Abstract
Background: Since the 1970s, studies have examined potential risk factors associated with adverse drug reactions (ADRs) in a variety of settings. However, no pharmacoepidemiological study exists that incorporates clinical and laboratory parameters in a multiple regression model in order to consider predictors for ADRs.
Objectives: To characterize risk factors associated with ADRs in patients admitted to university hospital departments of internal medicine.
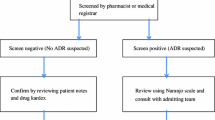
Design and Setting: Intensive pharmacovigilance was carried out in departments of internal medicine of two university hospitals. All admissions were followed prospectively for the occurrence of ADRs by members of a pharmacoepidemiological team consisting of physicians, pharmacologists and pharmacists. To identify patients at high risk for experiencing ADRs, patient histories and several clinical and laboratory data, determined at the time of admission, were taken into consideration. In addition to the drug prescribed, 40 parameters defined vital status at admission. These included temperature, heart rate, blood pressure (systolic-diastolic), body mass index, nicotine and alcohol use, and first laboratory test results after admission on nutrition status, inflammation, liver, kidney, pancreas or thyroid status, electrolytes, blood count and coagulation.
Results: 907 patients were observed during the study period. The mean age of the study population was 60 + 16 years. The median number of different drugs administered per patient during hospitalization was 9.6 + 7.7. In 345 patients, 592 ADRs were evaluated: 33.4% possible, 61.5% probable and 4.7% highly probable. Two ADR-related deaths were observed during the study period. Analysing ADR predictors, 17 of 40 parameters reached significance in univariate analysis, but only five in a multivariate binary regression model: raised temperature (odds ratio [OR] 1.609; 95% CI 1.133, 2.285), low erythrocyte levels (OR 0.386; 95% CI 0.194, 0.768), low thrombocyte levels (OR 0.788, 95% CI 0.627, 0.989), high number of drugs (OR 1.117; 95% CI 1.076, 1.159) and female sex (OR 1.562; 95% CI 0.785, 2.013) were independent predictors for ADRs.
Conclusion: For the patients investigated, of the large number of clinical data available only five independent factors predict ADR occurrence. Taking these results into account, physicians will be able to focus early on patients at risk for ADRs. To minimize ADR occurrence, ADR predictors should be integrated into the clinical pathway.




Similar content being viewed by others
References
Bates DW, Miller EB, Cullen DJ, et al. Patient risk factors for adverse drug events in hospitalized patients. ADE Prevention Study Group. Arch Intern Med 1999; 159(21): 2553–60
Bond CA, Raehl CL. Adverse drug reactions in United States hospitals. Pharmacotherapy 2006; 26(5): 601–8
Bowman L, Carlstedt BC, Hancock EF, et al. Adverse drug reaction (ADR) occurrence and evaluation in elderly inpatients. Pharmacoepidemiol Drug Saf 1996; 5(1): 9–18
Burgess CL, Holman CD, Satti AG, et al. Adverse drug reactions in older Australians 1981–2002. Med J Aust 2005; 182(6): 267–70
Caamaño F, Pedone C, Zuccalà G, et al. Socio-demographic factors related to the prevalence of adverse drug reaction at hospital admission in an elderly population. Arch Gerontol Geriatr 2005; 40(1): 45–52
Camargo AL, Cardoso Ferreira MB, Heineck I. Adverse drug reactions: a cohort study in internal medicine units at a university hospital. Eur J Clin Pharmacol 2006; 62(2): 143–9
Carbonin P, Pahor M, Bernabei R, et al. Is age an independent risk factor of adverse drug reactions in hospitalized medical patients? J Am Geriatr Soc 1991; 39(11): 1093–9
Domecq C, Naranjo CA, Ruiz I, et al. Sex-related variations in the frequency and characteristics of adverse drug reactions. Int J Clin Pharmacol Ther Toxicol 1980; 18(8): 362–6
Dormann H, Neubert A, Criegee-Rieck M, et al. Readmissions and adverse drug reactions in internal medicine: the economic impact. J Intern Med 2004; 255(6): 653–63
Drici MD, Clement N. Is gender a risk factor for adverse drug reactions? The example of drug-induced long QT syndrome. Drug Saf 2001; 24(8): 575–85
Edwards IR. The management of adverse drug reactions: from diagnosis to signal. Therapie 2001; 56(6): 727–33
Fattinger K, Roos M, Vergères P, et al. Epidemiology of drug exposure and adverse drug reactions in two Swiss departments of internal medicine. Br J Clin Pharmacol 2000; 49(2): 158–67
Gonzalez-Martin G, Yafiez CG, González-Contreras L, et al. Adverse drug reactions (ADRs) in patients with HIV infection: a prospective study. Int J Clin Pharmacol Ther 1999; 37(1): 34–40
