Abstract
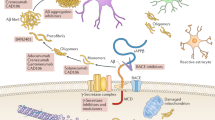
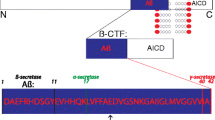
The underlying pathological cause of Alzheimer’s disease has been postulated to be an excess of amyloid-β (Aβ) which aggregates into toxic fibrillar deposits within the extracellular space of the brain, thereby disrupting neuronal and synaptic function and eventually leading to neuronal degeneration and dementia. As a result, therapeutic strategies have been developed that are designed to remove Aβ from the brain. Caution needs to be exercised concerning such strategies because, in addition to its presence in neuritic plaques, Aβ has a widespread distribution through the brain and body, even in cognitively normal individuals. Evidence indicates that instead of being a toxic peptide, soluble Aβ serves a variety of physiological functions, including modulation of synaptic function, facilitation of neuronal growth and survival, protection against oxidative stress, and surveillance against neuroactive compounds, toxins and pathogens. These physiological functions must be taken into account when strategies are developed to reduce Aβ load in Alzheimer’s disease. Ideally, such strategies should target forms of Aβ that are not bioavailable, such as fibrillar Aβ, or forms that are regarded to be overexpressed in Alzheimer’s disease (such as oligomers) while leaving normal soluble Aβ1–40 and β1–42 intact. At present none of the available therapeutic strategies appears to have such selectivity. Until these technical limitations and the uncertainties regarding the effect of depletion of Aβ from the brain are resolved, it would not be prudent to begin further clinical trials.
Similar content being viewed by others
References
Hardy J, Selkoe DJ. The amyloid hypothesis of Alzheimer’s disease: progress and problems on the road to therapeutics. Science 2002; 297(5580): 353–6
Selkoe DJ. The molecular pathology of Alzheimer’s disease. Neuron 1991; 6(4): 487–98
Robinson SR, Bishop GM. Aβ as a bioflocculant: implications for the amyloid hypothesis of Alzheimer’s disease. Neurobiol Aging 2002; 23(6): 1051–72
Schenk D, Barbour R, Dunn W, et al. Immunization with amyloid-β attenuates Alzheimer-disease-like pathology in the PDAPP mouse. Nature 1999; 400(6740): 173–7
Check E. Nerve inflammation halts trial for Alzheimer’s drug [letter]. Nature 2002; 415(6871): 462
Kunicki S, Richardson J, Mehta PD, et al. The effects of age, apolipoprotein E phenotype and gender on the concentration of amyloid-β (Aβ)40, Aβ542, apolipoprotein E and transthyretin in human cerebrospinal fluid. Clin Biochem 1998; 31(5): 409–15
Shoji M, Matsubara E, Kanai M, et al. Combination assay of CSF tau, Aβ1–40 and Aβ1–42(43) as a biochemical marker of Alzheimer’s disease. J Neurol Sci 1998; 158(2): 134–40
Tamaoka A, Fukushima T, Sawamura N, et al. Amyloid β protein in plasma from patients with sporadic Alzheimer’s disease. J Neurol Sci 1996; 141(1–2): 65–8
Coulson EJ, Paliga K, Beyreuther K, et al. What the evolution of the amyloid protein precursor supergene family tells us about its function. Neurochem Int 2000; 36(3): 175–84
Luo Y, Bolon B, Damore MA, et al. BACE1 (β-secretase) knockout mice do not acquire compensatory gene expression changes or develop neural lesions over time. Neurobiol Dis 2003; 14(1): 81–8
Hertz L, Dringen R, Schousboe A, et al. Astrocytes: glutamate producers for neurons. J Neurosci Res 1999; 57(4): 417–28
Ida N, Hartmann T, Pantel J, et al. Analysis of heterogeneous A4 peptides in human cerebrospinal fluid and blood by a newly developed sensitive Western blot assay. J Biol Chem 1996; 271(37): 22908–14
