Summary
Abstract
Vardenafil (Levitra®) is a potent and highly selective oral phosphodiesterase type 5 (PDE5) inhibitor.
Vardenafil improved erectile function in men with mild to severe erectile dysfunction (ED) of varying aetiology in two randomised, double-blind, multicentre, fixed-dose studies of 12 or 26 weeks’ duration. Men receiving vardenafil 10 or 20mg had significantly greater improvements in International Index of Erectile Function (IIEF) questionnaire erectile function domain scores than placebo recipients. Moreover, improvements in penetration and maintenance of erection (assessed using IIEF or Sexual Encounter Profile [SEP] questions) were significantly greater with vardenafil 5–20mg than with placebo. Improvements in IIEF intercourse satisfaction and orgasmic function domain scores were significantly greater with vardenafil 10 or 20mg than with placebo and the proportion of patients with a positive response to a Global Assessment Question (GAQ) concerning improvement in erections after 12 or 26 weeks’ therapy was significantly higher with vardenafil 5–20mg than with placebo.
Vardenafil improved erectile function in men with ED associated with diabetes mellitus or ED following unilateral or bilateral nerve-sparing radical retropubic prostatectomy in two randomised, double-blind, multicentre, fixed-dose, 3-month studies. In both studies, improvements from baseline in the erectile function domain score of the IIEF and in positive responses to SEP questions were significantly greater with vardenafil 10 or 20mg than with placebo. In addition, a significantly higher proportion of vardenafil 10 or 20mg recipients than placebo recipients had positive GAQ responses.
Vardenafil was generally well tolerated in men with ED; treatment-emergent adverse events were of mild to moderate intensity and transient in nature. The most commonly reported adverse events (typical of those seen with PDE5 inhibitors) in vardenafil 5–20mg recipients included headache, flushing, rhinitis, dyspepsia and sinusitis. There were no reports of abnormal colour vision in men with ED taking vardenafil at clinically recommended doses (5–20mg).
Conclusion: Vardenafil is a potent and highly selective oral PDE5 inhibitor. It is effective and generally well tolerated in men with mild to severe ED of varying aetiology, as well as in men with ED associated with diabetes mellitus or ED after radical prostatectomy. Vardenafil should be considered a first-line treatment option in men with ED who are suitable candidates for oral PDE5 inhibitor therapy.
Pharmacodynamic Profile
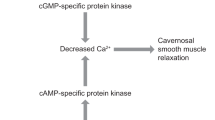
Vardenafil is a potent and highly selective inhibitor of phosphodiesterase type 5 (PDE5). The vardenafil concentration required to inhibit 50% of the activity (IC50) of PDE5 ranged from 0.11–0.7 nmol/L, depending on the assay used. Vardenafil, like sildenafil and tadalafil, showed highly selective inhibition of PDE5, with limited or no activity against other known PDE isoenzymes. Relative to PDE5, vardenafil (4-to 25-fold selectivity) and sildenafil (≈ 10-fold) demonstrated some selectivity for PDE6, whereas tadalafil (5-fold) had some activity against PDE11 A.
Both vardenafil and sildenafil inhibited cyclic guanosine monophosphate (cGMP) hydrolysis in a competitive manner in human corpus cavernosum smooth muscle cell extracts; IC50 values were ≈5-fold lower for vardenafil than for sildenafil. The accumulation of cGMP in human corpus cavernosum tissue induced by the nitric oxide donor sodium nitroprusside (SNP) was significantly augmented by vardenafil 3 nmol/L. This effect was also seen with sildenafil 30 nmol/L, but not with lower concentrations. Vardenafil significantly enhanced relaxation in human trabecular smooth muscle induced by SNP, acetylcholine or transmural electrical stimulation.
Oral vardenafil successfully induced penile erections in a conscious rabbit model; a dose-dependent erectile response was seen with oral vardenafil 1–10 mg/ kg. It has been suggested that the duration of effect of vardenafil may be much longer than its measured elimination half-life (t1/2). In a conscious rabbit model, a significant erectile response was obtained with SNP 7 hours after oral vardenafil administration (vardenafil t1/2 of 1.2 hours in rabbits).
Single-dose oral vardenafil 10–40mg significantly increased penile rigidity and tumescence during visual sexual stimulation in men with erectile dysfunction (ED) in two double-blind, randomised, placebo-controlled crossover studies. The mean total duration of erections with >60% rigidity was significantly longer with vardenafil 10–40mg than with placebo, and the mean total duration of erections with >80% rigidity was significantly longer with vardenafil 20 or 40mg than with placebo. Rigidity activity units and tumescence activity units were significantly higher with vardenafil 10–40mg than with placebo. The mean time to onset of the first erection was 26.8, 26.2 and 34.9 minutes with vardenafil 20 or 40mg or placebo.
