Summary
Abstract
Pranlukast (Onon®, Azlaire®), is an orally administered, selective, competitive antagonist of the cysteinyl leukotrienes (LT) C4, LTD4 and LTE4. It is indicated for the prophylactic treatment of chronic bronchial asthma in paediatric and adult patients.
The efficacy of pranlukast 225mg twice daily in adults with mild to moderate asthma was demonstrated in double-blind, placebo- or azelastine-controlled studies of 4 or 8 weeks' duration. The drug at this dosage was superior to both comparators in improving mean attack scores and morning and/or evening peak expiratory flow rates, and decreasing the use of rescue bronchodilators (p < 0.05). In limited clinical studies, pranlukast 225mg twice daily appeared to be as effective as montelukast 10mg once daily and zafirlukast 40mg twice daily in adults with mild to moderate asthma. Tachyphylaxis was absent when the drug was administered for up to 4 years. In patients requiring high-dose inhaled corticosteroid therapy, pranlukast 225mg twice daily plus a halved dosage of inhaled corticosteroid was as effective as the original dosage of inhaled corticosteroid. Pranlukast was also effective in patients with mild to severe asthma in a clinical practice setting. In a double-blind trial, greater improvements in most outcome measures were observed with pranlukast than with oxatomide in children and adolescents with asthma.
In clinical trials, pranlukast was well tolerated in adult and paediatric patients with asthma, with an adverse event profile similar to that of placebo. Gastrointestinal events and hepatic function abnormalities were the most commonly reported adverse events. No clinically significant differences in adverse event profiles between pranlukast, zafirlukast or montelukast were shown in limited comparisons. Although Churg-Strauss syndrome has been noted in pranlukast recipients, a direct causal relationship is unlikely.
Conclusions: Pranlukast is a well tolerated and effective preventative treatment in adult and paediatric patients with persistent asthma of all severities. In some patients, pranlukast may be beneficial when added to low-dose inhaled corticosteroids; it may also be a viable alternative to increasing inhaled corticosteroid dosages. The efficacy of pranlukast relative to placebo has been confirmed; its efficacy relative to other therapy awaits further investigation. Nonetheless, pranlukast is a useful therapeutic option (with as-required short-acting β2-agonists), either as preventative monotherapy for the treatment of mild persistent asthma or in conjunction with inhaled corticosteroids in the management of moderate or severe persistent asthma.
Pharmacodynamic Profile
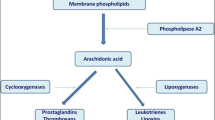
Pranlukast is a selective, competitive antagonist of the cysteinyl leukotrienes, leukotriene (LT) C4, LTD4 and LTE4, antagonising binding at the cysteinyl leukotriene type 1 (CysLTi) receptor. Like the other commercially-available CysLTi receptor antagonists montelukast and zafirlukast, pranlukast inhibits antigen-induced contraction of human bronchial smooth muscle in vitro.
In patients with asthma, oral pranlukast at clinically relevant dosages generally inhibited eosinophil activity and the presence of eosinophils in sputum (in the late asthma response), reduced exhaled nitric oxide levels, improved allergen-induced decreases in forced expiratory volume in 1 second and reduced the magnitude of airway hyperresponsiveness.
Pranlukast attenuated bronchoconstriction induced by exercise (dosage of 450 mg/day for 14 days), LTD4 (pranlukast 10-60mg as a single intravenous dose), aspirin (pranlukast 450 mg/day for 7 days), and bronchial hyperresponsiveness induced by methacholine and cold air (clinically relevant dosages), but did not ameliorate the effects of histamine, alcohol or propranolol.
Pharmacokinetic Profile
At least 12.5% of a dose of pranlukast is absorbed after oral administration, and maximum plasma concentrations are achieved within 2-6 hours when the drug is administered after food. Steady-state plasma concentrations of pranlukast are achieved within 7 days of twice-daily oral administration. Bioavailability is increased when the drug is administered before or after a meal; in addition, the bioavailability of pranlukast is greater after evening than after morning administration. Pranlukast is approximately 99% bound to plasma protein.
The mean plasma elimination half-life of oral pranlukast ranged from 3-9 hours after repeated doses in pharmacokinetic studies. The drug is cleared through metabolic transformation (primarily glucuronic acid conjugation). The majority of an oral dose is excreted via the faecal route, chiefly as the unchanged drug, within 72 hours of administration.
The pharmacokinetics of a single dose of oral pranlukast were generally similar in adults with asthma, elderly (aged >65 years) or young (aged 18-35 years) volunteers and, on a mg/kg basis, in children aged 8-11 years with moderate asthma.
Although the drug is metabolised by cytochrome P450 (CYP) enzymes in vitro, in a clinical setting pranlukast appears to have low potential for drug-drug interactions via this system. Urinary 6β-hydroxycortisol excretion (a surrogate marker of CYP3A4 activity) was unchanged with oral pranlukast 112.5-675mg twice daily in healthy volunteers. Prior treatment with clinically relevant doses of oral pranlukast (225mg twice daily) had no effect on the pharmacokinetics of intravenous aminophylline administered 4 hours after the final dose of pranlukast in healthy young adults.
Therapeutic Efficacy
In Adults: In patients with mild to moderate asthma receiving bronchodilators and/or inhaled corticosteroids, pranlukast 225mg twice daily for 4 or 8 weeks was more effective in improving asthma symptoms than twice-daily administration of placebo or azelastine 2mg. Mean attack scores, morning or evening peak expiratory flow rate (PEFR) and frequency of bronchodilator use improved to a significantly greater extent (p <0.05) with pranlukast than with placebo or azelastine at each 2-week timepoint in two randomised, double-blind, multicentre 4- or 8-week trials (n = 197 and 177).
In randomised, double-blind trials in patients with mild to moderate asthma, pranlukast 225mg twice daily showed similar efficacy to zafirlukast 40mg twice daily for 6 weeks in 298 patients and montelukast 10mg once daily for 4 weeks in 368 patients.
A subgroup analysis of preliminary data from European and US efficacy trials demonstrated that pranlukast 75–450mg twice daily for 12 weeks was effective in patients (n = 941–949) with mild to moderate asthma complicated by rhinitis.
In small (n = 10–70), nonblind 4- to 16-week studies, pranlukast 225mg twice daily was generally effective in patients with moderate or severe asthma receiving a concomitant inhaled corticosteroid (but not in those also receiving oral prednisolone) and in patients with severe asthma refractory to corticosteroids.