Shoji M, Kanai M, Matsubara E, et al. The levels of cerebrospinal fluid Aβ40 and Aβ42(43) are regulated age-dependently. Neurobiol Aging 2001; 22(2): 209–15
Blennow K, Sjogren M, Andreasen N. Usefulness of CSF total tau, Aβ42 and phospho tau as biomarkers for Alzheimer’s disease. In: Iqbal K, Winblad B, editors. Alzheimer’s disease and related disorders: research advances. Bucharest: ‘Ana Asian’ International Academy of Aging, 2003: 247–58
Mayeux R, Tang MX, Jacobs DM, et al. Plasma amyloid β-peptide 1–42 and incipient Alzheimer’s disease. Ann Neurol 1999; 46(3): 412–6
Mayeux R, Honig LS, Tang MX, et al. Plasma Aβ40 and Aβ42 and Alzheimer’s disease: relation to age, mortality, and risk. Neurology 2003; 61(9): 1185–90
Li QX, Whyte S, Tanner JE, et al. Secretion of Alzheimer’s disease Aβ amyloid peptide by activated human platelets. Lab Invest 1998; 78(4): 461–9
Armogida M, Petit A, Vincent B, et al. Endogenous β-amyloid production in presenilin-deficient embryonic mouse fibroblasts. Nat Cell Biol 2001; 3(11): 1030–3
Shibata M, Yamada S, Kumar SR, et al. Clearance of Alzheimer’s amyloid-β(l–40) peptide from brain by LDL receptor-related protein-1 at the blood-brain barrier. J Clin Invest 2000; 106(12): 1489–99
Savage MJ, Trusko SP, Howland DS, et al. Turnover of amyloid β-protein in mouse brain and acute reduction of its level by phorbol ester. J Neurosci 1998; 18(5): 1743–52
Mackic JB, Stins M, McComb JG, et al. Human blood-brain barrier receptors for Alzheimer’s amyloid-β 1β40: asymmetrical binding, endocytosis, and transcytosis at the apical side of brain microvascular endothelial cell monolayer. J Clin Invest 1998; 102(4): 734–43
Deane R, Du Yan S, Submamaryan RK, et al. RAGE mediates amyloid-β peptide transport across the blood-brain barrier and accumulation in brain. Nat Med 2003; 9(7): 907–13
Pan W, Solomon B, Maness LM, et al. Antibodies to β-amyloid decrease the blood-to-brain transfer of β-amyloid peptide. Exp Biol Med (Maywood) 2002; 227(8): 609–15
Dawson GR, Seabrook GR, Zheng H, et al. Age-related cognitive deficits, impaired long-term potentiation and reduction in synaptic marker density in mice lacking the β-amyloid precursor protein. Neuroscience 1999; 90(1): 1–13
Seabrook GR, Smith DW, Bowery BJ, et al. Mechanisms contributing to the deficits in hippocampal synaptic plasticity in mice lacking amyloid precursor protein. Neuro-pharmacology 1999; 38(3): 349–59
Huber G, Martin JR, Loffler J, et al. Involvement of amyloid precursor protein in memory formation in the rat: an indirect antibody approach. Brain Res 1993; 603(2): 348–52
Mileusnic R, Lancashire CL, Johnston AN, et al. APP is required during an early phase of memory formation. Eur J Neurosci 2000; 12(12): 4487–95
Dewachter I, Reverse D, Caluwaerts N, et al. Neuronal deficiency of presenilin 1 inhibits amyloid plaque formation and corrects hippocampal long-term potentiation but not a cognitive defect of amyloid precursor protein [V7171] transgenic mice. J Neurosci 2002; 22(9): 3445–53
Koistinaho M, Ort M, Cimadevilla JM, et al. Specific spatial learning deficits become severe with age in β-amyloid precursor protein transgenic mice that harbor diffuse β-amyloid deposits but do not form plaques. Proc Natl Acad Sci USA 2001; 98(25): 14675–80
Itoh A, Akaike T, Sokabe M, et al. Impairments of long-term potentiation in hippocampal slices of β-amyloid-infused rats. Eur J Pharmacol 1999; 382(3): 167–75
Raymond CR, Ireland DR, Abraham WC. NMDA receptor regulation by amyloid-β does not account for its inhibition of LTP in rat hippocampus. Brain Res 2003; 968(2): 263–72
Wu J, Anwyl R, Rowan MJ. β-amyloid-(l–40) increases long-term potentiation in rat hippocampus in vitro. Eur J Pharmacol 1995; 284(3): Rl–3