Administering oral vardenafil 10mg 1–24 hours before sublingual nitroglycerin (glyceryl trinitrate) 0.4mg in healthy men did not potentiate the blood pressure (BP)-lowering effect of the latter drug (the coadministration of vardenafil and nitrates in men with ED is contraindicated). Some healthy men who received concomitant administration of vardenafil 10 or 20mg and α-blockers experienced hypotension.
Oral vardenafil 10 or 20mg did not impair the ability of men with stable coronary artery disease to exercise to a level similar to or greater than that associated with sexual intercourse in two randomised, double-blind, placebo-controlled, crossover studies. In one study, the time to ST-segment depression of ≥1mm was significantly longer with vardenafil 10mg than with placebo (381 vs 341 seconds). Coadministration of oral vardenafil 20mg in men with essential hypertension did not alter the antihypertensive effect of extended-release nifedipine 30 or 60 mg/day to a clinically significantly extent. In men with ED, small decreases in mean BP occurred with oral vardenafil 5–20mg between 11 minutes and 5 hours after treatment (reduction in mean systolic and diastolic BP of −1.4 to −6.6mm Hg and −2.0 to −4.8mm Hg). Changes in systolic and diastolic BP were −0.4 to +0.6mm Hg and −1.3 to +1.5mm Hg with placebo.
Pharmacokinetic Profile
Plasma concentrations rose rapidly after oral administration of single-dose vardenafil 10–40mg in men with ED. The mean maximum plasma vardenafil concentration (Cmax) increased in an almost dose-proportional manner and the median time to Cmax (tmax) was slightly longer with vardenafil 10mg (≈0.9 hours) than with vardenafil 20 or 40mg (≈0.7 hours). Vardenafil has a mean absolute bioavailability of 15% and a mean volume of distribution at steady state of 208L. Both vardenafil and its major circulating metabolite (M1) are highly protein bound. In healthy men, the median tmax was prolonged by 1 hour after consumption of a high-fat meal, compared with the fasting state.
Vardenafil is predominantly metabolised by cytochrome P450 (CYP) 3A4 and to a lesser extent by CYP3A5 and CYP2C. The Ml metabolite is pharmacologically active with an estimated efficacy contribution of ≈7%. In men with ED, the mean t1/2 of single-dose vardenafil 10–40mg ranged from 3.94 to 4.79 hours. Vardenafil has a total body clearance of 56 L/h and the mean renal clearance of vardenafil was 2.3 L/h. Approximately 91–95% of the administered vardenafil dose is excreted as metabolites in faeces and ≈2–6% is excreted as metabolites in the urine.
Elderly volunteers (aged >65 years) had higher Cmax and area under the plasma concentration-time curve (AUC) values (by 34% and 52%) and reduced hepatic clearance compared with younger volunteers (aged 18–45 years) after receiving single-dose vardenafil 40mg; t1/2 was slightly longer in elderly than in young volunteers (6.0 vs 4.8 hours). Renal clearance was ≈50% lower in men with mild to severe renal impairment than in healthy volunteers. In addition, the Cmax was lower (18.4 vs 31.8 μg/L) and the tmax (1.4 vs 0.8 hours) and t1/2 (56.1 vs 4.7 hours) were prolonged in men with severe renal impairment compared with healthy volunteers. Vardenafil clearance was reduced in men with mild to moderate hepatic impairment in proportion to the degree of impairment.
Alterations in the pharmacokinetics of vardenafil were seen with concomitant administration of potent CYP3A4 inhibitors such as ritonavir, indinavir and ketoconazole, and the bioavailability of a single dose of vardenafil 20mg was increased by 12% when it was coadministered with cimetidine (a nonspecific inhibitor of CYP isoenzymes). Concomitant administration of vardenafil 5mg and the CYP3A4 inhibitor erythromycin increased the AUC of vardenafil 4-fold and the Cmax of vardenafil 3-fold.
Therapeutic Efficacy
Oral vardenafil improved erectile function in men with mild to severe ED of varying aetiology in two randomised, double-blind, multicentre, fixed-dose studies of 12 or 26 weeks’ duration (n = 580–895 [intent-to-treat]). Men receiving vardenafil 10 or 20mg had significantly greater improvements in International Index of Erectile Function (IIEF) questionnaire erectile function domain scores than placebo recipients after 12 or 26 weeks’ therapy. In addition, in the 26-week study, the proportion of patients responding ‘yes’ to two Sexual Encounter Profile (SEP) questions (‘Were you able to insert your penis into your partner’s vagina’ [SEP-2] and ‘Did your erection last long enough for you to have successful intercourse’ [SEP-3]) was significantly greater with vardenafil 5–20mg than with placebo at both weeks 12 and 26. In the 12-week study, the improvement from baseline in IIEF question 3 (‘When you attempted sexual intercourse how often were you able to penetrate your partner?’) and IIEF question 4 (‘During sexual intercourse how often were you able to maintain your erection after you had penetrated your partner?’) was significantly greater with vardenafil 5–20mg than with placebo.