Patients with mild to severe asthma who were responders to pranlukast (>15% increase in baseline PEFR after 16 weeks' treatment) have been effectively treated with pranlukast for up to 4 years. Patients receiving pranlukast had a significantly greater increase in percentage of predicted PEFR than patients receiving conventional therapy over the 4-year period.
Pranlukast allows for reduced inhaled corticosteroid dosages without deterioration in lung function in patients requiring high-dose inhaled corticosteroids. In a randomised, double-blind, placebo-controlled, 6-week trial in 79 patients with stable asthma who had been receiving inhaled beclomethasone ≥1500 µg/day, outcome measures did not change significantly from baseline values when inhaled corticosteroid doses were halved in patients receiving pranlukast 225mg twice daily. In contrast, a significant deterioration in outcome measures was demonstrated by patients receiving placebo. Similar results were observed in 8- and 16-week randomised trials.
Health-related quality of life (HRQOL) improved in adult patients with asthma receiving pranlukast 225–450mg twice daily in a placebo-controlled 12-week trial or nonblind 4- to 12-week trials.
In Children: Pranlukast was at least as effective as the H1 antihistamine oxatomide in a randomised, double-blind 4-week trial in 214 patients aged >1 year with mild to severe paediatric asthma. Pranlukast ≈7 mg/kg/day had a significantly better final global improvement rating than oxatomide ≈1 mg/kg/day (71.4% vs 37.2%) in both per-protocol and intention-to-treat analyses and was superior for several, but not all, other parameters.
In longer nonblind studies, treatment with pranlukast dry syrup for up to 24 months improved asthma control compared with baseline values.
Pranlukast dry syrup improved overall HRQOL, physical domain and emotional domain scores from baseline in children with mild to moderate asthma aged <4 years (n = 20; mean age 2.0 years) and ≥4 years (n = 36; mean age 6.6 years) in a nonblind, 4-week trial.
In Clinical Practice: Pranlukast was effective in the treatment of mild to severe asthma symptoms in the clinical practice setting, as shown by a large (completed by >2500 patients), nonblind, population-based study without specific inclusion and exclusion criteria. The efficacy of pranlukast was unaffected by concomitant use of inhaled corticosteroids, β2-agonists and/or theophylline.
Pharmacoeconomic Analyses
Preliminary data from retrospective Japanese pharmacoeconomic studies suggest that pranlukast may be a cost-effective addition to asthma treatment. Relative to placebo, pranlukast reduced the cost required to obtain an effectiveness increase of one point regardless of whether direct or overall costs were considered in a 4-week study in 166 patients with mild to moderate asthma (currency year 1995).
The initiation of pranlukast 225mg twice daily increased annual medication costs compared with the year before in 30 patients with moderate or severe asthma who responded to pranlukast, despite decreased asthma symptoms and signs.
Tolerability
Pranlukast was well tolerated in clinical trials in adults or children with asthma; most adverse events were experienced by <1 % of patients. In pre-approval clinical trials of pranlukast conducted in Japan, 7.4% of 472 evaluated adult patients and 5.6% of 320 evaluated paediatric patients with asthma reported an adverse event with pranlukast treatment. Gastrointestinal adverse events (e.g. nausea, diarrhoea, abdominal pain/stomach discomfort) and hepatic function abnormalities were the most commonly reported adverse events. In comparative trials, the frequency of adverse events with pranlukast was significantly less than with azelastine in adult patients (12.2% vs 26.4%), and similar to that with oxatomide dry syrup in paediatric patients (9.4% vs 11.1 %). Moreover, the frequency, severity and nature of adverse events with pranlukast 225–450mg twice daily were similar to those with placebo in clinical trials in Europe and the US.
In long-term treatment (for up to 4 years) in adult patients, pranlukast was well tolerated and laboratory data did not change significantly.
Comparative clinical studies have shown no clinically significant differences in adverse event profiles between pranlukast and zafirlukast or montelukast.
Churg-Strauss syndrome, a form of vasculitis, has been noted in patients receiving pranlukast; however, it is unlikely that there is a direct causal relationship between the drug and the emergence of this condition.
Dosage and Administration
Oral pranlukast is indicated for the prophylactic treatment of chronic bronchial asthma in Japan and for the prophylaxis and treatment of chronic bronchial asthma, including the prevention of exercise-induced asthma, in Latin America. It is approved for use in a capsule formulation in adult patients in Japan and in those aged ≥12 years in Latin America, and as a dry syrup formulation in paediatric patients aged ≥1 year (in Japan) or ≥2 years (Latin America). The recommended dosage of pranlukast is 450 mg/day in adult patients and 7 mg/kg/day (increasing to a maximum of 10 mg/kg/day according to age and symptoms, but not exceeding 450 mg/day) in paediatric patients, divided into two doses. As food affects the bioavailability of pranlukast, the drug is administered after breakfast and dinner. The manufacturer's prescribing information states that pranlukast should be withdrawn in patients with suspected liver dysfunction, interstitial or eosinophilic pneumonia, leukopenia, thrombocytopenia or risk of anaphylaxis. Close monitoring is required when the drug is administered in patients undergoing dosage reduction or withdrawal of oral corticosteroids. Dosage adjustments are recommended in elderly patients, and caution is required when prescribing pranlukast during pregnancy or when it is coadministered with drugs that are predominantly metabolised by or inhibit the activity of CYP3A4 enzymes.















Similar content being viewed by others
Notes
Use of tradenames is for product identification purposes only and does not imply endorsement.
