Genetics 9: 168–171 (1972a).
Schroder, H. and Evans, D.A.P.: Acetylation phenotype and adverse effects of sulphasalazine in healthy subjects. Gut 13: 278–284 (1972b).
Smith, S.E. and Kyi, T.: Inactivation of isoniazid in Burmese subjects. Nature (Lond.) 217: 1273 (1968).
Strandberg, J.; Boman, G.; Hassler, L. and Sjoqvist, F.: Acetylator phenotype in patients with hydralazine-induced lupoid syndrome. Acta Medica Scandinavica 200: 367–371 (1976).
Strong, J.M.; Dutscher, J.S.; Lee, W.-K. and Atkinson, A.J. Jr.: Pharmacokinetics in man of the N-acetylated metabolite of procainamide. Journal of Pharmacokinetics and Biopharmaceutics 3: 223–235 (1975).
Sunahara, S.; Urano, M. and Ogawa, M.: Genetical and geographic studies on isoniazid inactivation. Science 134: 1530–1531 (1961).
Talseth, T.: Studies on hydralazine I. Serum concentrations of hydralazine in man after a single dose and at steady state. European Journal of Clinical Pharmacology 10: 183–187 (1976b).
Talseth, T.: Studies on hydralazine II. Elimination rate and steady state concentration in patients with impaired renal function. European Journal of Clinical Pharmacology 10: 311–317 (1976b).
Tiitinen, H.; Mattila, M.J. and Eriksson, A.W.: Comparison of the isoniazid inactivation in Finns and Lapps. Annales Medicinae Internae Fenniae 57: 161–165 (1968).
Tuberculosis Chemotherapy Center, Madras: A controlled comparison of a twice-weekly and three once-weekly regimens in the initial treatment of pulmonary tuberculosis. Bulletin of the World Health Organization 43: 143–206 (1970).
Vizherova, A. and Slovekova, Z.: The determination of the acetylator phenotype of tuberculosis patients in Czechoslovakia using sulphadimidine. Tubercle 54: 67–71 (1973).
Walstad, R.A.: Inaktivisering av medikamenter ved acetylering. Tidsskrift for Den Norske Laegeforening 2: 100–103 (1975).
Weber, W.W.: Acetylating, deacetylating and amino acid conjugating enzymes; in Brodin and Gilette (Eds.) Handbook of Experimental Pharmacology, p.564–583, vol. 28, part 2 (Springer Verlag, Berlin 1971).
Weber, W.W.; Miceli, J.N.; Hearse, D.J. and Drummund, G.S.: N-acetylation of drugs. Pharmacogenetic studies in rabbits selected for their acetylator characteristics. Drug Metabolism and Disposition 4: 94–101 (1976).
Zacest, R. and Koch-Weser, J.: Relation of hydralazine plasma concentration to dosage and hypotensive action. Clinical Pharmacology and Therapeutics 13: 420–425 (1972).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lunde, P.K.M., Frislid, K. & Hansleen, V. Disease and Acetylation Polymorphism. Clin Pharmacokinet 2, 182–197 (1977). https://doi.org/10.2165/00003088-197702030-00003
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-197702030-00003