In both studies, improvements in IIEF intercourse satisfaction and orgasmic function domain scores were significantly greater with vardenafil 10 or 20mg than with placebo. Moreover, the proportion of patients with a positive response to the Global Assessment Question (GAQ) [‘Has the treatment you have taken over the past 4 weeks improved your erections?’] was significantly higher with vardenafil 5–20mg than with placebo after 12 and 26 weeks’ therapy (65–85% vs 28–39%). With regards to health-related quality of life (assessed using the Fugl-Meyer questionnaire), improvements from baseline in mean scores for the question relating to sex life satisfaction were significantly greater with vardenafil 5–20mg than with placebo. A dose-response was seen for improvement in IIEF domain scores and for GAQ.
In a randomised, double-blind, multicentre, fixed-dose, 52-week study in men with mild to severe ED of varying aetiology (n = 1000) who received vardenafil 10 or 20mg, the mean IIEF erectile function domain score increased from ≈13 at baseline to ≈23 after 52 weeks’ therapy; at 104 weeks the erectile function domain score was ≈25. Improvements in positive responses to SEP-2 and SEP-3 were also seen at week 104.
Pooled analyses of randomised, double-blind studies in men with ED who received vardenafil 5–20mg or placebo revealed that compared with placebo, vardenafil significantly improved erectile function irrespective of age, aetiology, baseline ED severity, the presence of comorbidities or whether or not patients were receiving concomitant antihypertensive therapy.
In a 12-week flexible-dose study in men with moderate to severe ED who had not responded to prior sildenafil therapy, the improvement from baseline in the IIEF erectile function domain score and SEP-2 and SEP-3 response rates were significantly greater with vardenafil than with placebo.
Vardenafil improved erectile function in men with ED associated with diabetes mellitus in a randomised, double-blind, multicentre, fixed-dose study (452 randomised patients). The improvements from baseline in the erectile function domain score of the IIEF and the proportion of positive SEP-2 and SEP-3 responses were significantly greater with vardenafil 10 or 20mg than with placebo. Significantly more vardenafil 10 or 20mg than placebo recipients had a positive GAQ response (57% and 72% vs 13%). Improvement with vardenafil occurred irrespective of glycaemic control at baseline. Following study completion, 340 men continued in a 3-month extension study in which placebo recipients switched to receive vardenafil 10 or 20mg. Erectile function domain scores improved in both men who had received vardenafil 10 or 20mg for the entire 6 months and in patients initially randomised to placebo who switched to vardenafil. Improvements were also seen in the percentage of patients able to maintain an erection during intercourse and the percentage of patients responding ‘yes’ to the GAQ
Vardenafil improved erectile function in men with ED following unilateral or bilateral nerve-sparing radical retropubic prostatectomy in a randomised, double-blind, multicentre, fixed-dose study (442 randomised patients). The improvements from baseline in the erectile function domain score of the IIEF and the proportion of positive SEP-2 and SEP-3 responses were significantly greater with vardenafil 10 or 20mg than with placebo. Positive GAQ response rates were significantly higher with vardenafil 10 or 20mg than with placebo (59% and 65% vs 13%).
Tolerability
Vardenafil was generally well tolerated in men with ED. Treatment-emergent adverse events were generally of mild to moderate intensity and transient in nature.
Across three large, well designed, placebo-controlled trials, the most commonly reported adverse events (typical of those seen with oral PDE5 inhibitors) in vardenafil 5–20mg recipients included headache (6.8–22%), flushing (5–13%) and rhinitis (2.8–17%). Dyspepsia (0.7–6.7%) and sinusitis (1–6%) were also reported. Serious adverse events occurred infrequently and were reported in 1–5% of vardenafil 5–20mg recipients and in 3–5% of placebo recipients. Adverse events resulting in treatment discontinuation occurred in 1–5% of vardenafil 5–20mg recipients and in 1% of placebo recipients.
Small decreases in mean BP occurred with vardenafil. In a pooled analysis of five randomised, double-blind trials, ECG abnormalities, oedema, syncope, angina pectoris, hypotension and myocardial ischaemia occurred with an incidence of 0% to <0.6% and were not dose related. Myocardial infarction occurred in one vardenafil and one placebo recipient. Another placebo recipient experienced a cerebrovascular accident and a third required cardiovascular surgery.