References
National Institutes of Health. Global initiative for asthma: global strategy for asthma management and prevention [online]. Available from URL: http://www.ginasthma.com [Accessed 2002 Oct 10]
Weersink EJM, Postma DS, Aabers R, et al. Early and late asthmatic reaction after allergen challenge. Respir Med 1994: 88: 103–14
O'Byrne PM. Leukotriene bronchoconstriction induced by allergen and exercise. Am J Respir Crit Care Med 2000; 161 (2): S68–72
Kumlin M. Measurement of leukotrienes in humans. Am J Respir Crit Care Med 2000; 161 (2): S102–6
Feldberg W, Kellaway CH. Liberation of histamine and formation of lysocithin-like substances by cobra venom. J Physiol 1938; 94: 187–226
Kellaway CH, Trethewie ER. The liberation of a slow reacting smooth-muscle-stimulating substance in anaphylaxis. Q J Exp Physiol 1940; 30: 121–45
Brocklehurst WE. The release of histamine and formation of a slow-reacting substance (SRS-A) during anaphylactic shock. J Physiol 1960; 151: 416–35
Samuelsson B. The discovery of the leukotrienes. Am J Respir Crit Care Med 2000; 161 (2): S2–6
Dunn CJ, Goa KL. Zafirlukast: an update of its pharmacology and therapeutic efficacy in asthma. Drugs 2001; 61(2): 285–315
Murphy RC, Hammarström S, Samuelsson B. Leukotriene C: a slow-reacting substance from murine mastocytoma cells. Proc Natl Acad Sci USA 1979 Sep; 76(9): 4275–9
Samuelsson B. Leukotrienes: mediators of immediate hypersensitivity reactions and inflammation. Science 1983 May 6; 220: 568–75
Lewis RA, Austen KF, Soberman RJ. Leukotrienes and other products of the 5-lipoxygenase pathway: biochemistry and relation to pathobiology in human diseases. N Engl J Med 1990 Sep 6; 323(10): 645–55
Lynch KR, O'Neill GP, Liu Q, et al. Characterization of the human cysteinyl leukotriene CysLT1 receptor. Nature 1999 Jun 24; 399: 789–93
Sarau HM, Ames RS, Chambers J, et al. Identification, molecular cloning, expression, and characterization of a cysteinyl leukotriene receptor. Mol Pharmacol 1999 Sep; 56(3): 657–63
Nothacker H-P, Wang Z, Zhu Y, et al. Molecular cloning and chararacterization of a second human cysteinyl leukotriene receptor: discovery of a subtype selective agonist. Mol Pharmacol 2000; 58(6): 1601–8
Heise CE, O'Dowd BF, Figueroa DJ, et al. Characterization of the human cysteinyl leukotriene 2 receptor. J Bio Chem 2000 Sep 29; 275(39): 30531–6
Leff AR. Discovery of leukotrienes and development of antileukotriene agents. Ann Allergy Asthma Immunol 2001; 86 Suppl.: 4–8
O'Byrne PM. Leukotrienes in the pathogenesis of asthma. Chest 1997 Feb; 111 Suppl.: 27S–34S
Dahlén B, Dahlén S-E. Leukotrienes as mediators of airway obstruction and inflammation in asthma. Clin Exp Allergy 1995; 25 Suppl. 2: 50–4
Dahlén S-E. Leukotriene receptors. Clin Rev Allergy Immunol 1999; 17: 179–91
Dahlén S-E. Pharmacological characterization of leukotriene receptors. Am J Respir Crit Care Med 2000; 161 (2): S41–5
Labat C, Ortiz JL, Norel X, et al. A second cysteinyl leukotriene receptor in human lung. J Pharmacol Exp Ther 1992; 263(2): 800–5
Walch L, Norel X, Back M, et al. Pharmacological evidence for a novel cysteinyl-leukotriene receptor subtype in human pulmonary artery smooth muscle. Br J Pharmacol 2002; 137: 1339–45
Zhang M-Q, Zwaagstra ME. Structural requirements for leukotriene CysLT1 receptor ligands. Curr Med Chem 1997; 4(4): 229–46
Barnes NC. Effects of antileukotrienes in the treatment of asthma. Am J Respir Crit Care Med 2000; 161: S73–6
Horwitz RJ, McGill KA, Busse WW. The role of leukotriene modifiers in the treatment of asthma. Am J Respir Crit Care Med 1998; 157 (5 Pt 1): 1363–71
Stokes JR, Busse WW. Antileukotriene therapy in asthma. Semin Respir Crit Care Med 1998; 19(6): 647–56
Leff AR. Regulation of leukotrienes in the management of asthma: biology and clinical therapy. Annu Rev Med 2001; 52: 1–14
Salvi SS, Krishna MT, Sampson AP, et al. The anti-inflammatory effects of leukotriene-modifying drugs and their use in asthma. Chest 2001; 119(5): 1533–46
Krawiec ME, Wenzel SE. Use of leukotriene antagonists in childhood asthma. Curr Opin Pediatr 1999 Dec; 11(6): 540–7
Claesson H-E, Dahlen S-E. Asthma and leukotrienes: antileukotrienes as novel anti-asthmatic drugs. J Intern Med 1999; 245(3): 205–27
Drazen JM, Israel E, O'Byrne PM. Treatment of asthma with drugs modifying the leukotriene pathway. N Engl J Med 1999: 340(3): 197–206
Nicosia S, Capra V, Rovati GE. Leukotrienes as mediators of asthma. Pulm Pharmacol Ther 2001; 14(1): 3–19
Smith LJ. Comparative efficacy of inhaled corticosteroids and antileukotriene drugs in asthma. Biodrugs 2001; 15(4): 239–49
Currie GP, Lipworth BJ. Bronchoprotective effects of leukotriene antagonists in asthma: a meta-analysis. Chest 2002; 122: 146–50
Nathan RA, Kemp JP. Efficacy of antileukotriene agents in asthma management. Ann Allergy Asthma Immunol 2001; 86 Suppl.: 9–17
Jarvis B, Markham A. Montelukast: a review of its therapeutic potential in persistent asthma. Drugs 2000; 59(4): 891–928
Dekhuijzen PNR, Koopmans PP. Pharmacokinetic profile of zafirlukast. Clin Pharmacokinet 2002; 41(2): 105–14
Adkins JC, Brogden RN. Zafirlukast: a review of its pharmacology and therapeutic potential in the management of asthma. Drugs 1998; 55(1): 121–44
Yamaguchi T, Kohrogi H, Honda I, et al. A novel leukotriene antagonist, ONO-1078, inhibits and reverses human bronchial contraction induced by leukotrienes C4 and D4 and antigen in vitro. Am Rev Respir Dis 1992 Oct; 146(4): 923–9
Fujiwara H, Kurihara N, Ohta, et al. Effect of a new leukotriene receptor antagonist, ONO-1078, on human bronchial smooth muscle in vitro. Prostaglandins Leukot Essent Fatty Acids 1993 Mar; 48(3): 241–6