Kim JH, Anwyl R, Suh YH, et al. Use-dependent effects of amyloidogenic fragments of (β)-amyloid precursor protein on synaptic plasticity in rat hippocampus in vivo. J Neurosci 2001; 21(4): 1327–33
Trubetskaya VV, Stepanichev MY, Onufriev MV, et al. Administration of aggregated β-amyloid peptide (25β35) induces changes in long-term potentiation in the hippocampus in vivo. Neurosci Behav Physiol 2003; 33(2): 95–8
Morton RA, Kuenzi FM, Fitzjohn SM, et al. Impairment in hippocampal long-term potentiation in mice under-expressing the Alzheimer’s disease related gene presenilin-1. Neurosci Lett 2002; 319(1): 37–40
Walsh DM, Klyubin I, Fadeeva JV, et al. Naturally secreted oligomers of amyloid β protein potently inhibit hippocampal long-term potentiation in vivo. Nature 2002; 416(6880): 535–9
Wang HY, Lee DH, Davis CB, et al. Amyloid peptide Aβ1–42 binds selectively and with picomolar affinity to α7 nicotinic acetylcholine receptors. J Neurochem 2000; 75(3): 1155–61
Freir DB, Herron CE. Nicotine enhances the depressive actions of Aβ1–40 on long-term potentiation in the rat hippocampal CA1 region in vivo. J Neurophysiol 2003; 89(6): 2917–22
Mattson MP, Partin J, Begley JG. Amyloid β-peptide induces apoptosis-related events in synapses and dendrites. Brain Res 1998; 807(1–2): 167–76
Dougherty JJ, Wu J, Nichols RA. β-amyloid regulation of presynaptic nicotinic receptors in rat hippocampus and neocortex. J Neurosci 2003; 23(17): 6740–7
Kamenetz F, Tomita T, Hsieh H, et al. APP processing and synaptic function. Neuron 2003; 37(6): 925–37
Ikegaya Y, Matsuura S, Ueno S, et al. β-amyloid enhances glial glutamate uptake activity and attenuates synaptic efficacy. J Biol Chem 2002; 277(35): 32180–6
Koudinov AR, Koudinova NV. Amyloid β protein restores hippocampal long term potentiation: a central role for cholesterol. Neurobiol Lipids 2003; 1(8): 45–6
Whitson JS, Selkoe DJ, Cotman CW. Amyloid β protein enhances the survival of hippocampal neurons in vitro. Science 1989; 243(4897): 1488–90
Yankner BA, Duffy LK, Kirschner DA. Neurotrophic and neurotoxic effects of amyloid β protein: reversal by tachykinin neuropeptides. Science 1990; 250(4978): 279–82
Wang C, Wurtman RJ, Lee RK. Amyloid precursor protein and membrane phospholipids in primary cortical neurons increase with development, or after exposure to nerve growth factor or Aβ(l–40). Brain Res 2000; 865(2): 157–67
Phinney AL, Deller T, Stalder M, et al. Cerebral amyloid induces aberrant axonal sprouting and ectopic terminal formation in amyloid precursor protein transgenic mice. J Neurosci 1999; 19(19): 8552–9
Wujek JR, Dority MD, Frederickson RC, et al. Deposits of Aβ fibrils are not toxic to cortical and hippocampal neurons in vitro. Neurobiol Aging 1996; 17(1): 107–13
DeWitt DA, Silver J. Regenerative failure: a potential mechanism for neuritic dystrophy in Alzheimer’s disease. Exp Neurol 1996; 142(1): 103–10
Plant LD, Boyle JP, Smith IF, et al. The production of amyloid P peptide is a critical requirement for the viability of central neurons. J Neurosci 2003; 23(13): 5531–5
Bishop GM, Robinson SR. Human Aβ1–42 reduces iron-induced toxicity in rat cerebral cortex. J Neurosci Res 2003; 73(3): 316–23
Bishop GM, Robinson SR. Neurotoxic and neuroprotective: the paradox of amyloid-metal complexes in vivo. In: 8th International Conference on Alzheimer’s Disease and Related Disorders; 2002 Jul 20–25; Stockholm. Neurobiol Aging 2002; 23Suppl. 1S: S26
Zou K, Gong JS, Yanagisawa K, et al. A novel function of monomeric amyloid β-protein serving as an antioxidant molecule against metal-induced oxidative damage. J Neurosci 2002; 22(12): 4833–41
