Summary
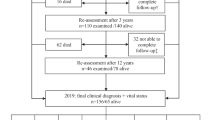
A recent study by the Parkinson’s Disease Research Group of the United Kingdom revealed higher mortality in patients with Parkinson’s disease who were treated with selegiline (deprenyl) compared with those who were not. In this article, the methodological limitations of the UK study are discussed.
Although several problems exist with this study, the mortality rate was correct, since the data were obtained from a verifiable source. The question then is whether or not the higher mortality rate can be ascribed to selegiline. It is difficult to find answers to this question in the study data, and we will have to wait for the study authors’ final report.
No other studies have reported higher mortality with selegiline. However, when prescribing selegiline in patients with early-stage Parkinson’s disease, it is important to provide them with all the available information so that treatment decisions can be made jointly. Meanwhile, the best indication for selegiline appears to be motor fluctuations in patients with moderately advanced Parkinson’s disease.
Similar content being viewed by others
References
Demarest K, Smith D, Azzaro A. The presence of the type A form of monoamine oxidase within nigrostriatal dopamine-containing neurons. J Pharmacol Exp Ther 1980; 215: 461–8
Damier P, Kastner A, Agid Y, et al. Does monoamine oxidase type B play a role in dopaminergic nerve cell death in Parkinson’s disease? Neurology 1996; 46: 1262–9
Levitt P, Pintar J, Breakfield X. Immunocytochemical demonstration of monoamine oxidase B in brain astrocytes and serotonergic neurons. Proc Natl Acad Sci USA 1982; 79: 6385–9
Chiba K, Trevor AJ, Castagnoli Jr N. Metabolism of the neurotoxic tertiary amine, MPTP, by brain monoamine oxidase. Biochem Biophys Res Commun 1984; 120: 574–8
Heikkila RE, Manzino L, Cabbat FS, et al. Protection against the dopaminergic neurotoxicity of 1-methyl-4-phenyl-1,2,5,6-tetrahydropyridine by monoamine oxidase inhibitors. Nature 1984; 311: 467–9
Parkinson Study Group. Effect of tocopherol and deprenyl on the progression of disability in early Parkinson’s disease. N Engl JMed 1993; 328: 176–83
Parkinson Study Group. Impact of deprenyl and tocopherol treatment on Parkinson’s disease in DATATOP subjects not requiring levodopa. Ann Neurol 1996; 39: 29–36
Olanow CW, Hauser RA, Gauger L, et al. The effect of deprenyl and levodopa on the progression of Parkinson’s disease. Ann Neurol 1995; 38: 771–7
Lees AJ. Parkinson’s Disease Research Group of the United Kingdom. Comparison of therapeutic effects and mortality data of levodopa and levodopa combined with selegiline in patients with early, mild Parkinson’s disease. BMJ 1995; 311: 1602–7
Parkinson’s Disease Research Group in the United Kingdom. Comparison of therapeutic effects of levodopa, levodopa and selegiline, and bromocriptine in patients with early, mild Parkinson’s disease: three-year interim report. BMJ 1993; 307: 469–72
Mäki-Ikola O, Kilkku O, Heinonen E. Effect of adding selegiline to levodopa in early, mild Parkinson’s disease. Other studies have not shown increased mortality [letter]. BMJ 1996; 312: 702
Anderson KE, Girdwood AC, Wilson JA. Stopping selegiline may lead to problems for patients [letter]. BMJ 1996; 312: 702
Olanow CW, Goodbold JH, Koller W. Patients taking selegiline may have received more levodopa than necessary [letter]. BMJ 1996; 312: 702–3
MacMahon DG, Bland R. Selegiline is effective and safe in early stages [letter]. BMJ 1996; 312: 703
Silva MT, Watts PM, Jenner P. Parkinson’s disease is rarely a primary cause of death [letter]. BMJ 1996; 312: 703
Yu PH, Lai CT, Boulton AA. Selegiline may be toxic in presence of increased dopamine concentrations [letter]. BMJ 1996; 312: 703–4
Gerlach M, Riederer P, Vogt H. ’On treatment’ rather than intention to treat analysis should have been used [letter). BMJ 1996; 312: 704
Jellinger KA. Causes of death need confirmation [letter]. BMJ 1996; 312: 704
