Abstract
Celecoxib, a selective cyclo-oxygenase-2 inhibitor, is a diaryl-substituted pyrazole derivative containing a sulfonamide substituent. Because of this structural component, celecoxib is contraindicated for use in patients who have demonstrated allergic reactions to sulfonamides. However, there is a lack of data demonstrating cross-reactivity among sulfonamide medications.
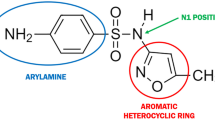
A sulfonamide is any compound with an SO2NH2 moiety. The major difference between sulfonamide antimicrobials and other sulfonamide-containing medications such as furosemide, thiazide diuretics and celecoxib, is that sulfonamide antimicrobials contain an aromatic amine group at the N4 position. This allows for division of the sulfonamides into 2 groups: aromatic amines (i.e., sulfonamide antimicrobials) and nonaromatic amines. In addition, sulfonamide antimicrobials contain a substituted ring at the N1-position; this group is not found with nonaromatic amine-containing sulfonamides.
Adverse reactions to sulfonamide antimicrobials include type I, or immunoglobulin (Ig) E-mediated reactions, hypersensitivity syndrome reactions, and severe skin reactions such as toxic epidermal necrolysis. The aromatic amine portion of the sulfonamide antimicrobial is considered to be critical in the development of latter 2 reactions. In susceptible individuals, the hydroxylamine metabolite is unable to be detoxified leading to a cascade of cytotoxic and immunological events that eventually results in the adverse reaction. Since celecoxib does not contain the aromatic amine, adverse reactions such as hypersensitivity syndrome reactions and toxic epidermal necrolysis would not be expected to occur at the same frequency as they do with sulfonamide antimicrobials. Similarly, for IgE-mediated reactions, the N1-substituent and not the sulphonamide moiety is important in determining specificity to antibodies. Celecoxib and other nonaromatic amine-containing sulfonamide medications do not contain the N1-substituent.
Cross-reactivity among the various sulfonamide-containing medications has also not been substantiated by published case reports. In fact, conflicting information exists in the literature. Reports showing lack of cross-reactivity balance the few case reports suggesting cross-reactivity.
Cross-reactivity between sulfonamide medications should be based on scientific data, including chemistry, metabolism, immune responses and clinical data. Based on the current information, there is no documentation for cross-reactivity between sulfonamide antimicrobials and other sulfonamide medications, such as celecoxib.





Similar content being viewed by others
References
Welbanks L, editor. CPS: compendium of pharmaceuticals and specialties, 35th ed. Ottawa: Canadian Pharmacists Association, 2000: 289
Hardman J, Limbird L, editors. Goodman & Gilman’s: the pharmacological basis of therapeutics. 9th ed. New York: McGraw-Hill, 1996: 1057
Dwenger C. ‘Sulpha’ hypersensitivity [letter]. Anaesthesia 2000; 55: 200
Venulet J, Ten Ham M. Methods for monitoring and documenting adverse drug reactions. Int J Clin Pharmacol Ther 1996; 34: 112–29
Pirmohamed M, Park B. The adverse effects of drugs. Hosp Med 1999; 60: 348–52
Management of drug hypersensitivity: a practice parameter. Ann Allergy Asthma Immunol 1999; 83 (Pt 3): 665–700
Cribb A, Lee B, Trepanier L, et al. Adverse reactions to sulphonamide and sulphonamide-trimethoprim antimicrobials: clinical syndromes and pathogenesis. Adverse Drug React Toxicol Rev 1996; 15: 9–50
Harle D, Baldo B, Wells J. Drugs as allergens: detection and combing site specificities of IgE antibodies to sulfamethoxazole. Mol Immunol 1988; 25: 1347–54
Gruchalla R, Sullivan T. Detection of human IgE to sulfamethoxazole by skin testing with sulfamethoxazoyl-poly-L-tyrosine. J Allergy Clin Immunol 1991; 88: 784–92
Meekins C, Sullivan T, Gruchalla R. Immunochemical analysis of sulfonamide drug allergy: identification of sulfamethoxazole-substituted human serum proteins. J Allergy Clin Immunol 1994; 94: 1017–24
Machtey I. Sudden death after intramuscular furosemide [letter]. Lancet 1968; II: 1301
Gould L, Reddy C, Zen B, et al. Life-threatening reaction to thiazides. NY State J Med 1980; 80: 1975–6
Heckbert S, Stryker W, Coltin K, et al. Serum sickness in children after antibiotic exposure: estimates of occurrence and morbidity in a Health Maintenance Organization population. Am J Epidemiol 1990; 132: 336–42
Roujeau JC, Stern R. Severe adverse cutaneous reactions to drugs. N Engl J Med 1994; 331(19): 1272–85
Gupta A, Eggo M, Uetrecht J, et al. Drug-induced hypothyroidism: The thyroid as a target organ in hypersensitivity reactions to anticonvulsants and sulfonamides. Clin Pharmacol Ther 1992; 51: 56–67