Ravasi S, Capra V, Mezzetti M, et al. A kinetic binding study to evaluate the pharmacological profile of a specific leukotriene C4 binding site not coupled to contraction in human lung parenchyma. Mol Pharmacol 2000; 57: 1182–9
Yonetomi Y, Fujita M, Nakagawa N, et al. Preclinical pharmacology of pranlukast. Clinical and Experimental Allergy Reviews 2001; 1(3): 210–7
Ravasi S, Capra V, Panigalli T, et al. Pharmacological differences among CysLTi receptor antagonists with respect to LTC4 and LTD4 in human lung parenchyma. Biochem Pharmacol 2002 Apr 15; 63(8): 1537–46
Panettieri Jr RA, Tan EML, Ciocca V, et al. Effects of LTD4 on human airway smooth muscle cell proliferation, matrix expression, and contraction In Vitro: differential sensitivity to cysteinyl leukotriene receptor antagonists. Am J Respir Cell Mol Biol 1998 Sep; 19(3): 453–61
Panettieri RA, Leonard T, Luttmann MA, et al. Pranlukast, but not montelukast, inhibits LTD4-induced potentiation of human airway smooth muscle proliferation [abstract]. Eur Respir J 1997 Sep; 10 Suppl. 25: 438S
Nakagawa N, Fujita M, Yonetomi Y, et al. CysLT1 antagonism of pranlukast, montelukast and zafirlukast in the presence of serum albumin [in Japanese]. Jpn Pharmacol Ther 2002; 30(3): 191–5
McAlexander MA, Myers AC, Undem BJ. Inhibition of 5-lipoxygenase diminishes neurally evoked tachykinergic contraction of guinea pig isolated airway. J Pharmacol Exp Ther 1998 May; 285(2): 602–7
Nakagawa N, Obata T, Kobayashi T, et al. In vivo pharmacologic profile of ONO-1078: a potent, selective and orally active peptide leukotriene (LT) antagonist. Jpn J Pharmacol 1992 Nov; 60(3): 217–25
ObataT, Nambu F, Kitagawa T, et al. ONO-1078: an antagonist of leukotrienes. Adv Prostaglandin Thromboxane Leukot Res 1987; 17A: 540–3
Underwood DC, Bochnowicz S, Osborn RR, et al. The cysteinyl-leukotriene (CystLT) receptor antagonist, pranlukast, attenuates airway inflammation [abstract]. J Allergy Clin Immunol 1996 Jan; 97 (Pt 3): 289
Bochnowicz S, Underwood DC. Dose-dependent mediation of leukotriene D4-induced airway microvascular leakage and bronchoconstriction in the guinea pig. Prostaglandins Leukot Essent Fatty Acids 1995 Jun; 52(6): 403–11
Kurosawa M, Yodonawa S, Tsukagoshi H, et al. Inhibition by a novel peptide leukotriene receptor antagonist ONO-1078 of airway wall thickening and airway hyperresponsiveness to histamine induced by leukotriene C4 or leukotriene D4 in guinea-pigs. Clin Exp Allergy 1994 Oct; 24(10): 960–8
Nakagawa N, Obata T, Kobayashi T, et al. Effect of a peptide leukotriene receptor antagonist, ONO-1078, on guinea-pig models of asthma. Eur J Pharmacol 1993 Apr 28; 235 (2-3): 211–9
Abraham WM, Ahmed A, Cortes A, et al. The effects of a cysteinyl leukotriene antagonist (ONO-1078) on antigen-induced responses in allergic sheep. Prostaglandins Leukot Essent Fatty Acids 1993 Mar; 48(3): 233–9
Ishii A, Nakagawa T, Nambu F, et al. Inhibition of endogenous leukotriene-mediated lung anaphylaxis in guinea pigs by a novel receptor antagonist ONO-1078. Int Arch Allergy Appl Immunol 1990; 92(4): 404–7
Hojo M, Suzuki M, Maghni K, et al. Role of cysteinyl leukotrienes in CD4+ T cell-driven late allergic airway responses. J Pharmacol Exp Ther 2000 May; 293(2): 410–6
Salmon M, Walsh DA, Huang T-J, et al. Involvement of cysteinyl leukotrienes in airway smooth muscle cell DNA synthesis after repeated allergen exposure in sensitized Brown Norway rats. Br J Pharmacol 1999 Jul; 127(5): 1151–8
Obata T, Okada Y, Nakagawa N, et al. Inhibitory effect of a peptide leukotriene antagonist ONO-1078 on LTD4- and antigen-induced thromboxane B2 production in guinea pig lungs. Life Sci 1993; 52 (12): PL97–102
Obata T, Kobayashi T, Okada Y, et al. Effect of a peptide leukotriene antagonist, ONO-1078 on antigen-induced airway microvascular leakage in actively sensitized guinea pigs. Life Sci 1992; 51(20): 1577–83
Wei E-Q, Liu J-W, Zhang L-F, et al. Effect of ONO-1078, a leukotriene antagonist, on capsaicin- and substance P-induced bronchoconstriction and airway microvascular leakage in guinea pigs. Zhongguo Yao Li Xue Bao 1996 May; 17(3): 209–12
Underwood DC, Osborn RR, Newsholme SJ, et al. Persistent airway eosinophilia after leukotriene (LT) D4 administration in the guinea pig: modulation by the LTD4 receptor antagonist, pranlukast, or an interleukin-5 monoclonal antibody. Am J Respir Crit Care Med 1996 Oct; 154 (4 Pt 1): 850–7
Horiguchi T, Tachikawa S, Kasahara J, et al. Effects of pranlukast hydrate on serum eosinophil cationic protein levels in patients with adult bronchial asthma. Arzneimittel Forschung 1999 Jan; 49(1): 35–7
Ishioka S, Hozawa S, Haruta Y, et al. Pranlukast, a cysteinyl leukotriene antagonist, reduces serum eosinophil cationic protein levels in patients with asthma. Hiroshima J Med Sci 1999 Dec; 48(4): 105–10
Yoshida S, Ishizaki Y, Shoji T, et al. Effect of pranlukast on bronchial inflammation in patients with asthma. Clin Exp Allergy 2000 Jul; 30(7): 1008–14
Hozawa S, Haruta Y, Tamagawa K, et al. Effect of a LT receptor antagonist, ONO-1078, on bronchial hyperrespon-siveness and serum ECP in patients with asthma. Am J Respir Crit Care Med 1998 Mar; 157 Suppl. Pt 2: 413
Yoshikawa T, Hirata K, Shiraishi S, et al. Effect of pranlukast on airway eosinophilic inflammation in patients with asthma: a randomized double-blind, placebo-controlled trial [abstract]. Am J Respir Crit Care Med 1999 Mar; 159 Suppl. Pt 2: 641
Isogai S, Taniguchi M, Anzai K, et al. Effects of peptido-leukotriene receptor antagonist, pranlukast on eosinophil counts in peripheral blood and sputum in patients with chronic asthma [abstract]. Am J Respir Crit Care Med 1998 Mar; 157 Suppl. Pt 2: 412