There were no reports of abnormal colour vision in men with ED taking vardenafil at clinically recommended doses (5–20mg). Transient vision changes such as mild haziness or an increase in the perceived brightness of light were reported infrequently (incidence of 1% in vardenafil and placebo recipients combined in one study).
Dosage and Administration
Vardenafil is approved in the US and the EU for the treatment of ED. The recommended dose of vardenafil in adult men is 10mg; however, the dose may be increased to the maximum recommended dose (20mg) or decreased to 5mg according to efficacy and tolerability. It is recommended that vardenafil not be taken more than once daily. Vardenafil should be taken orally prior to sexual intercourse. Vardenafil is not indicated for use in women or in individuals aged <18 years.
A starting dose of 5mg should be used in elderly men. EU prescribing information states that a starting dose of 5mg should also be considered in men with mild to moderate hepatic impairment and in men with severe renal impairment, and US prescribing information recommends a starting dose of 5mg in moderate hepatic impairment.
Vardenafil is contraindicated in the EU and not recommended in the US in men with severe hepatic impairment; end-stage renal failure needing dialysis; hypotension; a recent history of stroke or myocardial infarction; unstable angina pectoris; or known hereditary degenerative disorders of the retina (e.g. retinitis pig-mentosa). US prescribing information also states that the use of vardenafil is not recommended in patients with uncontrolled hypertension, a recent history of life-threatening arrhythmia or severe heart failure; vardenafil use should also be avoided in men with QT prolongation. Vardenafil should generally not be prescribed to men with ED for whom sexual activity is inadvisable.
Concomitant administration of vardenafil and nitrates or nitric oxide donors is contraindicated. In the EU, coadministration of vardenafil and potent CYP3A4 inhibitors such as ritonavir, indinavir, oral ketoconazole or oral itraconazole is contraindicated in men aged >75 years and should be avoided in younger men. In the US, reduced doses of vardenafil are recommended when it is administered in combination with ritonavir, indinavir, ketoconazole or itraconazole. Coadministration of vardenafil and grapefruit juice should be avoided and the vardenafil dose should not exceed 5mg when it is coadministered with the CYP3A4 inhibitor erythromycin. Coadministration of vardenafil and α-blockers is not recommended in the EU and contraindicated in the US.












Similar content being viewed by others
Notes
The use of tradenames is for product identification purposes only and does not imply endorsement.
References
Jardin A, Wagner G, Khoury S, et al. Recommendations of the 1st International Consultation on Erectile Dysfunction. Anderson KE, Becher E, Hendry B, et al. Erectile dysfunction. Plymouth, UK: Health Publication Ltd, 2000: 709–26
Melman A, Gingell JC. The epidemiology and pathophysiology of erectile dysfunction. J Urol 1999 Jan; 161(1): 5–11
Ralph D, McNicholas T, Erectile Dysfunction Alliance. UK management guidelines for erectile dysfunction. BMJ 2000 Aug 19; 321: 499–503
Benet AE, Melman A. The epidemiology of erectile dysfunction. Urol Clin North Am 1995 Nov; 22(4): 699–709
Nehra A, Pryor J, Althof SE, et al. Second international conference on management of erectile dysfunction: new perspectives on treatment. Int J Impot Res 2002; 14 Suppl. 1: S1–5
Vitezic D, Pelcic JM. Erectile dysfunction: oral pharmacotherapy options. Int J Clin Pharmacol Ther 2002 Sep; 40(9): 393–403
Lue TF. Drug therapy: erectile dysfunction. N Engl J Med 2000 Jun 15; 342(24): 1802–13
Nehra A, Barrett DM, Moreland RB. Pharmacotherapeutic advances in the treatment of erectile dysfunction. Mayo Clin Proc 1999 Jul; 74: 709–21
Rotella DP. Phosphodiesterase type 5 inhibitors: discovery and therapeutic utility. Drugs Future 2001; 26(2): 153–62
Melman A, Christ GJ. The hemodynamics of erection and the pharmacotherapies of erectile dysfunction. Heart Disease 2002; 4(4): 252–64
Giuliano F. Phosphodiesterase type 5 inhibition in erectile dysfunction: an overview. Eur Heart J Suppls 2002; 4 Suppl. H: H7–H12