Butterfield DA. Amyloid β-peptide (l–42)-induced oxidative stress and neurotoxicity: implications for neurodegeneration in Alzheimer’s disease brain: a review. Free Radic Res 2002; 36(12): 1307–13
Kontush A, Berndt C, Weber W, et al. Amyloid-β is an antioxidant for lipoproteins in cerebrospinal fluid and plasma. Free Radic Biol Med 2001; 30(1): 119–28
Atwood CS, Moir RD, Huang X, et al. Dramatic aggregation of Alzheimer Aβ by Cu(II) is induced by conditions representing physiological acidosis. J Biol Chem 1998; 273(21): 12817–26
Garzon-Rodriguez W, Yatsimirsky AK, Glabe CG. Binding of Zn(II), Cu(II), and Fe(II) ions to Alzheimer’s Aβ peptide studied by fluorescence. Bioorg Med Chem Lett 1999; 9(15): 2243–8
Mantyh PW, Ghilardi JR, Rogers S, et al. Aluminum, iron, and zinc ions promote aggregation of physiological concentrations of β-amyloid peptide. J Neurochem 1993; 61(3): 1171–4
Nunomura A, Perry G, Aliev G, et al. Oxidative damage is the earliest event in Alzheimer disease. J Neuropathol Exp Neurol 2001; 60(8): 759–67
Kontush A, Donarski N, Beisiegel U. Resistance of human cerebrospinal fluid to in vitro oxidation is directly related to its amyloid-β content. Free Radic Res 2001; 35(5): 507–17
Kawahara M, Muramoto K, Kobayashi K, et al. Aluminum promotes the aggregation of Alzheimer’s amyloid β-protein in vitro. Biochem Biophys Res Commun 1994; 198(2): 531–5
Smith MA, Harris PL, Sayre LM, et al. Iron accumulation in Alzheimer disease is a source of redox-generated free radicals. Proc Natl Acad Sci U S A 1997; 94(18): 9866–8
Lovell MA, Robertson JD, Teesdale WJ, et al. Copper, iron and zinc in Alzheimer’s disease senile plaques. J Neurol Sci 1998; 158(1): 47–52
Golabek A, Marques MA, Lalowski M, et al. Amyloid β binding proteins in vitro and in normal human cerebrospinal fluid. Neurosci Lett 1995; 191(1–2): 79–82
Oyama R, Yamamoto H, Titani K. Glutamine synthetase, hemoglobin α-chain, and macrophage migration inhibitory factor binding to amyloid β-protein: their identification in rat brain by a novel affinity chromatography and in Alzheimer’s disease brain by immunoprecipitation. Biochim Biophys Acta 2000; 1479(1–2): 91–102
de Haas CJ. New insights into the role of serum amyloid P component, a novel lipopolysaccharide-binding protein. FEMS Immunol Med Microbiol 1999; 26(3–4): 197–202
de Haas CJ, van Leeuwen EM, van Bommel T, et al. Serum amyloid P component bound to Gram-negative bacteria prevents lipopolysaccharide-mediated classical pathway complement activation. Infect Immun 2000; 68(4): 1753–9
Andersen O, Vilsgaard Ravn K, Juul Sorensen I, et al. Serum amyloid P component binds to influenza A virus haemagglutinin and inhibits the virus infection in vitro. Scand J Immunol 1997; 46(4): 331–7
Bishop GM, Robinson SR. The amyloid hypothesis: let sleeping dogmas lie? Neurobiol Aging 2002; 23(6): 1101–5
Balin BJ, Gerard HC, Arking EJ, et al. Identification and localization of Chlamydia pneumoniae in the Alzheimer’s brain. Med Microbiol Immunol (Berl) 1998; 187(1): 23–42
Sigurdsson EM, Scholtzova H, Mehta PD, et al. Immunization with a nontoxic/nonfibrillar amyloid-β homologous peptide reduces Alzheimer’s disease-associated pathology in transgenic mice. Am J Pathol 2001; 159(2): 439–47
Bard F, Cannon C, Barbour R, et al. Peripherally administered antibodies against amyloid β-peptide enter the central nervous system and reduce pathology in a mouse model of Alzheimer disease. Nat Med 2000; 6(8): 916–9
Munch G, Robinson SR. Potential neurotoxic inflammatory responses to Aβ vaccination in humans. J Neural Transm 2002; 109(7β8): 1081–7
Robinson SR, Bishop GM, Lee H-G, et al. Lessons from the AN 1792 Alzheimer vaccine: lest we forget. Neurobiol Aging 2004; 25: 609–16