Obase Y, Shimoda T, Tomari SY, et al. Effects of pranlukast on chemical mediators in induced sputum on provocation tests in atopic and aspirin-intolerant asthmatic patients. Chest 2002 Jan; 121(1): 143–50
Yamauchi K, Tanifuji Y, Pan LH, et al. Effects of pranlukast, a leukotriene receptor antagonist, on airway inflammation in mild asthmatics. J Asthma 2001 Feb; 38(1): 51–7
Nakamura Y, Hoshino M, Sim JJ, etal. Effect of the leukotriene receptor antagonist pranlukast on cellular infiltration in the bronchial mucosa of patients with asthma. Thorax 1998 Oct: 53(10): 835–41
Kobayashi H, Takahashi Y, Mitsufuji H, et al. Decreased exhaled nitric oxide in mild persistent asthma patients treated with a leukotriene receptor antagonist, pranlukast. Jpn J Physiol 1999 Dec; 49(6): 541–4
Hamilton A, Faiferman I, Stober P, et al. Pranlukast, a cysteinyl leukotriene receptor antagonist, attenuates allergen-induced early- and late-phase bronchoconstriction and airway hyper-responsiveness in asthmatic subjects. J Allergy Clin Immunol 1998 Aug; 102(2): 177–83
Taniguchi Y, Tamura G, Honma M, et al. The effect of an oral leukotriene antagonist, ONO-1078, on allergen-induced immediate bronchoconstriction in asthmatic subjects. J Allergy Clin Immunol 1993 Oct; 92(4): 507–12
Okudaira H. Challenge studies of a leukotriene receptor antagonist. Chest 1997 Feb; 111 Suppl.: 46S–51S
Obase Y, Shimoda T, Tomari S, et al. Effects of pranlukast on aspirin-induced bronchoconstriction: differences in chemical mediators between aspirin-intolerant and tolerant asthmatic patients. Ann Allergy Asthma Immunol 2001 Jul; 87(1): 74–9
Suguro H, Majima T, Hattori T, et al. Effect of a leukotriene antagonist, pranlukast hydrate, on exercise-induced asthma for 14 days [in Japanese]. Arerugi 2000 Aug; 49(8): 646–52
Smith LJ, Jorkasky DK, Carr A, et al. Intravenous pranlukast (Ultair) inhibits LTD4-induced bronchoconstriction in patients with asthma [abstract]. J Allergy Clin Immunol 1997 Jan; 99 (Pt 2): 328
Ishioka S, Hozawa S, Haruta Y, et al. Effects of pranlukast, a cysteinyl leukotriene antagonist, on bronchial responsiveness to methacholine in aspirin-intolerant asthmatics treated with corticosteroids. Hiroshima J Med Sci 2000 Jun; 49(2): 105–8
Yoshida S, Sakamoto H, Ishizaki Y, et al. Efficacy of leukotriene receptor antagonist in bronchial hyperresponsiveness and hypersensitivity to analgesic in aspirin-intolerant asthma. Clin Exp Allergy 2000 Jan; 30(1): 64–70
Fujimura M, Sakamoto S, Kamio Y, et al. Effect of a leukotriene antagonist, ONO-1078, on bronchial hyperresponsiveness in patients with asthma. Respir Med 1993 Feb; 87(2): 133–8
Strek ME, Sedy J, Solway J, et al. Pranlukast (Ultair) inhibits cold air-induced bronchoconstriction in patients with asthma [abstract]. J Allergy Clin Immunol 1997 Jan; 99 (Pt 2): 329
Shimoda TS, Asai SA, Kohno SK. Inhibitory effects of antiallergic drugs in alcohol-induced asthma [abstract]. Allergy Clin Immunol Int 2000; Suppl. 2: 126–7
Myou S, Fujimura M, Kamio Y, et al. Effect of a cysteinyl leukotriene antagonist, pranlukast hydrate, on acetaldehyde-induced bronchoconstriction in asthmatic patients. Pros-taglandins Leukot Essent Fatty Acids 2001 Jul; 65(1): 41–4
Fujimura M, Abo M, Kamio Y, et al. Effect of leukotriene and thromboxane antagonist on propranolol-induced bronchoconstriction. Am J Respir Crit Care Med 1999; 160: 2100–3
Ohmichi M, Tanaka H, Tanaka S, et al. Sputum eosinophil cationic protein, interleukin-8 and urinary eicosanoids in asthmatic patients with a long term treatment of pranlukast [abstract]. Eur Respir J 1998 Sep; 12 Suppl. 28: 361
Ramsay CF, van Kan CI, Sterk PJ, et al. Pranlukast improves spirometry and bronchial hyperresponsiveness in patients with mild asthma [abstract]. Am J Respir Crit Care Med 1998 Mar; 157 Suppl. Pt 2: 411
Obase Y, Shimoda T, Matsuo N, et al. Effects of cysteinylleukotriene receptor antagonist, thromboxane A2 receptor antagonist, and thromboxane A2 synthetase inhibitor on antigen-induced bronchoconstriction in patients with asthma. Chest 1998 Oct; 114(4): 1028–32
Hirata K, Kurihara N, Kamimori T, et al. Exercise-induced asthma and leukotriene receptor antagonist [in Japanese]. Rinsho Iyaku 1993; 9 Suppl. 1: 225–8
Christie PE, Tagari P, Ford-Hutchison AW, et al. Urinary leukotriene E4 concentrations increase after aspirin challenge in aspirin-sensitive asthmatic subjects. Am Rev Respir Dis 1994; 143: 1025–9
Shimoda T, Kohno S, Takao A, et al. Investigation of the mechanism of alcohol-induced bronchial asthma. J Allergy Clin Immunol 1996; 97(1): 74–84
Sano Y, Suzuki N, To M, et al. Effects of pranlukast hydrate on allergic airway inflammation in patients with mild to moderate asthma [abstract]. Allergy Clin Immunol Int 2000; Suppl. 2: 48
Brocks DR, Upward J, Hust R, et al. The pharmacokinetics of pranlukast in healthy young and elderly subjects. Int J Clin Pharmacol Ther 1996 Sep; 34(9): 375–9
Brocks DR, Upward JW, Georgiou P, et al. The single and multiple dose pharmacokinetics of pranlukast in healthy volunteers. Eur J Clin Pharmacol 1996; 51 (3–4): 303–8
Brocks DR, Upward J, Davy M, et al. Evening dosing is associated with higher plasma concentrations of pranlukast, a leukotriene receptor antagonist, in healthy male volunteers. Br J Clin Pharmacol 1997 Sep; 44(3): 289–91
Nakashima M, Uematsu T, Kanamaru M, et al. A phase I clinical study of a leukotriene C4, D4 and E4 receptor antagonist; ONO-1078 in healthy volunteers [in Japanese]. Rinsho Iyaku 1993; 9 Suppl. 1: 3–29