Andersson KE, Hedlund P. New directions for erectile dysfunction therapies. Int J Impot Res 2002; 14 Suppl. 1: S82–92
Saenz de Tejada I, Angulo J, Cuevas P, et al. The phosphodiesterase inhibitory selectivity and the in vitro and in vivo potency of the new PDE5 inhibitor vardenafil. Int J Impot Res 2001 Oct; 13(5): 282–90
Saenz de Tejada I, Angulo Frutos J, Gaudo M, et al. Comparative selectivity profiles of tadalafil, sildenafil, and vardenafil using an in vitro phosphodiesterase activity assay [abstract no. P-081]. 5th Congress of the European Society for Sexual and Impotence Research; 2002 Dec 1–4; Hamburg
Gbekor E, Bethell S, Fawcett L, et al. Phosphodiesterase 5 inhibitor profiles against all human phosphodiesterase families: implications for use as pharmacological tools [abstract no. 967]. J Urol 2002 May; 167 (4 Suppl.): 246
Ballard SA, Gingell CJ, Tang K, et al. Effects of sildenafil on the relaxation of human corpus cavernosum tissue in vitro and on the activities of cyclic nucleotide phosphodiesterase isozymes. J Urol 1998 Jun; 159: 2164–71
Kim NN, Huang Y-H, Goldstein I, et al. Inhibition of cyclic GMP hydrolysis in human corpus cavernosum smooth muscle cells by vardenafil, a novel, selective phosphodiesterase type 5 inhibitor. Life Sci 2001 Sep 28; 69(19): 2249–56
Bischoff E, Niewoehner U, Haning H, et al. The oral efficacy of vardenafil hydrochloride for inducing penile erection in a conscious rabbit model. J Urol 2001 Apr; 165(4): 1316–8
Bischoff E, Schneider K. A conscious-rabbit model to study vardenafil hydrochloride and other agents that influence penile erection. Int J Impot Res 2001 Aug; 13(4): 230–5
Bischoff E, Mondritzki T, Niewoehner U, et al. Vardenafil improved erections in rabbits longer than expected from plasma half-life [abstract]. 5th Congress of the European Society for Sexual and Impotence Research; 2002 Dec 1–4; Hamburg
Choi S, O’Connell L, Min K, et al. Efficacy of vardenafil and sildenafil in facilitating penile erection in an animal model. J Androl 2002; 23(3): 332–7
Klotz T, Sachse R, Heidrich A, et al. Vardenafil increases penile rigidity and tumescence in erectile dysfunction patients: a RigiScan and pharmacokinetic study. World J Urol 2001 Feb; 19(1): 32–9
Stark S, Sachse R, Liedl T, et al. Vardenafil increases penile rigidity and tumescence in men with erectile dysfunction after a single oral dose. Eur Urol 2001 Aug; 40(2): 181–90
Padma-Nathan H, Kaufman J, Taylor T. Earliest time of onset of erections with vardenafil determined in an at-home setting [abstract no. 41]. Satellite Symposium at the 98th Annual Meeting of the American Urological Association; 2003 Apr 25; Chicago (IL)
Mazzu AL, Nicholls AJ, Zinny M. Vardenafil, a new selective PDE-5 inhibitor, interacts minimally with nitroglycerin in healthy middle-aged male subjects [abstract no. 19]. Int J Imp Res 2001; 13 Suppl. 5: S64
Young JM. Vardenafil. Expert Opin Investig Drugs 2002 Oct; 11(10): 1487–96
European Agency for Evaluation of Medicinal Products. Vardenafil: summary of product characteristics [online]. Available from URL: http://www.emea.eu.int [Accessed 2003 Mar 20]
Bayer Pharmaceuticals Corporation/GlaxoSmithKline. Levitra® (vardenafil HCl) tablets: prescribing information [online]. Available from URL: http://www.levitra.com [Accessed 2003 Aug 22]
Thadani U, Smith W, Nash S, et al. The effect of vardenafil, a potent and highly selective phosphodiesterase-5 inhibitor for the treatment of erectile dysfunction, on the cardiovascular response to exercise in patients with coronary artery disease. J Am Coll Cardiol 2002 Dec 4; 40(11): 2006–12
Thadani U, Chrysant S, Mazzu A, et al. The effect of vardenafil, a selective PDE5 inhibitor, on ischemic threshold, exercise tolerance, and circulatory responses during treadmill exercise in men with stable angina pectoris [abstract no. 1026-104]. J Am Coll Cardiol 2003 Mar 19; 41 (6 Suppl. A): 336
Rohde G, Jordan PJ. Influence of vardenafil on blood pressure and pharmacokinetics in hypertensive patients on nifedipine therapy [abstract no. 90]. Pharmacotherapy 2002 Mar; 22(3): 418
Hellstrom WJG, Gittelman M, Karlin G, et al. Vardenafil for treatment of men with erectile dysfunction: efficacy and safety in a randomized, double-blind, placebo-controlled trial. J Androl 2002; 23(6): 763–71