Cribb A, Spielberg S, Griffen G. N4-hydroxyation of sulfamethoxazole by cytochrome P450 of the CYP2C subfamily and microsomal reduction of its hydroxylamine metabolite in human and rat hepatic microsomes. Drug Metab Disp 1995; 23: 406–14
Cribb A, Miller M, Leeder J, et al. Reactions of the nitroso and hydroxylamine metabolites of sulfamethoxazole with reduced glutathione. Drug Metab Disp 1991; 19: 900–6
Spielberg S, Leeder J, Cribb A, et al. Is sulfamethoxazole hydroxylamine the proximal toxin for sulfamethoxazole toxicity? Eur J Clin Pharmacol 1989; 36Suppl.: A146
Rieder M, M Mask, Bird I. Production of tumour necrosis factor by cells exposed to sulphonamide reactive metabolites. Can J Physiol Pharmacol 1992; 70: 719–22
Rieder M, Sisson F, Bird I. Suppression of T lymphocyte proliferation by sulphonamide hydroxylamine. Int J Immunopharmacol 1992; 14: 1175–80
Naibitt D, Hough S, Gill H, et al. Cellular disposition of sulphamethoxazole and its metabolites: implications for hypersensitivity. Br J Pharmacol 1999; 126: 1393–407
Shear N, Spielberg S, Grant D, et al. Differences in Metabolism of Sulfonamides Predisposing to Idiosyncratic Toxicity. Ann Intern Med 1986; 105: 179–84
Shear N, Spielberg S. In vitro evaluation of a toxic metabolite of sulfadiazine. Can J Physiol Pharmacol 1985; 63: 1370–2
Rieder M, Uetrecht J, Shear N, et al. Diagnosis of sulfonamide hypersensitivity reactions by in-vitro ‘rechallenge’ with hydroxylamine metabolites. Ann Intern Med 1989; 110: 286–9
Shepherd G, Burton D. Administration of cephalosporin antibiotics to patients with a history to penicillin [abstract]. J Allergy Clin Immunol 1993; 91(1 Pt 2): 262
Pumphrey R, Davis S. Under-reporting of antibiotic anaphylaxis may put patients at risk. Lancet 1999; 353: 1157–8
Szezeklik A, Stevenson D. Aspirin-induced asthma: advances in pathogenesis and management. J Allergy Clin Immunol 1999; 104: 5–13
Shear N, Spielberg S. Anticonvulsant hypersensitivity syndrome, in vitro assessment of risk. J Clin Invest 1988; 82: 1826–32
Knowles S, Shapiro L, Shear N. Anticonvulsant hypersensitivity syndrome: incidence, prevention and management. Drug Saf 1999; 21: 489–501
Knowles S, Uetrecht J, Shear N. Idiosyncratic drug reactions: the reactive metabolite syndromes. Lancet 2000; 356: 1587–91
Bretza J. Thrombocytopenia due to sulfonamide cross-sensitivity. Wiscon Med J 1982; 81: 21–3
Hansbrough J, Wedner JH, Chaplin D. Anaphylaxis to intravenous furosemide. J Allergy Clin Immunol 1987; 80: 538–41
Ummenhofer B, Jawari D. Kreuzallergie zwischen Sulfonamid-Diuretika, Probenecid, Sulfamethoxazol und Sulfonylharnstoffen. Dtsch Med Wschr 1979; 104: 514–7
Conner C. Cross-sensitivity of sulfonamide antibacterials and antihypertensives. Drug Ther 1975; 5: 63–64
Goerz V, Ippen H, Meiers H. Sulfonamid-Uberempfindlichkeit: gekreuzte Reaktionen zwischen antibakteriellen Sulfonamiden und Diuretika. Dtsch Med Wschr 1964; 89: 1301–3
Sullivan T. Cross-reactions among furosemide, hydrochlorothiazide and sulfonamides. JAMA 1991; 265: 120–1
De Barrio M, Tornero P, Zubeldia J, et al. Fixed drug eruption induced by indapamide. Cross-reactivity with sulfonamides. Invest Allergol Clin Immunol 1998; 8: 253–5
Stricker B, Biriell C. Skin reactions and fever with indapamide. BMJ 1987; 295: 1313–4
Barrio M, Tornero P, Baeza M, et al. Cross-reactivity among para-amine group in sulfonamide-induced urticaria and fixed drug eruption [abstract]. J Allergy Clin Immunol 1991; 87(1 Pt 2): 230
Moseley V, Baroody N. Some observations on the use of acetazoleamide as an oral diuretic in various edmatous states and in uremia with hyperkaliemia. Am Pract Digest Treat 1955; 6: 558–66
Stock J. Sulfonamide hypersensitivity and acetazolamide. Arch Ophthalmol 1990; 108: 634–5
Newman L, Lay C, O’Connor K, Russell M. Lack of cross-reactivity to sumatriptan in patients allergic to sulfonamides: a retrospective chart review [abstract]. Headache 1999; 39: 372
Patterson R, Bello A, Lefkowtih J. Immunologic tolerability profile of celecoxib. Clin Ther 1999; 21: 2065–73
Data on file, Searle (Pharmacia), 2000
Roujeau J, Kelly J, Naldi L, et al. Medication use and the risk of Stevens-Johnson syndrome or toxic epidermal necrolysis. N Engl J Med 1995; 333: 1600–7
McMorran M, Morawiecka I. Celecoxib (Celebrex): one year later. Can Adv Drug React Newslett 2000; 10: 1–3
Physicians’ Desk Reference, 54th ed. Montvale (NJ): Medical Economics Company, 2000
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Knowles, S., Shapiro, L. & Shear, N.H. Should Celecoxib Be Contraindicated in Patients Who Are Allergic to Sulfonamides?. Drug-Safety 24, 239–247 (2001). https://doi.org/10.2165/00002018-200124040-00001
Published:
Issue Date:
DOI: https://doi.org/10.2165/00002018-200124040-00001