Mena MA, Pardo B, Casarejos MJ, et al. Neurotoxicity of levodopa on catecholamine-rich neurons. Mov Disorders 1992; 7: 23–31
Spencer JPE, Jenner A, Aruoma OJ, et al. Intense oxidative DNA damage promoted by L-DOPA and its metabolites: implications for neurodegenerative disease. FEBS Lett 1994; 353: 246–50
Tanaka M, Sotomatsu A, Kanai H, et al. Combined histochemical and biochemical demonstration of nigral vulnerability to lipid peroxidation induced by DOPA and iron. Neurosci Lett 1992: 140: 42–6
Hoehn MM. Parkinsonism treated with levodopa: progression and mortality. J Neural Transm 1983; Suppl. 19: 253–64
Diamond SG, Markham CH, Hoehn MM, et al. Multi-center study of Parkinson mortality with early versus later dopa treatment. Ann Neurol 1987; 22: 8–12
Scigliano G, Musicco M, Soliveri P, et al. Mortality associated with early and late levodopa therapy initiation in Parkinson’s disease. Neurology 1990; 40: 265–9
Mytilineou C, Han SK, Cohen G. Toxic and protective effects of L-DOPA on mesencephalic cell cultures. J Neurochem 1993; 61: 1470–8
Han SK, Mytilineou C, Cohen G. L-DOPA up-regulates glutathione and protects mesencephalic cultures against oxidative stress. J Neurochem 1996; 66: 501–10
Duarte J, Almuina JV, Sevillano MD, et al. Atrial fibrillation induced by selegiline. Parkinsonism Related Disord 1996; 2: 125–6
Lees AJ, Shaw KM, Kohout LJ, et al. Deprenyl in Parkinson’s disease. Lancet 1977; II: 791–5
Rinne UK, Siirtola T, Sonninen V. L-Deprenyl treatment of on-off phenomena in Parkinson’s disease. J Neural Transm 1978; 43: 253–62
Schachter M, Marsden CD, Parkes JD. Deprenyl in the management of response fluctuations in patients with Parkinson’s disease on levodopa. J Neurol Neurosurg Psychiatry 1980; 43: 1016–21
Brodersen P, Philbert A, Gulliksen G, et al. The effect of L-deprenyl on on-off phenomena in Parkinson’s disease. Acta Neurol Scand 1985; 71: 494–7
Golbe LI, Lieberman AN, Muenter MD, et al. Deprenyl in the treatment of symptom fluctuations in advanced Parkinson’s disease. Clin Neuropharmacol 1988; 11: 45–55
Golbe LI. Long-term efficacy and safety of deprenyl (selegiline) in advanced Parkinson’s disease. Neurology 1989; 39: 1109–11
Cedarbaum JM, Silvestri M, Clark M, et al. L-deprenyl, levodopa pharmacokinetics, and response fluctuations in Parkinson’s disease. Clin Neuropharmacol 1990; 13: 29–35
Myllylä VV, Sotaniemi KA, Vuorinen JA, et al. Selegiline in de nova parkinsonian patients: the Finnish study. Mov Disord 1993; 1: S41–4
Tetrud JW, Langston JW. The effect of deprenyl (selegiline) on the natural history of Parkinson’s disease. Science 1989; 45: 519–22
Allain H, Pollak P, Neukirch HC, et al. Symptomatic effect of selegiline in de nova parkinsonian patients. Mov Disord 1993; 8: S36–40
Teräväinen H. Selegiline in Parkinson’s disease. Acta Neurol Scand 1990; 81: 333–6
Brannan T, Yahr M. Comparative study of selegiline plus L-dopa-carbidopa versus L-dopa-carbidopa alone in the treatment of Parkinson’s disease. Ann Neurol 1995; 37: 95–8
Rinne UK. Combination of a dopamine agonist, MAO-B inhibitor and levodopa: a new strategy in the treatment of early Parkinson’s disease. Acta Neurol Scand 1989; 126: 165–9
Baronti F, Davis TL, Boldry RC, et al. Deprenyl effects on levodopa pharmacodynamics, mood, and free radical scavenging. Neurology 1992; 42: 541–4
Elizan TS, Yahr MD, Moros DA, et al. Selegiline as an adjunct to conventional levodopa therapy in Parkinson’s disease: experience with this type B monoamine oxidase inhibitor in 200 patients. Arch Neurol 1989; 48: 1280–3
Yahr MD. [editorial]. Parkinsonism Related Disord 1996; 2: 123–4
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mizuno, Y., Kondo, T. Mortality Associated with Selegiline in Parkinson’s Disease. Drug-Safety 16, 289–294 (1997). https://doi.org/10.2165/00002018-199716050-00001
Published:
Issue Date:
DOI: https://doi.org/10.2165/00002018-199716050-00001