Robinson SR, Bishop GM, Munch G. Alzheimer vaccine: amyloid-β on trial. Bioessays 2003; 25(3): 283–8
Schenk D. Amyloid-β immunotherapy for Alzheimer’s disease: the end of the beginning. Nat Rev Neurosci 2002; 3(10): 824–8
Hyman BT, Smith C, Buldyrev I, et al. Autoantibodies to amyloid-β and Alzheimer’s disease. Ann Neurol 2001; 49(6): 808–10
Du Y, Dodel R, Hampel H, et al. Reduced levels of amyloid β-peptide antibody in Alzheimer disease. Neurology 2001; 57(5): 801–5
Weksler ME, Relkin N, Turkenich R, et al. Patients with Alzheimer disease have lower levels of serum anti-amyloid peptide antibodies than healthy elderly individuals. Exp Gerontol 2002; 37(7): 943–8
Hock C, Konietzko U, Papassotiropoulos A, et al. Generation of antibodies specific for β-amyloid by vaccination of patients with Alzheimer disease. Nat Med 2002; 8(11): 1270–5
Monsonego A, Zota V, Kami A, et al. Increased T cell reactivity to amyloid β protein in older humans and patients with Alzheimer disease. J Clin Invest 2003; 112(3): 415–22
Nath A, Hall E, Tuzova M, et al. Autoantibodies to amyloid β-peptide (Aβ) are increased in Alzheimer’s disease patients and Aβ antibodies can enhance Aβ neurotoxicity: implications for disease pathogenesis and vaccine development. Neuromolecular Med 2003; 3(1): 29–39
McGreevy K, Wang DS, Dickson DW. Is lowering amyloid levels a therapeutic target for Alzheimer’s disease: insights from macrophage-mediated resorption of amyloid in cortical infarcts. In: Iqbal K, Winblad B, editors. Alzheimer’s disease and related disorders: research advances. Bucharest: ‘Ana Asian’ International Academy of Aging, 2003: 569–80
Cai H, Wang Y, McCarthy D, et al. BACE1 is the major β-secretase for generation of Aβ peptides by neurons. Nat Neurosci 2001; 4(3): 233–4
Roberds SL, Anderson J, Basi G, et al. BACE knockout mice are healthy despite lacking the primary β-secretase activity in brain: implications for Alzheimer’s disease therapeutics. Hum Mol Genet 2001; 10(12): 1317–24
Luo Y, Bolon B, Kahn S, et al. Mice deficient in BACE1, the Alzheimer’s β-secretase, have normal phenotype and abolished β-amyloid generation. Nat Neurosci 2001; 4(3): 231–2
Yu H, Saura CA, Choi SY, et al. APP processing and synaptic plasticity in presenilin-1 conditional knockout mice. Neuron 2001; 31(5): 713–26
Feng R, Rampon C, Tang YP, et al. Deficient neurogenesis in forebrain-specific presenilin-1 knockout mice is associated with reduced clearance of hippocampal memory traces. Neuron 2001; 32(5): 911–26
Ohno M, Sametsky EA, Younkin LH, et al. BACE1 deficiency rescues memory deficits and cholinergic dysfunction in a mouse model of Alzheimer’s disease. Neuron 2004; 41(1): 27–33
Gong Y, Chang L, Viola KL, et al. Alzheimer’s disease-affected brain: presence of oligomeric Aβ ligands (ADDLs) suggests a molecular basis for reversible memory loss. Proc Natl Acad Sci U S A 2003; 100(18): 10417–22
Kayed R, Head E, Thompson JL, et al. Common structure of soluble amyloid oligomers implies common mechanism of pathogenesis. Science 2003; 300(5618): 486–9
Acknowledgements
We wish to thank the Department of Psychology, Monash University, Australia, for providing resources that assisted in the preparation of this review.
The authors have no conflicts of interest that are directly relevant to the content of this review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bishop, G.M., Robinson, S.R. Physiological Roles of Amyloid-β and Implications for its Removal in Alzheimer’s Disease. Drugs Aging 21, 621–630 (2004). https://doi.org/10.2165/00002512-200421100-00001
Published:
Issue Date:
DOI: https://doi.org/10.2165/00002512-200421100-00001