Goldstein I, Young JM, Fischer J, et al. Vardenafil, a new phosphodiesterase type 5 inhibitor, in the treatment of erectile dysfunction in men with diabetes: a multicenter double-blind placebo-controlled fixed-dose study. Diabetes Care 2003 Mar; 26(3): 777–83
Kloner RA, Mohan P, Norenberg C, et al. Cardiovascular safety of vardenafil, a potent, highly selective PDE-5 inhibitor in patients with erectile dysfunction; an analysis of five placebocontrolled clinical trials [abstract no. 304]. Pharmacotherapy 2002 Oct; 22(10): 1371
Bauer RJ, Rohde G, Taylor T, et al. A single oral dose of vardenafil had no acute effect on sperm motility in healthy males [abstract no. 2]. J Androl 2002; 23 Suppl. 1: 26
Klotz T, Bauer R-J, Rohde G, et al. Effect of renal impairment on the single-dose pharmacokinetics of vardenafil 20 mg, a selective PDE5 inhibitor, for the treatment of erectile dysfunction [abstract no. 92. Pharmacotherapy 2002; 22(3): 418
Steidle CP, Feldman RA, Lettieri J, et al. Pharmacokinetics of vardenafil (a new selective PDE5 inhibitor) in the elderly and subgroup data of efficacy and safety in elderly patients with erectile dysfunction [abstract no. P289]. J Am Geriatr Soc 2001 Apr; 49(4): S103
Rohde G, Wensing G, Sachse R. The pharmacokinetics of vardenafil, a new selective PDE5 inhibitor, are not affected by the antacid, Maalox 70™ [abstract no. 2]. Pharmacotherapy 2001 Oct; 21(10): 1254
Rohde G, Wensing G, Unger S, et al. The pharmacokinetics of vardenafil, a new selective PDE5 inhibitor, is minimally affected by coadministration with cimetidine or ranitidine [abstract no. 3]. Pharmacotherapy 2001 Oct; 21(10): 1254
Rohde G, Bauer R-J, Unger S, et al. Vardenafil, a new selective PDE5 inhibitor, produces no interaction with digoxin [abstract no. 4. Pharmacotherapy 2001 Oct; 21(10): 1254
Bauer RJ, Rohde G, Wensing G. The interaction of alcohol with vardenafil, a potent and highly selective phosphodiesterase type 5 inhibitor [abstract no. 91]. Pharmacotherapy 2002 Mar; 22(3): 418
Rajagopalan P, Mazzu A, Xia C, et al. Effect of high-fat breakfast and moderate-fat evening meal on the pharmacokinetics of vardenafil, an oral phosphodiesterase-5 inhibitor for the treatment of erectile dysfunction. J Clin Pharmacol 2003 Mar; 43(3): 260–7
Porst H, Rosen R, Padma-Nathan H, et al. The efficacy and tolerability of vardenafil, a new, oral, selective phosphodiesterase type 5 inhibitor, in patients with erectile dysfunction: the first at-home clinical trial. Int J Impot Res 2001 Aug; 13(4): 192–9
Saenz de Tejada I, Glina S, Becher E, et al. Vardenafil exhibits long-term efficacy and safety for up to 52 weeks [abstract no. P-090]. 5th Congress of the European Society for Sexual and Impotence Research; 2002 Dec 1–4; Hamburg
Porst H, Giuliano F, Liyanage N. 6 month study shows sustained efficacy of vardenafil in treating ED [abstract no. PS-4]. 5th Congress of the European Society for Sexual and Impotence Research; 2002 Dec 1–4; Hamburg
Sotomavor M, Rubio E, Tapia R, et al. Vardenafil improved erectile function in Mexico: a 3-month, open-label study [abstract no. CP5.08]. Int J Impot Res 2002 Sep; 14 Suppl. 3: S88
Hatzichristou D, Montorsi F, Porst H, et al. A flexible dose regimen of vardenafil for erectile dysfunction: a placebo-controlled trial [abstract no. 014]. Eur Urol Suppl 2003; 2(1): 176
Carson C, Hatzichristou D, Carrier S, et al. Vardenafil exhibits efficacy in men with erectile dysfunction unresponsive to prior sildenafil therapy: results of a phase III clinical trial —Patient Response with VardENafil in sildenafil nonresponders (PROVEN) [abstract no. 31]. Int J Impot Res 2003; 15 Suppl. 5: S175
Brock G, Taylor T, Seger M. Efficacy and tolerability of vardenafil in men with erectile dysfunction following radical prostatectomy [abstract no. 598]. Eur Urol Suppl 2002; 1(1): 152 and poster from the XVIIth Congress of the European Association of Urology; 2002 Feb 23–26; Birmingham
Vezina JG, Nehra A, Brock G, et al. Vardenafil improved patient satisfaction with erection hardness, orgasmic function, and sexual experience in men with erectile dysfunction following nerve-sparing radical prostatectomy [abstract no. 949]. J Urol 2003 Apr; 169 (4 Suppl.): 245