Gray SL, Sagar M, Lestico MR, et al. Adverse drug events in hospitalized elderly. J Gerontol A Biol Sci Med Sci 1998; 53(1): M59–63
Hanlon JT, Pieper CF, Hajjar ER, et al. Incidence and predictors of all and preventable adverse drug reactions in frail elderly persons after hospital stay. J Gerontol A Biol Sci Med Sci 2006; 61(5): 511–5
Hoigné R, Sollberger J, Zoppi M, et al. Significance of age, sex, kidney function, atopy and number of prescriptions for the occurrence of adverse drug reactions, studied by multivariate statistical methods: results from the Comprehensive Hospital Drug Monitoring Berne (CHDMB) [in German]. Schweiz Med Wochenschr 1984; 114(49): 1854–7
Kando JC, Yonkers KA, Cole JO. Gender as a risk factor for adverse events to medications. Drugs 1995; 50(1): 1–6
Lazarou J, Pomeranz BH, Corey PN. Incidence of adverse drug reactions in hospitalized patients: a meta-analysis of prospective studies. JAMA 1998; 279(15): 1200–5
Levy M, Kewitz H, Altwein W, et al. Hospital admissions due to adverse drug reactions: a comparative study from Jerusalem and Berlin. Eur J Clin Pharmacol 1980; 17(1): 25–31
Levy M, Lipshitz M, Eliakim M. Hospital admissions due to adverse drug reactions. Am J Med Sci 1979; 277(1): 49–56
Mittmann N, Knowles SR, Gomez M, et al. Evaluation of the extent of under-reporting of serious adverse drug reactions: the case of toxic epidermal necrolysis. Drug Saf 2004; 27(7): 477–87
Onder G, Landi F, Delia Vedova C, et al. Moderate alcohol consumption and adverse drug reactions among older adults. Pharmacoepidemiol Drug Saf 2002; 11(5): 385–92
Onder G, Pedone C, Landi F, et al. Adverse drug reactions as cause of hospital admissions: results from the Italian Group of Pharmacoepidemiology in the Elderly (GIFA). J Am Geriatr Soc 2002; 50(12): 1962–8
Pirmohamed M, James S, Meakin S, et al. Adverse drug reactions as cause of admission to hospital: prospective analysis of 18 820 patients. BMJ 2004; 329(7456): 15–9
Rademaker M. Do women have more adverse drug reactions? Am J Clin Dermatol 2001; 2(6): 349–51
Ramesh M, Pandit J, Parthasarathi G. Adverse drug reactions in a south Indian hospital: their severity and cost involved. Pharmacoepidemiol Drug Saf 2003; 12(8): 687–92
Schneider JK, Mion LC, Frengley JD. Adverse drug reactions in an elderly outpatient population. Am J Hosp Pharm 1992; 49(1): 90–6
Tegeder I, Levy M, Muth-Selbach U, et al. Retrospective analysis of the frequency and recognition of adverse drug reactions by means of automatically recorded laboratory signals. Br J Clin Pharmacol 1999; 47(5): 557–64
Tran C, Knowles SR, Liu BA, et al. Gender differences in adverse drug reactions. J Clin Pharmacol 1998; 38(11): 1003–9
Trifiro G, Calogero G, Ippolito FM, et al. Adverse drag events in emergency department population: a prospective Italian study. Pharmacoepidemiol Drug Saf 2005; 14(5): 333–40
Tschepik W, Segal R, Sherrin TP, et al. Therapeutic riskassessment model for identifying patients with adverse drug reactions. Am J Hosp Pharm 1990; 47(2): 330–4
Vakil BJ, Kulkarni RD, Chabria NL, et al. Intense surveillance of adverse drag reactions: an analysis of 338 patients. J Clin Pharmacol 1975; 15(5–6): 435–41
van der Hooft CS, Sturkenboom MC, van Grootheest K, et al. Adverse drag reaction-related hospitalisations: a nationwide study in The Netherlands. Drug Saf 2006; 29(2): 161–8
Wu FL, Yang CC, Shen LJ, et al. Adverse drug reactions in a medical ward. J Formos Med Assoc 1996; 95(3): 241–6
Blomqvist P, Feltelius N, Löfberg R, et al. A 10-year survey of inflammatory bowel diseases-drug therapy, costs and adverse reactions. Aliment Pharmacol Ther 2001; 15(4): 475–81
Dormann H, Criegee-Rieck M, Neubert A, et al. Implementation of a computer-assisted monitoring system for the detection of adverse drag reactions in gastroenterology. Aliment Pharmacol Ther 2004; 19(3): 303–9
Dormann H, Krebs S, Muth-Selbach U, et al. Adverse drug reactions in patients with gastroenterological diseases: does age increase the risk? Aliment Pharmacol Ther 2001; 15(2): 171–80
Labenz J, Petersen KU, Rösch W, et al. A summary of Food and Drug Administration-reported adverse events and drug interactions occurring during therapy with omeprazole, lansoprazole and pantoprazole. Aliment Pharmacol Ther 2003; 17(8): 1015–9
Lancashire RJ, Cheng K, Langman MJ. Discrepancies between population-based data and adverse reaction reports in assessing drags as causes of acute pancreatitis. Aliment Pharmacol Ther 2003; 17(7): 887–93
Hallas J, Haghfelt T, Gram LF, et al. Drug related admissions to a cardiology department; frequency and avoidability. J Intern Med 1990; 228(4): 379–84