Kemp JP, Tinkelman D, Sublett J, et al. Pranlukast (Ultair) pharmacokinetics in children consistent with that of adults. Am J Respir Crit Care Med 1998 Mar; 157 Suppl. Pt 2: 411
Dennis M, Minthorn E, Stelman G, et al. Effect of food on the absorption of pranlukast in healthy subjects. Pharm Res 1994 Oct; 11 Suppl.: 430
Murdoch RD, Upward JW, Dennis M, et al. The safety and pharmacokinetics of IV aminophylline during co-administration with an oral LTD4 receptor antagonist pranlukast [abstract]. Eur Respir J 1995 Sep; 8 Suppl. 19: 48S
Grossman J, Faiferman I, Dubb JW, et al. Results of the first U.S. double-blind, placebo-controlled, multicenter clinical study in asthma with pranlukast, a novel leukotriene receptor antagonist. J Asthma 1997; 34(4): 321–8
PLM Latina. Azlaire capsulas [online]. Available from URL: http://www.plmlatina.com/sp/sp_alz.htm [Accessed 2002 Dec 19]
Barnes NC, de Jong B, Miyamoto T. Worldwide clinical experience with the first marketed leukotriene receptor antagonist. Chest 1997 Feb; 111 Suppl.: 52S–60S
Yoo SH, Park SH, Song JS, et al. Clinical effects of pranlukast, an oral leukotriene receptor antagonist, in mild-to-moderate asthma: a 4 week randomized multicentre controlled trial. Respirology 2001 Mar; 6(1): 15–21
The Japan Pharmaceutical Manufacturers Association. Japan Pharmaceutical Reference: Onon capsules [online]. Available from URL: http://www.jpma.or.jp/jpr/ [Accessed 2002 Oct 29]
Luan L, Sugiyama T, Takai S, et al. Purification and characterization of pranlukast hydrolase from rat liver microsomes: the hydrolase is identical to carboxylesterase pI 6. 2. Biol Pharm Bull 1997 Jan; 20(1): 71–5
Chervinsky P, Wenzel S, Kerwin E, et al. Pranlukast, a leukotriene receptor antagonist, improves quality of life in adult asthma patients [abstract]. Eur Respir J 1997 Sep; 10 Suppl. 25: 439S
Korenblat P, Wenzel S, Chervinsky P, et al. Reduction in health care utilization associated with pranlukast in adult patients with asthma [abstract]. Eur Respir J 1997 Sep; 10 Suppl. 25: 439
Spector SL, Bronsky E, Faiferman I, et al. Pranlukast (Ultair) decreases symptoms in patients with asthma complicated by rhinitis [abstract]. Am J Respir Crit Care Med 1998 Mar; 157 Suppl. Pt 2: A617
Silvers WS, Strek ME, Faiferman I, et al. Pranlukast (Ultair) decreases beta-2-agonist use in patients with asthma complicated by rhinitis [abstract]. Am J Respir Crit Care Med 1998 Mar; 157 Suppl. Pt 2: A617
Rosenthal R, Lang DM, Faiferman I, et al. Pranlukast (Ultair) improves lung function in patients with asthma complicated by rhinitis [abstract]. Am J Respir Crit Care Med 1998 Mar; 157 Suppl. Pt 2: A617
Tomari S, Shimoda T, Kawano T, et al. Effects of pranlukast, a cysteinyl leukotriene receptor 1 antagonist, combined with inhaled beclomethasone in patients with moderate or severe asthma. Ann Allergy Asthma Immunol 2001 Aug; 87(2): 156–61
Wada K, Minoguchi K, Kohno Y, et al. Effect of a leukotriene receptor antagonist, pranlukast hydrate, on airway inflammation and airway hyperresponsiveness in patients with moderate to severe asthma. Allergol Int 2000; 49(1): 63–8
Oosaki R, Mizushima Y, Kashii T, et al. Therapeutic effect of pranlukast, a selective cysteinyl leukotriene receptor antagonist, on bronchial asthma. Int Arch Allergy Immunol 1997 Sep: 114(1): 97–100
Miyamoto T, Nakajima S, Mano K, et al. Phase III comparative study of ICI 204,219 (zafirlukast, Accolate), a cysteinyl-leukotriene (LT) receptor antagonist, in adult patients with bronchial asthma: a double-blind comparative study with pranlukast hydrate. Rinsho Iyaku 2001; 17(12): 1683–723
Kohrogi H, Iwagoe H, Fujii K, et al. The role of cysteinyl leukotrienes in the pathogenesis of asthma: clinical study of leukotriene antagonist pranlukast for 1 year in moderate and severe asthma. Respirology 1999 Sep; 4(3): 319–23
Obase Y, Shimoda T, Tomari S, et al. Efficacy and safety of long-term treatment of asthmatic patients with pranlukast, a cysteinyl-leukotriene-receptor antagonist: four-year followup study. Ann Allergy Asthma Immunol 2001 Jul; 87(1): 43–7
Yokoyama A, Kohno N, Sakai K, et al. Effect of pranlukast, a leukotriene receptor antagonist, in patients with severe asthma refractory to corticosteroids. J Asthma 1998; 35(1): 57–62
Tamaoki J, Kondo M, Sakai N, et al. Leukotriene antagonist prevents exacerbation of asthma during reduction of high-dose inhaled corticosteroid. The Tokyo Joshi-Idai Asthma Research Group. Am J Respir Crit Care Med 1997 Apr; 155(4): 1235–40
Mikawa H, Baba M, Nakashima M. Clinical usefulness of a leukotriene antagonist; pranlukast dry syrup on pediatric bronchial asthma in multi-center comparative double-blind clinical study with oxatomide dry syrup [in Japanese]. Rinsho Iyaku 1997; 13(2): 423–56
Miyamoto T, Takishima T, Makino S, et al. Utility of a leukotrienes C4, D4 and E4 antagonist; ONO-1078, on adult bronchial asthma in multi-center comparative double-blind study with azelastine hydrochloride [in Japanese]. Rinsho Iyaku 1993; 9 Suppl. 1: 71–107
Toyoshima K, Kameda M, Mikawa H, et al. Efficacy and safety of a long-term (12 weeks) use of a leukotriene C4, D4 and E4 antagonist, ONO-1078 dry syrup, in asthmatic children [in Japanese]. Rinsho Iyaku 1997; 13(4): 1041–71
Iikura Y, Saito H, Sakaguchi N, et al. Efficacy and safety of a long-term use of a leukotriene C4, D4 and E4 antagonist, ONO-1078 dry syrup, in pediatric bronchial asthma [in Japanese]. Rinsho Iyaku 1997; 13(4): 1015–39
Tomita K, Hashimoto K, Matsumoto S, et al. Pranlukast allows reduction of inhaled steroid dose without deterioration in lung function in adult asthmatics [in Japanese]. Jpn J Allergol 1999 Apr; 48(4): 459–65