Donatucci C, Pommerville P, Niederberger C, et al. Vardenafil improved patient satisfaction with erection hardness, orgasmic function, and sexual experience in men with erectile dysfunction [abstract no. 950]. J Urol 2003 Apr; 169 (4 Suppl.): 246
Rosen RC, Riley A, Wagner G, et al. The International Index of Erectile Function (IIEF): a multidimensional scale for assessment of erectile dysfunction. Urology 1997 Jun; 49(6): 822–30
Fugl-Meyer AR, Lodnert G, Bränholm I-B, et al. On life satisfaction in male erectile dysfunction. Int J Impot Res 1997 Sep; 9(3): 141–8
Padma-Nathan H, Eardley I, Collins O, et al. Vardenafil restores normal functioning to men with erectile dysfunction [abstract no. 710]. J Urol 2002 Apr; 167 (4 Suppl.): 177
Donatucci C, Eardley I, McVary KT, et al. Vardenafil improves erectile function regardless of etiology or baseline severity in men with erectile dysfunction [abstract no. 715]. J Urol 2002 Apr; 167 (4 Suppl.): 178
Donatucci C, Karlin G, Goldfischer E, et al. Influence of age on the efficacy and safety of vardenafil, a novel phosphodiesterase-5 inhibitor, for the treatment of erectile dysfunction [abstract no. P293]. J Amer Ger Soc 2002; 50 Suppl. 4: S107
Goldfischer E, Eardley I, Segerson T. Vardenafil improves erectile function in men with significant comorbidities associated with erectile dysfunction [abstract no. 714]. J Urol 2002 Apr; 167 (4 Suppl.): 178
Hatzichristou DG, Pescatori ES. Current treatments and emerging therapeutic approaches in male erectile dysfunction. BJU Int 2001; 88 (Suppl. 3): 11–7
Porst H, Padma-Nathan H, Thibonnier M, et al. Efficacy and safety of vardenafil, a selective phosphodiesterase 5 inhibitor, in men with erectile dysfunction on antihypertensive therapy [abstract no. 599]. Eur Urol Suppl 2002 Jan; 1(1): 152
Valiquette L, Hellstrom W, Gittelman M, et al. Vardenafil provides reliable efficacy over time in men with erectile dysfunction [abstract no. CP5.07]. Int J Impot Res 2002 Sep; 14 Suppl. 3: S88
Young JM, Goldstein I, Taylor T. Long-term efficacy and safety of vardenafil in diabetic men with erectile dysfunction [abstract no. 1033]. Diabetologia 2002 Aug; 45 Suppl. 2: 334–5
Young J, Goldstein I, Fishcer J, et al. Vardenafil (Levitra®) improved satisfaction with erectile hardness, orgasmic function, and sexual experience in men with diabetes with erectile dysfunction [abstract no. 68-OR]. Diabetes 2003 Jun; 52 Suppl. 1: A16
Stief C, Porst H, Saenz de Tejada I, et al. Sustained efficacy and tolerability of vardenafil over two years in men with erectile dysfunction [abstract no. 100]. Prog Urol 2003; 13 Suppl. 2: 31
Montorsi F, Hellstrom WJG, Valiquette L, et al. Reliable efficacy over time of vardenafil, a potent, highly selective PDE-5 inhibitor in men with erectile dysfinction: a retrospective analysis of two pivotal phase III studies [abstract no. 98]. Prog Urol 2003; 13 Suppl. 2: 31
Goldstein I, Young JM, Fishcer JS, et al. Vardenafil (Levitra®) improved patient satisfaction with erectile hardness, orgasmic function, and sexual experience in men with erectile dysfunction and diabetes irrespective of level of glycemic control [abstract no. 917]. Diabetologia 2003; 46 Suppl. 2: A317
Hellstrom WJG, Gittelman M, Karlin G, et al. Sustained efficacy and tolerability of vardenafil, a highly potent selective phosphodiesterase type 5 inhibitor, in men with erectile dysfunction: results of a randomized, double-blind, 26-week placebo-controlled pivotal trial. Urology 2003 Apr; 61 Suppl. 4A: 8–14
Porst H, Young JM, Schmidt AC, et al. Efficacy and tolerability of vardenafil for treatment of erectile dysfunction in patient subgroups. Urology 2003 Sep; 62(3): 519–23; discussion 523-4
Dey J, Shepherd MD. Evaluation and treatment of erectile dysfunction in men with diabetes mellitus. Mayo Clin Proc 2002; 77(3): 276–82
Feldman HA, Goldstein I, Hatzichristou DG, et al. Impotence and its medical and psychosocial correlates: results of the Massachusetts Male Aging Study. J Urol 1994 Jan; 151(1): 54–61
Wespes E, Amar E, Hatzichristou D, et al. Guidelines on erectile dysfunction. Eur Urol 2002 Jan; 41(1): 1–5