Bordet R, Gautier S, Le Louet H, et al. Analysis of the direct cost of adverse drag reactions in hospitalised patients. Eur J Clin Pharmacol 2001; 56(12): 935–41
Egger T, Dormann H, Ahne G, et al. Identification of adverse drug reactions in geriatric inpatients using a computerised drag database. Drags Aging 2003; 20(10): 769–76
Nebert DW, Jorge-Nebert L, Vesell ES. Pharmacogenomics and “individualized drug therapy”: high expectations and disappointing achievements. Am J Pharmacogenomics 2003; 3(6): 361–70
Nebert DW, Vesell ES. Advances in pharmacogenomics and individualized drug therapy: exciting challenges that lie ahead. Eur J Pharmacol 2004; 500(1–3): 267–80
G-DRG-System 2006. InEK Institute for the Hospital Remuneration System [online]. Available from URL: http://www.gdrg.de [Accessed 2008 Aug 4]
Dormann H, Neubert A, Ackermann A, et al. Model-Projekt OntoDrug: Befundpräsentation Arzneimittelnebenwirkung DMW 2006; 131(34/35):1888
WHO Collaboration Centre for Drug Statistics. Methodology guidelines for ATC classification 2006 [online]. Available from URL: http://www.whocc.no/atcvet/about_atcvet.html [Accessed 2008 Mar 17]
Edwards IR, Aronson JK. Adverse drug reactions: definitions, diagnosis, and management. Lancet 2000; 356(9237): 1255–9
Naranjo CA, Busto U, Sellers EM, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther 1981; 30(2): 239–45
National Cancer Institute. Common toxicity criteria. Bethesda (MD): Division of Cancer Treatment, National Cancer Institute, 1988
Aziz Z, Siang TC, Badarudin NS. Reporting of adverse drug reactions: predictors of under-reporting in Malaysia. Pharmacoepidemiol Drag Saf 2007; 16(2): 223–8
Cosentino M, Leoni O, Banfi F, et al. Attitudes to adverse drug reaction reporting by medical practitioners in a Northern Italian district. Pharmacol Res 1997; 35(2): 85–8
Hartmann K, Kuhn M, Gartmann J. Spontaneous reporting system for the assessment of new, unknown and rare undesirable effects of drags [in German]. Schweiz Med Wochenschr 1992; 122(38): 1409–13
Li Q, Zhang SM, Chen HT, et al. Awareness and attitudes of healthcare professionals in Wuhan, China to the reporting of adverse drug reactions. Chin Med J (Engl) 2004; 117(6): 856–61
Rehan HS, Vasudev K, Tripathi CD. Adverse drug reaction monitoring: knowledge, attitude and practices of medical students and prescribes. Natl Med J India 2002; 15(1): 24–6
Schumock GT, Thornton JP. Focusing on the preventability of adverse drug reactions [letter]. Hosp Pharm 1992; 27(6): 538
Giroux L, Smeesters C, Boury F, et al. Adriamycin and adriamycin-DNA nephrotoxicity in rats. Lab Invest 1984; 50(2): 190–6
Gurwitz JH, Avom J. Old age: is it a risk for adverse drug reactions? Agents Actions 1990; 29 Suppl.: 13–25
Zhang Q, Matsumura Y, Teratani T, et al. The application of an institutional clinical data warehouse to the assessment of adverse drag reactions (ADRs): evaluation of aminoglycoside and cephalosporin associated nephrotoxicity. Methods Inf Med 2007; 46(5): 516–22
Sanai T, Okuda S, Motomura K, et al. Effect of phosphate binders on the course of chronic renal failure in rats with focal glomeralar sclerosis. Nephron 1989; 51(4): 530–5
Zhao SZ, Reynolds MW, Lejkowith J, et al. A comparison of renal-related adverse drag reactions between rofecoxib and celecoxib, based on the World Health Organization/Uppsala Monitoring Centre safety database. Clin Ther 2001; 23(9): 1478–91
Acknowledgements
We would like to thank Ulrich Rothe (Head of the Pharmacy at the University of Regensburg) and Prof. Dr Jürgen Schölmerich (Director of the Medical Department I) for the opportunity to implement KLASSE at the Department of Internal Medicine of the University Hospital of Regensburg, in order to establish computerized intensive drug surveillance studies.
We also thank Prof. Dr Kay Brune Doerenkamp (Professor for Innovations in Animal and Consumer Protection at the Department of Experimental and Clinical Pharmacology and Toxicology at the University of Erlangen Nuremberg) and Prof. Micha Levy (the incumbent of the Wilfred P. and Rose J. Cohen Chair in Internal Medicine and the former Chairman of Medicine at Hadassah-Hebrew University School of Medicine) for the early discussions on this topic and for their comments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zopf, Y., Rabe, C., Neubert, A. et al. Risk Factors Associated with Adverse Drug Reactions Following Hospital Admission. Drug-Safety 31, 789–798 (2008). https://doi.org/10.2165/00002018-200831090-00007
Published:
Issue Date:
DOI: https://doi.org/10.2165/00002018-200831090-00007