Tamura G, Iwasaki T, Shirato K, et al. Effect of pranlukast, a CysLTl receptor antagonist, on asthmatics treated with high doses of inhaled beclomethasone [abstract]. Allergy Clin Immunol Int 2000; Suppl. 2: 136
Takahashi H, Odagiri S, Yoshiike Y, et al. Pranlukast improves quality of life in patients treated with fluticasone propionate [abstract]. Allergy Clin Immunol Int 2000; Suppl. 2: 125
Terashima T, Yamaguchi K. Effect of pranlukast on health-related quality of life in patients with moderate persistent asthma [poster]. Am J Respir Crit Care Med 2002 Apr; 165 (Pt 2): 186
Yano T, Arikawa K, Saito S, et al. Clinical evaluation of the leukotriene receptor antagonist (Pranlukast) on health-related quality of life in patients with asthma [abstract]. Am J Respir Crit Care Med 1999 Mar; 159 Suppl. Pt 2: 641
Kondo N, Teramoto T, Inoue R, et al. Evaluation before and after pranlukast administration with the QOL questionnaire (revised version 2001) for pediatrie patients with bronchial asthma and their parents or caregivers [in Japanese]. Arerugi 2002 May; 51(5): 421–9
Calhoun WJ, Weisberg SC, Faiferman I, et al. Pranlukast (Ultair) is effective in improving asthma: results of a 12-week, multicenter, dose-range study [abstract]. J Allergy Clin Immunol 1997 Jan; 99 (Pt 2): 318
Miyamoto T, Takishima T, Tamura G, et al. Dose finding study of leukotriene antagonist; ONO-1078, on adult bronchial asthma [in Japanese]. Rinsho Iyaku 1993; 9 Suppl. 1: 53–70
Miyamoto T, Takishima T, Tamura G, et al. Efficacy and safety of leukotriene antagonist; ONO-1078, on adult bronchial asthma [in Japanese]. Rinsho Iyaku 1993; 9 Suppl. 1: 31–51
Barnes NC, Pujet J-C. Pranlukast, a novel leukotriene receptor antagonist: results of the first European, placebo controlled, multicentre clinical study in asthma. Thorax 1997 Jun; 52(6): 523–7
Mikawa H, Baba M. Efficacy and safety of a leukotriene antagonist; ONO-1078 dry syrup, on pediatrie bronchial asthma-dose finding study in a multi-center, open trial [in Japanese]. Rinsho Iyaku 1997; 13(2): 395–421
Tinkelman DG, Bucholtz GA, Kemp JA, et al. Evaluation of the safety and efficacy of multiple doses of azelastine to adult patients with bronchial asthma over time. Am Rev Respir Dis 1990 Mar; 141(3): 569–74
Chand N, Sofia RD. Azelastine — a novel in vivo inhibitor of leukotriene biosynthesis: a possible mechanism of action: a mini review. J Asthma 1995; 32(3): 227–34
Miyamoto T. Activity of pranlukast in asthma and allergic rhinitis [abstract]. Allergy 1996; 51 Suppl. 30: 116
Miyamoto T, Makino S, Kitamura S, et al. Cysteinyl leukotriene receptor 1 antagonist; MK-476, on adult bronchial asthma in multi-center comparative double-blind clinical study with pranlukast hydrate [in Japanese]. Rinsho Iyaku 2001; 17: 519–58
Kishimoto T, Okahara M, Chikamori K. Effectiveness of pranlukast for asthmatics [in Japanese]. Jpn J Chest Dis 1998: 57(11): 899–906
Nagano H, Hirose T, Yoshida M, et al. Clinical experience with long-term use of leukotriene antagonist, ONO-1078, in adult bronchial asthma [in Japanese]. Rinsho Iyaku 1993; 9 Suppl. 1: 185–207
Satake T, Takagi K, Suzuki R, et al. Study on efficacy and safety of the long-term administration of a leukotriene C4, D4 and E4 antagonist, ONO-1078, on adult bronchial asthma [in Japanese]. Rinsho Iyaku 1993; 9 Suppl. 1: 159–84
Hata D, Shiota M, Tamura Y, et al. The effectiveness of the long-term treatment with pranlukast in pediatric patients with mild to moderate asthma [in Japanese]. Arerugi 2002 May; 51(5): 400–10
Chiu C-P, Huang J-L, Lin T-Y, et al. Double-blind placebo-controlled study of oxatomide in the treatment of childhood asthma. Acta Paed Sin 1997 Jan 28; 38(1): 14–20
Marone G, Granata F, Spadaro G, et al. Anti-inflammatory effects of oxatomide. Invest Allergol Clin Immunol 1999 Jul 31; 9(4): 207–14
Tamura G, Inoue H, Chihara J, et al. Population-based open-label clinical effectiveness, assessment of the cysteinyl leukotriene receptor antagonist pranlukast. Allergology International 2000; 49(3): 189–94
Fujino S, Sato K, Imai H, et al. Cost effectiveness analysis of pranlukast (selective leukotriene C4, D4, E4 antagonist) in bronchial asthma therapy [in Japanese]. Rinsho Iyaku 1995; 11(11): 2303–23
Fujita K, Kohrogi H, Hirata N, et al. The economic impact of long-term therapy by leukotriene antagonist in asthma in Japan [abstract]. Am J Respir Crit Care Med 2001 Apr; 163 Suppl. Pt 2: A430
The Japan Pharmaceutical Manufacturers Association. Japan Pharmaceutical Reference: Onon dry syrup [online]. Available from URL: http://www.jpma.or.jp/jpr/ [Accessed 2002 Oct 29]
Ohnishi-Inoue Y, Mitsuya K, Horio T. Aspirin-sensitive urticaria: provocation with a leukotriene receptor antagonist. Br J Dermatol 1998 Mar; 138(3): 483–5
Wechsler ME, Pauwels R, Drazen JM. Leukotriene modifiers and Churg-Strauss syndrome: adverse effect or response to corticosteroid withdrawal? Drug Saf 1999 Oct; 21(4): 241–51
Wechsler ME, Drazen JM. Leukotriene modifiers and Churg-Strauss syndrome: an update. Am J Respir Crit Care Med 2000 Mar; 161 Suppl. Pt 2: 195
Stirling RG, Chung KF. Leukotriene antagonists and Churg-Strauss syndrome: the smoking gun. Thorax 1999 Oct; 54: 865–6
Hashimoto M, Fujishima T, Tanaka H, et al. Churg-Strauss syndrome after reduction of inhaled corticosteroid in a patient treated with pranlukast for asthma. Intern Med 2001 May; 40(5): 432–4
Kuwahara H, Arimoto T, Hara H. A case of pranlukast-induced Churg-Strauss syndrome [in Japanese]. Jpn J Chest Dis 2001: 60(11): 1041–5