Canadian Urological Association Guidelines Committee. Erectile dysfunction practice guidelines. Can J Urol 2002 Aug; 9(4): 1583–7
Costabile RA. Alprostadil for the treatment of erectile dysfunction. Expert Opin Invest Drugs 1999 Jun; 8(6): 877–84
Lyseng-Williamson KA, Wagstaff AJ. Management of erectile dysfunction: defining the role of sildenafil. Dis Manage Health Outcomes 2002; 10(7): 431–52
Fulgham PF, Cochran JS, Denman JL, et al. Disappointing initial results with transurethral alprostadil for erectile dysfunction in a urology practice setting. J Urol 1998 Dec; 160: 2041–6
Altwein JE, Keuler FU. Oral treatment of erectile dysfunction with apomorphine SL. Urol Int 2001; 67: 257–63
European Agency for Evaluation of Medicinal Products. Apomorphine: summary of product characteristics [online]. Available from URL: http://emea.eu.int [Accessed 2003 Mar 24]
Borer J, Armstrong P. Proceedings of the 99th meeting of the Food and Drug Administration Cardiovascular and Renal Drugs Advisory Committee: May 29th and 30th, 2003. Circulation 2003 Jun 17; 107(23): e9052
Pfizer Inc.. Viagra® (sildenafil citrate) prescribing information [online]. Available from URL: http://www/viagra.com/professional/pi/proPacklnsert.asp [Accessed 2003 Mar 24]
Jackson G, Betteridge J, Dean J, et al. A systematic approach to erectile dysfunction in the cardiovascular patient: a consensus statement —update 2002. Int J Clin Pract 2002; 56(9): 663–71
Cheitlin MD, Hutter AM, Brindis RG, et al. Use of sildenafil (Viagra) in patients with cardiovascular disease. Circulation 1999; 99: 168–77
European Agency for Evaluation of Medicinal Products. Tadalafil: summary of product characteristics [online]. Available from URL: http://www.emea.eu.int [Accessed 2003 Mar 24]
Langtry HD, Markham A. Sildenafil: a review of its use in erectile dysfunction. Drugs 1999 Jun; 57: 967–89
Porst H. IC351 (tadalafil, Cialis): update on clinical experience. Int J Impot Res 2002; 14 Suppl. 1: S57–64
Porst H, Rosen RC, Padma-Nathan H, et al. Tadalafil allows men with erectile dysfunction to have successful intercourse up to 36 hours postdose [abstract no. 709]. J Urol 2002 Apr; 167 Suppl.: 177
Author information
Authors and Affiliations
Additional information
Various sections of the manuscript reviewed by: K.-E. Andersson, Department of Clinical Pharmacology, Lund University Hospital, Lund, Sweden; J.D. Corbin, Department of Molecular Physiology and Biophysics, Vanderbilt University School of Medicine, Nashville, Tennessee, USA; W.J.G. Hellstrom, Department of Urology, Tulane University Medical Center, New Orleans, Louisiana, USA; M.K. Li, Department of Surgery, National University Hospital, Singapore; C.G. McMahon, Australian Centre for Sexual Health, St Luke’s Hospital, Sydney, New South Wales, Australia; J. Pryor, Lister Hospital, London, UK; D. Ralph, Institute of Urology, London, UK; E. Wespes, Department of Urology, CHU de Charleroi, Belgium; J.M. Young, South Orange County Urological Medical Associates, Laguna Woods, California, USA.
Data Selection
Sources: Medical literature published in any language since 1980 on vardenafil, identified using Medline and EMBASE, supplemented by AdisBase (a proprietary database of Adis International). Additional references were identified from the reference lists of published articles. Bibliographical information, including contributory unpublished data, was also requested from the company developing the drug.
Search strategy: Medline search terms were ‘vardenafil’. EMBASE search terms were ‘vardenafil’. AdisBase search terms were ‘vardenafil’. Searches were last updated 3 November 2003.
Selection: Studies in men with erectile dysfunction who received vardenafil. Inclusion of studies was based mainly on the methods section of the trials. When available, large, well controlled trials with appropriate statistical methodology were preferred. Relevant pharmacodynamic and pharmacokinetic data are also included.
Index terms: Vardenafil, erectile dysfunction, pharmacodynamics, pharmacokinetics, therapeutic use.
Rights and permissions
About this article
Cite this article
Keating, G.M., Scott, L.J. Vardenafil. Drugs 63, 2673–2702 (2003). https://doi.org/10.2165/00003495-200363230-00010
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-200363230-00010