Kinoshita M, Shiraishi T, Koga T, et al. Churg-Strauss syndrome after corticosteroid withdrawal in an asthmatic patient treated with pranlukast. J Allergy Clin Immunol 1999 Mar: 103 (3 Pt 1): 534–5
Baba K, Niwa S, Yagi T, et al. Churg-Strauss syndrome during corticosteroid tapering in a patient with bronchial asthma receiving pranlukast [in Japanese]. Arerugi 2000 Jun; 49(6): 512–5
Hayashi S, Furuya S, Imamura H. Fulminant eosinophilic endomyocarditis in an asthmatic patient treated with pranlukast after corticosteroid withdrawal. Heart 2001 Sep; 86 (3): e7
Schurman SJ, Alderman JM, Massanari M, et al. Tubulointer-stitial nephritis induced by the leukotriene receptor antagonist pranlukast. Chest 1998 Oct; 114(4): 1220–3
Takahashi N, Iwanaga T, Aizawa H, et al. Acute interstitial pneumonia induced by ONO-1078 (pranlukast), a leukotriene receptor antagonist. Intern Med 2001 Aug; 40(8): 791–4
PLM Latina. Azlaire granulado [online]. Available from URL: http://www.plmlatina.com/sp/sp_alz_cap.htm [Accessed 2002 Dec 19]
JCN Newswire. Ono announces Schering-Plough launched leukotriene antagonist pranlukast in Mexico [online]. Available from URL: http://www.japancorp.net/Article.Asp?Art_ID=3641 [Accessed 2002 Oct 11]
British Thoracic Society. Scottish Intercollegiate Guidelines Network. British guideline on the management of asthma. Thorax 2003; 58 Suppl. 1: 1–94
Ilowite JS, Spiegler PA. Leukotriene-modifying agents: new therapy in asthma. Clin Pulmonary Med 1999; 6(2): 87–94
Flood-Page P, Barnes NC. What are the alternatives to increasing inhaled corticosteroids for the long term control of asthma? Biodrugs 2001; 15(3): 185–98
Warner JO. The role of leukotriene receptor antagonists in the treatment of chronic asthma in childhood. Allergy 2001; 56 Suppl. 66: 22–9
Chung KF, Naya I. Compliance with an oral asthma medication: a pilot study using an electronic monitoring device. Respir Med 2000; 94: 852–8
Kelloway JS, Wyatt RA, Adlis SA. Comparison of patients' compliance with prescribed oral and inhaled asthma medications. Arch Intern Med 1994; 154: 1349–52
Busse W, Nelson H, Wolfe J, et al. Comparison of inhaled salmeterol and oral zafirlukast in patients with asthma. J Allergy Clin Immunol 1999; 103: 1075–80
Malmstrom K, Rodriguez-Gomez G, Guerra J, et al. Oral montelukast, inhaled beclomethasone and placebo for chronic asthma: a randomized, controlled trial. Ann Intern Med 1999: 130: 487–95
Yurdakul AS, Çalasir HC, Tunçtan B, et al. Comparison of second controller medications in addition to inhaled corticosteroid in patients with moderate asthma. Respir Med 2002; 96: 322–9
Terashima T, Amakawa K, Matsumaru A, et al. Correlation between cysteinyl leukotriene release from leukocytes and clinical response to a leukotriene inhibitor. Chest 2002; 122: 1566–70
Laitinen LA, Naya IP, Binks S, et al. Comparative efficacy of zafirlukast and low dose steroids in asthmatics on prn β2-agonists [abstract no. 2716]. Eur Respir J 1997; 10 Suppl. 25: 419S
Smith LJ. Newer asthma therapies. Ann Intern Med 1999; 130(6): 531–2
Suissa S, Ernst P. Use of anti-inflammatory therapy and asthma mortality in Japan. Eur Respir J 2003; 21: 101–4
Fujimura M, Tachibana H, Myou S. Comparison between effects of leukotriene and thromboxane A2 antagonist in asthma: a preliminary trial [abstract]. Eur Respir J 2000 Aug; 16 Suppl. 31: 522
Babu KS, Salvi SS. Aspirin and asthma. Chest 2000; 118(5): 1470–6
Author information
Authors and Affiliations
Corresponding author
Additional information
Various sections of the manuscript reviewed by: P.E. Korenblat, Washington University School of Medicine, St Louis, Missouri, USA; R. F. Lockey, Division of Allergy and Clinical Immunology, Department of Internal Medicine, University of South Florida, Tampa, Florida, USA; Y. Obase, Second Department of Internal Medicine, Nagasaki University School of Medicine, Nagasaki, Japan; G.E. Rovati, Department of Pharmacological Sciences, University of Milan, Milan, Italy; A.P. Sampson, Director, Respiratory Cell and Molecular Biology, University of Southampton School of Medicine, Southampton, UK; L.J. Smith, Northwestern University Medical School, Chicago, Illinois, USA; G. Tamura, First Department of Internal Medicine, Tohoku University School of Medicine, Sendai, Japan.
Data Selection
Sources: Medical literature published in any language since 1980 on pranlukast, identified using Medline and EMBASE, supplemented by AdisBase (a proprietary database of Adis International). Additional references were identified from the reference lists of published articles. Bibliographical information, including contributory unpublished data, was also requested from the company developing the drug.
Search strategy: Medline search terms were ‘pranlukast’ or ‘ONO-1078’ and ‘asthma’. EMBASE search terms were ‘pranlukast’ and ‘asthma’. AdisBase search terms were ‘pranlukast’ or ‘ONO-1078’ and ‘asthma’ and ‘pranlukast’ and ‘PK’ or ‘PD’. Searches were last updated 31 March 2003.
Selection: Studies in patients with asthma who received pranlukast. Inclusion of studies was based mainly on the methods section of the trials. When available, large, well controlled trials with appropriate statistical methodology were preferred. Relevant pharmacodynamic and pharmacokinetic data are also included.
Index terms: Asthma, pranlukast, leukotriene antagonists, pharmacodynamics, pharmacokinetics, therapeutic use, tolerability.
Rights and permissions
About this article
Cite this article
Keam, S.J., Lyseng-Williamson, K.A. & Goa, K.L. Pranlukast. Drugs 63, 991–1019 (2003). https://doi.org/10.2165/00003495-200363100-00005
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-200363100-00005